Hemophagocytic lymphohistiocytosis (hlh), Langerhans cell histiocytosis dr vijitha
- 2. Hemophagocytic lymphohistiocytosis (HLH) & Langerhans cell histiocytosis PRESENTER –Dr VIJITHA A S
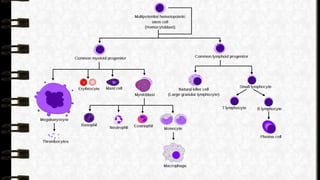
- 3. • Histiocyte is a normal immune cell that is found in bone marrow, blood, skin, liver, lungs, lymph glands and spleen. These include monocytes, macrophages, and dendritic cells • Childhood Histiocytosis is a generic name for a group of syndromes characterized by prominent proliferation or accumulation of histiocytes (monocyte, macrophage system of bone marrow-myeloid orgin)
- 5. LANGERHANS CELL HISTIOCYTOSIS INTRODUCTION Langerhans cell histiocytosis (LCH) is a rare clonal disease of the monocyte-macrophage system characterized by uncontrolled proliferation and accumulation of CD1a+/ CD207+ dendritic cells (DCs) as a result of continuous immune stimulation
- 6. Epidemiology • can occur at any age peak incidence between 1 - 4 years • Male:female 1.4 : 1 • Incidence is rare ; 4-10 /1million population
- 7. Histiocytosis X • Langerhans cell histiocytosis (LCH) previously called as histiocytosis X includes clinical entities 1. Eosinophilic granuloma 2. Hand schuller – Christian disease 3. Letterer siwe disease
- 8. Eosinophilic granuloma • Multifocal LCH • Eosinophilic granuloma of bone • Eosinophilic granuloma of lung
- 9. Hand schuller – Christian disease • Multifocal LCH • Triad of - 1. Calvarial bone defect(osteophytic bone lesion) 2. Exophthalmos 3. Diabetes Insipudus
- 10. Letterer siwe disease • Multifocal multisystem involvement: • <2years of age • Aggressive systemic disorder; rapidly fatal • c/f – fever, anemia, thrombocytopenia, pulmonary infiltrate, enlargement of spleen, liver, and lymph node
- 11. CLASSIFICATION • At present, LCH is classified into three distinct forms: 1. single-system single site (SS-s) 2. single-system multi-site (SS-m) 3. multisystem type (MS)
- 14. DIAGNOSIS • The diagnosis of LCH is based on histological criteria established by the Histiocyte Society in 1987 • Diagnosis = histological examination with immunophenotyping • The identification of characteristic cells + presence of markers( CD1a and/or CD207 (Langerin) definitive diagnosis of LCH. • The Birbeck granule - is a LCH-specific tennis racquet-shaped organelle detected by electron microscopy. • The presence of Birbeck granules, once considered the “gold standard” in the diagnostics, • Currently they have been replaced by langerin, which is a cell-surface receptor that induces formation of Birbeck granules
- 15. Birbeck granule
- 16. Treatment • Systemic therapy is indicated for all patients with SS-m and MS –LCH as well as for special localizations • Standard two drug regimen - vinblastine and prednisone consisting of an initial intensive phase for 6–12 weeks, followed by • maintenance therapy for a total treatment duration of at least 12 months, is recommended
- 17. Treatment… • A single bone lesion involving the frontal, parietal, occipital bones and any other bones - curettage/curettage with a local injection of methylprednisolone • skull lesions (mastoid, temporal or orbital) should be treated with chemotherapy • Vertebral or femoral bone lesions at risk for collapse require - isolated radiotherapy • CNS lesions (enlargement of the hypothalamus and the pituitary gland or grey and white matter, the choroid plexus, and leptomeningeal lesions) - systemic chemotherapy
- 18. Hemophagocytic lymphohistiocytosis (HLH) • INTRODUCTION • Hemophagocytosis-: Phagocytosis by macrophages of erythrocytes, leucocytes, platelets & their precursors in BM & other tissues • Rare, Aggressive and life threatening • It is associated with various infections, autoimmune diseases malignancies, and immunocompromised states • MC age group 0- 18 months, but it can present in all age groups
- 19. TERMINOLOGY (HLH) • In 1952 Farquhar & Claireaux - Familial Hemophagocytic reticulosis Other terms - primary - secondary - Macrophage activation syndrome
- 20. DEFENITION HLH • Benign proliferation of mature histiocytes and uncontrolled phagocytosis of the platelet, erythrocytes, lymphocytes, and their hematopoietic precursors in the bonemarrow & other tissues
- 21. Epidemiology • Incidence - 1 in 3000 children • Infants are most commonly affected with highest incidence in <3 months. • Male to female ratio is 1:1 • Up to 25% of HLH cases have been reported to be familial.
- 22. ETIOLOGY
- 23. PRIMARY HLH • Incidence 1.2/1000000 per year • Occurs in children with genetic abnormalities of the cytotoxic function of natural killer (NK) cells and T cells • Several genetic defects in primary HLH recently identified which include - perforin (PRF1) Munc13-4 (UNC13D) syntaxin 11 (STX11), Munc18-2 (STXBP2)
- 24. • HLH is also a common manifestation of certain - pigmentary disorders, - X-linked lymphoproliferative diseases, - Epstein-Barr virus (EBV) susceptibility disorders, - certain CDC42 mutations & activating mutations in NLRC4. • All of these disorders can be collectively termed genetic HLH diseases. • Term primary HLH can also be used as a category for some or all of these diseases.
- 26. HLH IN PRIMARY IMMUNODEFICIENCIES HLH develops in various primary immunodeficiencies, • Chediak–Higashi syndrome (CHS) • Griscelli syndrome (GS) type 2 • Hermansky–Pudlak syndrome (HPS) type 2 • X-linked lymphoproliferative disease
- 29. Hermansky–Pudlak syndrome (HPS) type 2 • decreased pigmentation (albinism) • visual impairment • blood platelet dysfunction with prolonged bleeding. • Some patients have lung fibrosis, colitis, or an abnormal storage of a fatty-like substance (ceroid lipofuscin) in various tissues of the body.
- 30. HLH
- 31. • HLH is a syndrome of excessive inflammation and tissue destruction due to abnormal immune activation • This excessive inflammation is caused by a failure of normal down regulation of activated macrophages and lymphocytes • Macrophage activation results in secretion of excessive amounts of cytokines leading to severe tissue damage and organ failure PATHOGENESIS
- 32. • Cytokine storm is due to the persistent activation of macrophages, NK cells and CTLs leading on to excessive cytokine production - responsible for multiorgan failure and the high mortality • Cytokines found at extremely high levels in the plasma of patients with HLH - interferon gamma (IFN-γ) - tumour necrosis factor alpha (TNF-α) - interleukins (IL) - IL-6, IL-10, IL-12, soluble IL-2 receptor (CD25)
- 33. PATHOGENESIS… • Three intertwined cellular processes: 1. Genetic failure or transient impairment of cytotoxic cells (CD8-T-Cells and NK Cells) to induce cell lysis when encountered with an antigen presenting cell 2. Hyperactivation of lymphocytes and macrophages due to the failure to terminate an immune response leads to host tissue destruction ( Ex. Macrophages get out of control and engulf host cells including RBC, Platelets, and WBC causing cytopenias thus the name hemo (blood) phagocytosis (eating). 3. Excessive proliferation of inflammatory cytokines resulting in further immune stimulation and progressive multi-organ dysfunction •
- 34. PATHOGENESIS
- 39. CLINICAL FEATURES • From various studies conducted in India, the predominant presenting features - prolonged fever and hepatosplenomegaly. - CNS symptoms (36%) - Anemia (Hb <9gm/ dL)97% -Thrombocytopenia (platelets <1, 00,000/mm3)72% • Biochemical markers - hyperferritinemia 97% - hypofibrinogenemia & high LDH in 92%. • Bone marrow examination -hemophagocytosis in 84%. • Infectious agents were identified in 42% children, with viruses accounting for two thirds of them
- 40. CLINICAL FEATURES…. • Patients with HLH are at risk of developing posterior reversible encephalopathy syndrome (PRES). • Respiratory abnormalities may lead to an urgent need for ventilatory support and death from acute respiratory distress syndrome (ARDS). Deteriorating respiratory function may be due to worsening of the HLH or due to an infection • STXBP2 mutations - hypogammaglobulinemia, severe diarrhea, bleeding and sensorineural hearing loss.
- 41. INVESTIGATIONS • Initial tests • CBC with differentials • Coagulation profile • Serum ferritin • LFT,RFT • S.triglycerides • Full septic • Serology & PCR • BM evaluation • ECG,EEG,CXR • MRI,CT, abdomen US
- 42. INVESTIGATIONS…. Specialized tests Immunological profile Genetic testing Human leukocyte antigen testing
- 44. DIAGNOSIS OF HLH
- 46. Differential diagnosis • Systemic inflammatory response syndrome (SIRS) due to other causes. • Macrophage activation syndrome (mas) • Infection/sepsis • Liver disease/liver failure • Multiple organ dysfunction syndrome • Encephalitis • Autoimmune lymphoproliferative syndrome (ALPS) • Drug reaction with eosinophilia and systemic symptoms (DRESS) • Kawasaki disease • Cytophagic histiocytic panniculitis, • Thrombotic thrombocytopenic purpura (TTP) • Hemolytic uremic syndrome (HUS) • Transfusion-associated graft-versushost disease (ta-gvhd).
- 48. TREATMENT • The aim of therapy - whether for primary or secondary disease is to interrupt the amplification cascades of cytokines and suppress the hyper inflammation, with a secondary aim of killing antigen presenting cells to remove ongoing stimulus for inflammatory responses
- 49. HLH TREATMENT • Until 1994 , HLH therapy ineffective with 90% fatalities • HLH – 94 - first international study on HLH treatment - included combination of chemotherapy, immunotherapy and steroids as well as antibiotics and antiviral drugs followed by stem cell transplant • Two phases – initial phase (8weeks) – continuation phase • Survival rate – 55% at median follow up up of 3.1 years
- 50. HLH 2004 • Cyclosporin A started at the onset of therapy instead at week 9
- 51. HLH TREATMENT 1. Immediate goals 2. Long term Goals
- 53. LONG TERM GOALS Replace the defective immune system • Allogenic haematopoietic stem cell transplantation • Best overall cure rate • Needed for patient with • Genetic mutaions diagnosed or family history • Patients who responded poorly with initial eight weeks of chemotherapy • Patients with CNS disease • Non –myeloablative or reduced intensity transplantation- use of fludarabine, melphalan and alemtuzumab (anti-D52 antibody)before transplantation improves survival rates
- 54. Other treatment approaches Anti thymocyte globulin IVIG Rituximab (EBV HLH) HIT-HLH trial – combined use of ATG,Etoposide,intrathecal methotrexate &hydrocortisone)
- 55. HLH TREATMENT • Dexamethasone – 10 mg/kg/day first 2 weeks – 5mg/kg/day week 3 and 4 –2.5 mg/kg/day for week 5 and 6 – 1.25 mg/kg/day for week 7 – Tapering to zero over the 8th week • Etoposide (VP-16) – 150 mg/m2 IV twice weekly for first two weeks
- 56. Points to Remember • Hemophagocytic lymphohistiocytosis (HLH) is a frequently fatal but underdiagnosed condition. • Clinical features mimic many illnesses. • Fever, lymphadenopathy, hepatosplenomegaly, rash along with bicytopenia, elevated liver enzymes and ferritin should make one suspect HLH. • Well defined criteria help in making a definitive diagnosis of HLH. • Corticosteroids, etoposide and cyclosporine A form the basis of the treatment. • Hematopoietic stem cell transplant will be needed in primary cases to correct the underlying immune defect and to prevent recurrence. • Supportive care is essential
- 57. THANK YOU