Jeopardy chemestry game
•Download as PPT, PDF•
1 like•240 views
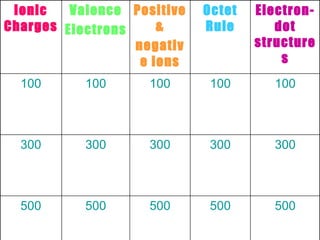
The document discusses various concepts in chemistry including ionic charges, valence electrons, ion formation, the octet rule, and electron dot structures. It contains questions about using ionic charges to determine ion identities, the charges of group 1 and 2 metal ions, the definition and location of valence electrons, the definition of an ion and ionic charge, how negative ions are named, the number of electrons atoms want in the outer shell to be stable, how compounds are formed using the octet rule, what electron dot structures are, what happens with more than 4 electrons, and what valence electrons show in dot structures.
Report
Share
Report
Share
More Related Content
Similar to Jeopardy chemestry game
Similar to Jeopardy chemestry game (20)
Recently uploaded
Model Call Girl in Bikash Puri Delhi reach out to us at 🔝9953056974🔝

Model Call Girl in Bikash Puri Delhi reach out to us at 🔝9953056974🔝9953056974 Low Rate Call Girls In Saket, Delhi NCR
call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️

call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️9953056974 Low Rate Call Girls In Saket, Delhi NCR
9953330565 Low Rate Call Girls In Rohini Delhi NCR

9953330565 Low Rate Call Girls In Rohini Delhi NCR9953056974 Low Rate Call Girls In Saket, Delhi NCR
Recently uploaded (20)
Model Call Girl in Bikash Puri Delhi reach out to us at 🔝9953056974🔝

Model Call Girl in Bikash Puri Delhi reach out to us at 🔝9953056974🔝
Introduction to ArtificiaI Intelligence in Higher Education

Introduction to ArtificiaI Intelligence in Higher Education
call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️

call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️
A Critique of the Proposed National Education Policy Reform

A Critique of the Proposed National Education Policy Reform
9953330565 Low Rate Call Girls In Rohini Delhi NCR

9953330565 Low Rate Call Girls In Rohini Delhi NCR
Call Girls in Dwarka Mor Delhi Contact Us 9654467111

Call Girls in Dwarka Mor Delhi Contact Us 9654467111
Jeopardy chemestry game
- 1. 500 500 500 500 500 300 300 300 300 300 100 100 100 100 100 Electron-dot structures Octet Rule Positive & negative ions Valence Electrons Ionic Charges
- 2. 100 Ionic Charges Question 1.- What can be used to determine the ionic charges of the ions?