Preformulation introduction and organoleptic properties.pptx
•Download as PPTX, PDF•
0 likes•502 views
..Preformulation introduction and organoleptic properties.pptx..
Report
Share
Report
Share
Recommended
Recommended
DOCUMENTATION IN PHARMACEUTICAL INDUSTRY, BY Dr. UMESH KUMAR SHARMADocumentation in pharmaceutical industry, by dr. umesh kumar sharma and anu m...

Documentation in pharmaceutical industry, by dr. umesh kumar sharma and anu m...Dr. UMESH KUMAR SHARMA
More Related Content
What's hot
DOCUMENTATION IN PHARMACEUTICAL INDUSTRY, BY Dr. UMESH KUMAR SHARMADocumentation in pharmaceutical industry, by dr. umesh kumar sharma and anu m...

Documentation in pharmaceutical industry, by dr. umesh kumar sharma and anu m...Dr. UMESH KUMAR SHARMA
What's hot (20)
Current Good Manufacturing Practices(cGMP) and industrial management

Current Good Manufacturing Practices(cGMP) and industrial management
Documentation in pharmaceutical industry, by dr. umesh kumar sharma and anu m...

Documentation in pharmaceutical industry, by dr. umesh kumar sharma and anu m...
Pilot plan scale up for semisolid and parenteral by Khushboo kunkulol

Pilot plan scale up for semisolid and parenteral by Khushboo kunkulol
Similar to Preformulation introduction and organoleptic properties.pptx
Similar to Preformulation introduction and organoleptic properties.pptx (20)
Pharmacology Theory_Introduction & Routes of Administration.pptx

Pharmacology Theory_Introduction & Routes of Administration.pptx
Recently uploaded
❤️Amritsar Escort Service☎️98151-129OO☎️ Call Girl service in Amritsar☎️ Amritsar Call Girls Service ☎️ Call Girls In Amritsar BEST Call Girls in Amritsar Escort Service provide Cute Nice sweet and Sexy Models in beautiful Amritsar city cash in hand to hand call girl in Amritsar and Amritsar escorts. HOT & SEXY MODELS // COLLEGE GIRLS IN Amritsar AVAILABLE FOR COMPLETE ENJOYMENT WITH HIGH PROFILE INDIAN MODEL AVAILABLE HOTEL & HOME ★ SAFE AND SECURE HIGH CLASS SERVICE AFFORDABLE RATE ★ 100% SATISFACTION,UNLIMITED ENJOYMENT. ★ All Meetings are confidential and no information is provided to any one at any cost.
★ EXCLUSIVE Profiles Are Safe and Consensual with Most Limits Respected
★ Service Available In: - HOME & 24x7 :: 3 * 5 *7 *Star Hotel Service .In Call & Out call
Services :
★ A-Level (5 star escort)
★ Strip-tease
★ BBBJ (Bareback Blowjob)Receive advanced sexual techniques in different mode make their life more pleasurable.
★ Spending time in hotel rooms
★ BJ (Blowjob Without a Condom)
★ Completion (Oral to completion)
★ Covered (Covered blowjob Without a Condom)Amritsar Call Girls❤️Amritsar Escort Service☎️98151-129OO☎️ Call Girl service in Amritsar☎️ Amri...

❤️Amritsar Escort Service☎️98151-129OO☎️ Call Girl service in Amritsar☎️ Amri...shallyentertainment1
❤️ Call Girls service In Panchkula☎️9815457724☎️ Call Girl service in Panchkula☎️ Panchkula Call Girls Service ☎️ Call Girls In Panchkula BEST CALL GIRL ESCORTS SERVICE IN PANCHKULA CALL WATTSAPP 9815457724 THE MOST BEAUTIFUL INDEPENDENT ESCORT CALL GIRL SERVICE In Panchkula WE ARE PROVIDING GENUINE CALL GIRL SERVICE
I AM A a NATURAL BRUNETTES, SLIM BODY, NATURAL LONG HAIR AND ALL TYPE OF HAIR IS A NATURAL BRUNETTE IN THE MOST BEAUTIFUL MODELS INDEPENDENT ESCORT GIRL I AM A NATURAL BRUNETTE WITH ROOM AND HOTEL AND A NATURAL BRUNETTE WITH A BODY MADE FOR SIN AND ALL TYPE OF ME ALL THE TIME
I SEND YOU A HAIR, VERY SOCIABLE AND FUNNY, READY TO ENTERTAIN TO ENTERTAIN U AND MAKE FORGET ABOUT TO AGET ENTERTAINMENT YOU AND MAKE FORGET ABOUT ALL THE PROBLEMS. LET'S HAVE A WONDERFUL TIME TOGETHER AND FORGET ABOUT EVERYTHING ALL TYPE SERVICE ENJOYMENT SAFE AND SECURE IN CALL OUT CALL HOME AND HOTEL ANYTIME AVAILABLE
AND ALL TYPE SERVICE ENJOYMENTPANCHKULA INDEPENDENT BEST CALL GIRL ESCORTS SERVICE IN PANCHKULA INDEPENDENT CALL GIRLS❤️ Call Girls service In Panchkula☎️9815457724☎️ Call Girl service in Panchku...

❤️ Call Girls service In Panchkula☎️9815457724☎️ Call Girl service in Panchku...Rashmi Entertainment
🍑👄Ludhiana Escorts Service☎️98157-77685🍑👄 Call Girl service in Ludhiana☎️Ludhiana Call Girls Service 🍑👄 Call Girls In Ludhiana Book Now :- 98157-77685
Our agency presents a selection of young, charming call girls available for bookings at Oyo Hotels. Experience high-class escort services at pocket-friendly rates, with our female escorts exuding both beauty and a delightful personality, ready to meet your desires. Whether it's Housewives, College girls, Russian girls, Muslim girls, or any other preference, we offer a diverse range of options to cater to your tastes.
We provide both in-call and out-call services for your convenience. Our in-call location in Delhi ensures cleanliness, hygiene, and 100% safety, while our out-call services offer doorstep delivery for added ease.
We value your time and money, hence we kindly request pic collectors, time-passers, and bargain hunters to refrain from contacting us. l Ludhiana, Majestic Grand Hotel, Ramada by Wyndham Ludhiana City Centre, Park Plaza Ludhiana, Windsor Fountain, G.T Road Ludhiana escort all Ludhiana service Russian available model female girls in Ludhiana VIP Lo price personal Ludhiana off class call girls payment high profile model and female escort 70% Off On Your First Booking Ludhiana Call Girls Service Cash Payment
Welcome to DILPREET Ludhiana Call Girl Service, the Trusted call girl agency around. We Offer 70% Discount On Your First Booking For Ludhiana Call Girls Service Cash Payment is available.🍑👄Ludhiana Escorts Service☎️98157-77685🍑👄 Call Girl service in Ludhiana☎️Ludh...

🍑👄Ludhiana Escorts Service☎️98157-77685🍑👄 Call Girl service in Ludhiana☎️Ludh...dilpreetentertainmen
❤️ Escorts Service in Bangalore ☎️81279-924O8☎️ Call Girl service in Bangalore☎️ Bangalore Call Girls Service ☎️ Call Girls In Bangalore BEST Call Girls in Bangalore Escort Service provide Cute Nice sweet and Sexy Models in beautiful Bangalore city cash in hand to hand call girl in Bangalore and Bangalore escorts. HOT & SEXY MODELS // COLLEGE GIRLS IN BANGALORE AVAILABLE FOR COMPLETE ENJOYMENT WITH HIGH PROFILE INDIAN MODEL AVAILABLE HOTEL & HOME ★ SAFE AND SECURE HIGH CLASS SERVICE AFFORDABLE RATE ★ 100%SATISFACTION,UNLIMITED ENJOYMENT. ★ All Meetings are confidential and no information is provided to any one at any cost. ★ EXCLUSIVE Profiles Are Safe and Consensual with Most Limits Respected ★ Service Available In: - HOME & 24x7 :: 3 * 5 *7 *Star Hotel Service .In Call & Out call Services :
★ A-Level (5 star escort)
★ Strip-tease
★ BBBJ (Bareback Blowjob)Receive advanced sexual techniques in different mode make their life more pleasurable.
★ Spending time in hotel rooms
★ BJ (Blowjob Without a Condom)
★ Completion (Oral to completion)
★ Covered (Covered blowjob Without a Condom)-Bangalore Call Girls❤️Bangalore Call Girls ☎️81279-924O8☎️ Call Girl service in Bangalore☎️ Bangalore Call Girls Service ☎️ Call Girls In Bangalore
❤️ Escorts Service in Bangalore ☎️81279-924O8☎️ Call Girl service in Bangalor...

❤️ Escorts Service in Bangalore ☎️81279-924O8☎️ Call Girl service in Bangalor...chandigarhentertainm
Recently uploaded (19)
👉Bangalore Call Girl Service👉📞 6378878445 👉📞 Just📲 Call Manisha Call Girls Se...

👉Bangalore Call Girl Service👉📞 6378878445 👉📞 Just📲 Call Manisha Call Girls Se...
💞 Safe And Secure Call Girls Mysore 🧿 9332606886 🧿 High Class Call Girl Servi...

💞 Safe And Secure Call Girls Mysore 🧿 9332606886 🧿 High Class Call Girl Servi...
💸Cash Payment No Advance Call Girls Nagpur 🧿 9332606886 🧿 High Class Call Gir...

💸Cash Payment No Advance Call Girls Nagpur 🧿 9332606886 🧿 High Class Call Gir...
💞 Safe And Secure Call Girls gaya 🧿 9332606886 🧿 High Class Call Girl Service...

💞 Safe And Secure Call Girls gaya 🧿 9332606886 🧿 High Class Call Girl Service...
❤️Amritsar Escort Service☎️98151-129OO☎️ Call Girl service in Amritsar☎️ Amri...

❤️Amritsar Escort Service☎️98151-129OO☎️ Call Girl service in Amritsar☎️ Amri...
❤️ Call Girls service In Panchkula☎️9815457724☎️ Call Girl service in Panchku...

❤️ Call Girls service In Panchkula☎️9815457724☎️ Call Girl service in Panchku...
TIME FOR ACTION: MAY 2024 Securing A Strong Nursing Workforce for North Carolina

TIME FOR ACTION: MAY 2024 Securing A Strong Nursing Workforce for North Carolina
❤️ Zirakpur Call Girl Service ☎️9878799926☎️ Call Girl service in Zirakpur ☎...

❤️ Zirakpur Call Girl Service ☎️9878799926☎️ Call Girl service in Zirakpur ☎...
💸Cash Payment No Advance Call Girls Kanpur 🧿 9332606886 🧿 High Class Call Gir...

💸Cash Payment No Advance Call Girls Kanpur 🧿 9332606886 🧿 High Class Call Gir...
💸Cash Payment No Advance Call Girls Hyderabad 🧿 9332606886 🧿 High Class Call ...

💸Cash Payment No Advance Call Girls Hyderabad 🧿 9332606886 🧿 High Class Call ...
👉Indore Call Girl Service👉📞 7718850664 👉📞 Just📲 Call Anuj Call Girls In Indor...

👉Indore Call Girl Service👉📞 7718850664 👉📞 Just📲 Call Anuj Call Girls In Indor...
🍑👄Ludhiana Escorts Service☎️98157-77685🍑👄 Call Girl service in Ludhiana☎️Ludh...

🍑👄Ludhiana Escorts Service☎️98157-77685🍑👄 Call Girl service in Ludhiana☎️Ludh...
💞 Safe And Secure Call Girls Coimbatore 🧿 9332606886 🧿 High Class Call Girl S...

💞 Safe And Secure Call Girls Coimbatore 🧿 9332606886 🧿 High Class Call Girl S...
❤️ Escorts Service in Bangalore ☎️81279-924O8☎️ Call Girl service in Bangalor...

❤️ Escorts Service in Bangalore ☎️81279-924O8☎️ Call Girl service in Bangalor...
Making change happen: learning from "positive deviancts"

Making change happen: learning from "positive deviancts"
💸Cash Payment No Advance Call Girls Pune 🧿 9332606886 🧿 High Class Call Girl ...

💸Cash Payment No Advance Call Girls Pune 🧿 9332606886 🧿 High Class Call Girl ...
Call Girls In Indore 💯Call Us 🔝 9987056364 🔝 💃 Independent Escort Service Ind...

Call Girls In Indore 💯Call Us 🔝 9987056364 🔝 💃 Independent Escort Service Ind...
Preformulation introduction and organoleptic properties.pptx
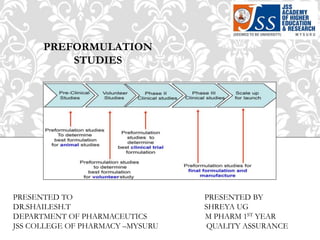
- 1. PREFORMULATION STUDIES PRESENTED TO PRESENTED BY DR.SHAILESH.T SHREYA UG DEPARTMENT OF PHARMACEUTICS M PHARM 1ST YEAR JSS COLLEGE OF PHARMACY –MYSURU QUALITY ASSURANCE
- 2. 1.Introduction 2.Defination 3.Objective and concepts of preformulation 4.Preformulation parameters 5.Benefits 5.Organoleptic properties 6.References CONTENTS
- 3. Prior to the development of any dosage form , it is essential that the physical chemical and fundamental properties of the drug molecule must be investigated. Preformulation is the first step in the rational development of a drug substance. It’s a branch of pharmaceutical sciences that utilizes biopharmaceutical principles to find the pharmaceutical properties of the drug molecule. INTRODUCTION
- 4. Investigation of physciochemical properties of a new drug compond that could affect drug performance and development of an efficious dosage form. It is the phase of reasearch and development characterized physical and chemical properties of a drug molecule in order to develop safe effective and stable dosage form. DEFINITION
- 5. The preformulation investigations confirm that there are no significant barriers to the compound’s development as a marketed drug. To establish the necessary physicochemical parameters of a new drug substance To establish compatibility of drug with excipients The formulation scientist uses these informations to develop dosage forms OBJECTIVES AND CONCEPTS OF PREFORMULATION
- 6. Bulk characteristic 1. Organoleptic properties 2. Particle size & surface area 3. Crystallinity & polymorphism Solubility Analysis 1. Aqueous solobility 2. Dissolution Stability analysis 1. Solid state and solution state stability 1. Oxidation 2. Hydrolysis 3. Photolysis 4. polymerization Physial characteristic Chemical characteristic PREFORMULATION PARAMETERS
- 7. Gives direction for development of formulation in choice of dosage form , excipents, compostion, physical compatibility Helps in adjustment of pharmacokinetic biopharmaceutical properties Support for process development of drug substance Support for development of PAT(Process Analytical Technology) BENEFITS
- 8. Color Odour Taste Off -white Pungent Acidic Cream - Yellow Sulfurous Bitter Shiny Fruity Sweet Tan Aromatic Tasteless Odourless Bland Intense Organoleptic properties are the aspects that can be experienced through one’s senses like taste, smell, sight, touch. The color, odour and taste of the new drug must be recorded using descriptive terminology. ORGANOLEPTIC PROPERTIES
- 9. color is generally a function of drug’s inherent chemical structure relating to a certain level of unsaturation. Color intensity relates to extend of conjugated unsaturation as well as the presence of chromophores. The substance may exhibit an inherent odor characteristic of major functional group present. Taste is considered as unpalatable, consideration is given to the use of a less soluble chemical form of the drug Both of these can be suppressed by using appropriate flavours and excipients. Color Odour &Taste ORGANOLEPTIC PROPERTIES
- 10. Leon Lachman , Lieberman H A and Joseph L K: The Theory and Practice of Industrial Pharmacy. Varghese publishing house, Bombay. Vilegave, K., Vidyasagar, G. and Chandankar, P., 2013. Preformulation studies of pharmaceutical new drug molecule and products: An Overview. The American Journal of Pharmacy. Verma, G. and Mishra, M.K., 2016. Pharmaceutical preformulation studies in formulation and development of new dosage form: A review. Int. J. Pharma Res. Rev, 5(10). Jain NK and Sharma SN: A Text book of professional pharmacy. Brahmankar DM and Jaiswal SB: Absorption of drugs in:. Biopharmaceutics and Pharmacokinetics A treatise. REFERENCES
- 11. THANK YOU