Revisiting PD-L1 as an Immunotherapy Biomarker Across the Cancer Spectrum: Current and Emerging Standards of Testing, Scoring, and Assay Interpretation
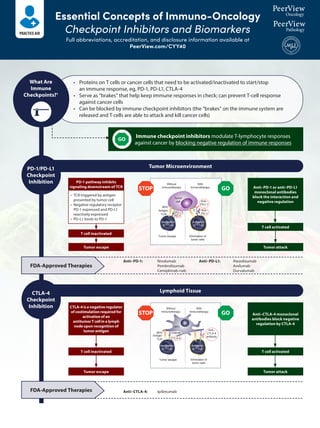
- 1. Essential Concepts of Immuno-Oncology Checkpoint Inhibitors and Biomarkers Full abbreviations, accreditation, and disclosure information available at PeerView.com/CYY40 Tumor Microenvironment Lymphoid Tissue Immune checkpoint inhibitors modulate T-lymphocyte responses against cancer by blocking negative regulation of immune responses PD-1 pathway inhibits signaling downstream of TCR • TCR triggered by antigen presented by tumor cell • Negative regulatory receptor PD-1 expressed and PD-L1 reactively expressed • PD-L1 binds to PD-1 T cell inactivated T cell inactivated T cell activated T cell activated Anti–PD-1 or anti–PD-L1 monoclonal antibodies block the interaction and negative regulation Anti–CTLA-4 monoclonal antibodies block negative regulation by CTLA-4 CTLA-4 is a negative regulator of costimulation required for activation of an antitumor T cell in a lymph node upon recognition of tumor antigen What Are Immune Checkpoints?1 PD-1/PD-L1 Checkpoint Inhibition CTLA-4 Checkpoint Inhibition FDA-Approved Therapies FDA-Approved Therapies Without Immunotherapy With Immunotherapy MHC Antigen TCR PD-1 PD-L1 Anti– PD-L1 Anti– PD-1 Tumor cell Tumor escape Inactivation of T Cell Activation of T Cell Elimination of tumor cells Without Immunotherapy With Immunotherapy MHC CD80/86 CTLA-4 Anti– CTLA-4 antibody APC Antigen TCR Inactivation of T Cell Activation of T Cell Tumor escape Elimination of tumor cells STOP STOP GO GO GO Tumor escape Tumor escape Tumor attack Tumor attack Anti–PD-1: Nivolumab Pembrolizumab Cemiplimab-rwlc Anti–CTLA-4: Ipilimumab Anti–PD-L1: Atezolizumab Avelumab Durvalumab • Proteins on T cells or cancer cells that need to be activated/inactivated to start/stop an immune response, eg, PD-1, PD-L1, CTLA-4 • Serve as “brakes” that help keep immune responses in check; can prevent T-cell response against cancer cells • Can be blocked by immune checkpoint inhibitors (the “brakes” on the immune system are released and T cells are able to attack and kill cancer cells)
- 2. Essential Concepts of Immuno-Oncology Checkpoint Inhibitors and Biomarkers Full abbreviations, accreditation, and disclosure information available at PeerView.com/CYY40 1. Adapted from: Ribas A, Wolchock JD. Science. 2018;359:1350-1355. 2. Mayeux R. NeuroRx. 2004;1:182-188. 3. Naylor S. Expert Rev Mol Diagn. 2003;3:525-529. What Are Biomarkers?1-3 • PD-L1: a ligand for the immune checkpoint receptor PD-1 • PD-L1 expression can be measured by IHC and detected on tumor and immune cells PD-L1 expression FDA-approved pan-cancer biomarker used as either a companion or complementary diagnostic for multiple checkpoint inhibitors in different settings PD-L1 PD-L2 PD-1 T cell Tumor cell • MSI-H/dMMR status are indicators of genomic instability – MSI-H: change in number of nucleotide repeats in DNA sequences different number of repeats than in inherited DNA – dMMR: loss of function in the MMR pathway, a key DNA-repair system MSI/MMR FDA-approved pan-cancer biomarker for pembrolizumab, and in mCRC, for nivolumab and nivolumab + ipilimumab • TMB = collective no. of somatic mutations within the tumor’s genome; TMB levels vary in different tumors • Measured as total number of mutations or mutations per megabase • Thresholds for high vs low TMB still in flux; depend on assay + histology TMB • FDA-approved biomarker for pembrolizumab for patients with unresectable or metastatic TMB-high (≥10 mut/Mb) solid tumors that have progressed on prior treatment COMPANION Test result is required for prescription of the drug • Specified on drug label • Typically used when the test is among inclusion criteria for the trial (there are some exceptions) COMPLEMENTARY Testresultispredictive,butnot requiredforprescriptionofthedrug • Nice to have, but do not need to have • Mostly used when the assay is integrated into the trial, but not used among inclusion criteria Predictive Diagnostic Categories • Biomarkers are defined as “biological characteristics that can be objectively measured and evaluated as an indicator of normal biological processes, pathogenic processes, or pharmacological responses to a therapeutic intervention” • Biomarkers can be used to support the optimal clinical use (and development) of anticancer immunotherapies
