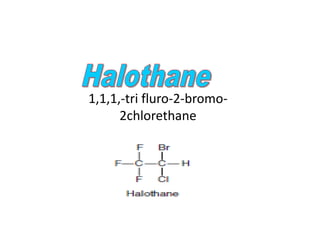
Halothine
- 2. Halothane was not chemically inert and prolonged usage of this agent damage metal rubber and some plastic component of the anesthetic circuit Halothane is susceptible to spontaneous oxidation and photochemical decomposition Requiring storage in tinted glass bottle (amber glass bottle) containing 0.01% thymol ( it renders its stability)
- 3. Colorless liquid, relatively pleasant smell, non irritant , decomposed by light in to hydrochloric acid (HCL), hydrobromic acid (Hbr), chloride (CL-), bromide (Br-). Potent with a MAC of 0.75%. Carbon fluoride is responsible for non- flammable and explosive nature.
- 4. Induction The potency and relative lack of irritation favor the use of halothane for rapid smooth inhalation induction of anesthesia especially when it is administered together with 60-70% N₂O.
- 5. Halothane has a relatively low blood gas solubility coefficient of 2.5 and thus Induction of anesthesia is relatively fast in pediatrics However it may take at least 30min for the alveolar concentration to reach 50% of the inspired conc. This is slower than for Enflurane or Sevoflurane
- 6. As with all volatile anesthetics it is customary to use the techniques of administration of a higher partial pressure of anesthetic (PI) than the alveolar concentration (PA) (over pressurization) Induce halothane anesthesia with concentration 2-3× higher than the MAC value . The inspired value can be reduced when a stable level of anesthesia has been achieved.
- 7. For halothane MAC is almost -1.1% in neonate. -0.9% in infants. -0.9% at 1-2 years. -0.75% at 4-5 years. -0.65% at 80 yrs.
- 8. • Recovery from halothane is slower compared to newer drugs( induction is also slower) because of its higher tissue gas coefficient During awakening after halothane anesthesia patient remain drowsy for several hours. Because of the reactive metabolite bromide This is due to higher solubility in brain,
- 9. The greater solubility of halothane in those tissues increase the amount of halothane that accumulate during anesthesia Increase the time it takes to clear halothane from those compartments after administration is discontinued Re-Distribution Delayed awakening
- 10. Cardiovascular effects Potent direct myocardial depressant effect The most prominent circulatory effect of halothane is dose dependent arterial hypotension Decreased HR and coronary blood flow Slow conduction to AV node lead to Bradycardia
- 11. 1/18/2019 11
- 12. During controlled ventilation halothane is associated with dose dependent depression of COP by decreasing myocardial contractility (vasodilatation) thus there is reduction of ABP.
- 13. The hypotensive effect of halothane is augmented by reduction in HR Antagonism of bradycardia with atropine usually leads to increased arterial BP The reduction in myocardial contractility and low HR leads to reduction in myocardial oxygen demand and coronary blood flow So halothane is advantageous in patients with coronary artery disease. Because of reduced oxygen demand caused by low HR and decreased
- 14. • The depressant effect of halothane on COP is aggravated in the presence of β- blocker • Inadequate anesthesia or exogenous administration of CA’s increases myocardial sensitization leading to myocardial dysarrythmia and also cardiac arrest • During local infiltration with adrenaline containing local anesthetic, caution should be taken
- 15. -Over dosage of halothane causes bradycardia and hypotension ,so treat with atropine and discontinue halothane. Guidelines Avoid hypoxemia and hypercapnia Avoid concentration of adrenaline greater than 1:100,000 Avoid a dosage in adults exceeding 10ml of 1:100,000 adrenaline in 10 min. or 30ml/hr.
- 16. Respiratory Effects Alveolar hypoventilation (hypoxia) and arterial hypercapnia occurs in a dose dependent manner during halothane anesthesia in a spontaneously breathing patient so patient breathing should be assisted or controlled.
- 17. 1/18/2019 17
- 18. • Non irritant, pleasant to breath during induction of anesthesia • The respiratory pattern associated with halothane anesthesia is characterized by rapid shallow respiration.
- 19. 1/18/2019 19
- 20. In awake individual hypercapnia does not occur because even small increase in arterial CO₂ stimulates the respiratory drive to increase minute ventilation. Halothane and other volatile anesthetics abolish physiologic mechanism that protect against hypercapnia
- 21. Rapid loss of pharyngeal and laryngeal reflexes might lead to risk of aspiration. Inhibition of salivary and bronchial secretion.
- 22. 1/18/2019 22
- 23. PaCO₂ increases as the depth of anesthesia increases and patient becomes hypoxic(PaCO2 increase PaO₂ decrease) Decrease in mucociliary function which may persist several hours after halothane anesthesia. This may contribute to post op. hypoxia and atelectasis
- 24. 1/18/2019 24
- 25. CNS Cerebral vasodilatation Increase CBF Increase ICP
- 26. Other systems Potentiate action of NDMR by direct relaxation of skeletal muscle. Trigger malignant hyperthermia. Post op. shivering is common in old age (this increase O₂ requirement ⇒300% and result in hypoxemia unless O₂ is
- 27. GI motility is inhibited – paralytic illus PONV are seldom severe. Decrease HBF this is proportional to COP. Hepatic artery vasoconstriction
- 28. Biotransformation Major route of elimination is lung 80% 10-20% is bio-transformed in the liver Small amount diffuse out through skin
- 29. Hepatic biotransformation occurs through the cytochrome P450 system resulting in the release of bromide and chloride ion and the formation of fluorine containing compounds mostly trifluoroacetic acid Many believe that the hepatic complication of halothane results from its biotransformation
- 30. Emergence Awakening is prompt but may take several hours because of higher solubility of halothane in brain, muscle, fat increase accumulation Clearing time is increased after discontinuation PONV
- 31. Halothane hepatitis Defined as the appearance of liver damage within 28 days of halothane exposure in a person in whom other known causes of liver disease have been excluded. Approximately 20% of halothane is metabolized in liver by the oxidative pathway, the end product excreted in urine.
- 32. The major metabolites are bromine, chlorine, trifluoroacetic acid and trifluoroacetyl-ethanol amide. A small proportion of halothane may undergo reductive metabolism, particularly in the presence of hypoxemia and when the hepatic microsomal enzymes has been stimulated by enzyme inducing agents such as phenobarbitone
- 33. Reductive metabolism may result in the formation of reactive metabolite Chlorine when absorbed or contact with dry soda lime and will get broken down to BCDFE (2-bromo-2- chloro-1,1-difluoroethene) which has organ toxicity in animal models Halothane hepatitis The product of reductive metabolic pathway are more toxic than those produced by oxidative pathway.
- 34. In mild cases halothane increase enzyme of liver, but in several cases halothane hepatitis and liver necrosis Incidence is 1:35,000
- 35. This is supported by the fact that the risk of post operative liver dysfunction is increased in the presence of Obesity which increase hypoxia and greater storage of halothane Hypoxemia A short interval b/n administrations of the drug Enzyme induction produced by drugs e.g.- phenobarbitone, phenytoin
- 36. • The incidence of hepatic toxicity is high in obese middle age women but less in pediatric (halothane is the drug of choice in pediatrics) • As a result of this concern the committee on safety of medicine has made the following recommendations in respect halothane.
- 37. 1. A careful anesthetic history should be taken to determine previous exposure and any previous reaction to halothane. 2. Repeated exposure to halothane with in a period of three months should be avoided unless there are over riding clinical conditions. 3. A history of jaundice or pyrexia after previous exposure to halothane is an absolute C/I to its future use in that patient.
- 38. Precaution • Space occupying lesion • Pheochromocytoma • MHT • History of PPH,APH, hypovolaemic • Unexplained liver dysfunction
- 39. Indication • Induction of anesthesia in children • Maintenance of anesthesia • In air way obstructions
- 40. Advantage Disadvantage -rapid and smooth induction -poor analgesia -effective in low conc. -CA’s induced arrhythmia -Minimal stimulation of salivary and bronchial secretions -post op. shivering -bronchodilation -liver toxicity -Muscle relaxation -slow recovery -relative rapid recovery, cheap -Halothane hepatitis -less stable
- 41. • Dose – induction-2-3% in adult -1-2% child - maintenance-0.8%-1.5%
- 42. 1/18/2019 42