Chromatographic Column chemistry
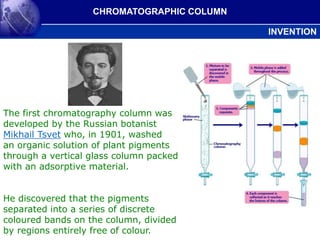
- 1. The first chromatography column was developed by the Russian botanist Mikhail Tsvet who, in 1901, washed an organic solution of plant pigments through a vertical glass column packed with an adsorptive material. He discovered that the pigments separated into a series of discrete coloured bands on the column, divided by regions entirely free of colour. CHROMATOGRAPHIC COLUMN INVENTION
- 2. CHROMATOGRAPHIC COLUMN Details of column
- 3. CHROMATOGRAPHIC COLUMN Details of column polyimid, 350 oC Silica Stationary phase microbore: d < 150 m standard capillary: 150 m < d < 500 m widebore: d > 500 m d
- 4. CHROMATOGRAPHIC COLUMN Stationary Phase - Silica
- 5. CHROMATOGRAPHIC COLUMN Stationary Phase - Silica
- 6. CHROMATOGRAPHIC COLUMN Physical Characteristics of column Default Column Optimum Column† Column Bed Dimensions 150 x 4.6mm 250 x 4.6mm Particle Size 5µm 3* or 5µm Surface Area 200m2/g >200m2/g Pore Size 100Å 100Å Carbon Load 10% 16 - 20% Bonding Type Monomeric Mono- or Polymeric Base Material Silica Silica Particle Shape Spherical Spherical * mobile phase backpressure may be excessive † Optimum Column: Alltima C18™, 5µm, 250 x 4.6mm (Part No. 88056) *Note that the choice may represent a compromise. Here, the “optimum” column for resolution sacrifices speed.
- 7. CHROMATOGRAPHIC COLUMN Physical Characteristics of column Column Dimensions Length and internal diameter of packing bed • Effect on chromatography Short (30-50mm) - short run times, low backpressure Long (250-300mm) - higher resolution, long run times Narrow ( 2.1mm) - higher detector sensitivity Wide (10-22mm) - high sample loading
- 8. CHROMATOGRAPHIC COLUMN Physical Characteristics of column Particle Shape : Spherical or irregular Effect on chromatography Spherical particles offer reduced back pressures and longer column life when using viscous mobile phases like 50:50 MeOH:H2O.
- 9. CHROMATOGRAPHIC COLUMN Physical Characteristics of column Particle Size The average particle diameter, typically 3-20µm Effect on chromatography Smaller particles offer higher efficiency, but also cause higher backpressure. Choose 3µm particles for resolving complex, multi-component samples. Otherwise, choose 5 or 10µm packings.
- 10. CHROMATOGRAPHIC COLUMN Physical Characteristics of column Surface Area Sum of particle outer surface and interior pore surface, in m2/gram Effect on chromatography High surface area generally provides greater retention, capacity and resolution for separating complex, multi- component samples. Low surface area packings generally equilibrate quickly, especially important in gradient analyses.
- 11. CHROMATOGRAPHIC COLUMN Physical Characteristics of column Pore Size Average size of pores or cavities in particles, ranging from 60-10,000Å Effect on chromatography Larger pores allow larger solute molecules to be retained longer through maximum exposure to the surface area of the particles. Choose a pore size of 150Å or less for sample MW 2000. Choose a pore size of 300Å or greater for sample MW > 2000.
- 12. CHROMATOGRAPHIC COLUMN Physical Characteristics of column Bonding Type Monomeric - single-point attachment of bonded phase molecule Si R R (CH2)17CH3Si CH3 CH3 (CH2)17CH3XOH + monomeric bonding Si CH3 X (CH2)17CH3X+ polymeric bonding OH OH O O Si CH3 (CH2)17CH3 Polymeric - multi-point attachment of bonded phase molecule
- 13. CHROMATOGRAPHIC COLUMN Physical Characteristics of column
- 14. CHROMATOGRAPHIC COLUMN Physical Characteristics of column Monomeric - single-point attachment of bonded phase molecule Polymeric - multi-point attachment of bonded phase molecule Effect on chromatography This product presents a very high stability, batch-to-batch reproducibility and good hydrophobic properties. The fact that the silane possesses only one bond with the surface makes this phase less stable at low pH, which may lead to silane hydrolysis and consequently leaching. Effect on chromatography The silica surface is more hydrophobic, has greater stability in strong acidic condition (pH 2, 3) and has a longer lifetime.
- 15. CHROMATOGRAPHIC COLUMN Physical Characteristics of column Carbon Load Amount of bonded phase attached to base material, expressed as %C Effect on chromatography Higher carbon loads generally offer greater resolution and longer run times. Low carbon loads shorten run times and many show a different selectivity.
- 16. CHROMATOGRAPHIC COLUMN Physical Characteristics of column End capping “Capping” of exposed sialons with short hydrocarbon chains after the primary bonding step Effect on chromatography End capping reduces peak-tailing of polar solutes that interact excessively with the otherwise exposed, mostly acidic silanols. Non-end capped packings provide a different selectivity than do end capped packings, especially for such polar samples.
- 17. CHROMATOGRAPHIC COLUMN Physical Characteristics of column End capping “Capping” of exposed sialons with short hydrocarbon chains after the primary bonding step
