Driving Change in Multiple Myeloma: Updates on Novel Agent Classes and Next-Generation Therapeutics
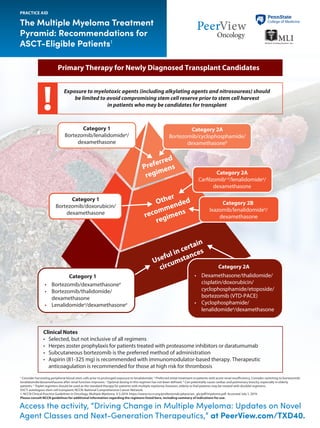
- 1. Access the activity, “Driving Change in Multiple Myeloma: Updates on Novel Agent Classes and Next-Generation Therapeutics,” at PeerView.com/TXD40. The Multiple Myeloma Treatment Pyramid: Recommendations for ASCT-Eligible Patients1 PRACTICE AID a Consider harvesting peripheral blood stem cells prior to prolonged exposure to lenalidomide. b Preferred initial treatment in patients with acute renal insufficiency. Consider switching to bortezomib/ lenalidomide/dexamethasone after renal function improves. c Optimal dosing in this regimen has not been defined. d Can potentially cause cardiac and pulmonary toxicity, especially in elderly patients. e Triplet regimens should be used as the standard therapy for patients with multiple myeloma; however, elderly or frail patients may be treated with doublet regimens. ASCT: autologous stem cell transplant; NCCN: National Comprehensive Cancer Network. 1. NCCN Clinical Practice Guidelines in Oncology: Multiple Myeloma. V.3.2019. https://www.nccn.org/professionals/physician_gls/pdf/myeloma.pdf. Accessed July 1, 2019. Please consult NCCN guidelines for additional information regarding the regimens listed here, including summary of indications for use. Other recommended regimens Preferred regimens Useful in certain circumstances Primary Therapy for Newly Diagnosed Transplant Candidates Exposure to myelotoxic agents (including alkylating agents and nitrosoureas) should be limited to avoid compromising stem cell reserve prior to stem cell harvest in patients who may be candidates for transplant Category 2A Bortezomib/cyclophosphamide/ dexamethasoneb Category 1 Bortezomib/lenalidomidea / dexamethasone Category 1 Bortezomib/doxorubicin/ dexamethasone Category 2B Ixazomib/lenalidomidea / dexamethasone Category 2A Carfilzomibc,d /lenalidomidea / dexamethasone Category 1 • Bortezomib/dexamethasonee • Bortezomib/thalidomide/ dexamethasone • Lenalidomidea /dexamethasonee Category 2A • Dexamethasone/thalidomide/ cisplatin/doxorubicin/ cyclophosphamide/etoposide/ bortezomib (VTD-PACE) • Cyclophosphamide/ lenalidomidea /dexamethasone Clinical Notes • Selected, but not inclusive of all regimens • Herpes zoster prophylaxis for patients treated with proteasome inhibitors or daratumumab • Subcutaneous bortezomib is the preferred method of administration • Aspirin (81-325 mg) is recommended with immunomodulator-based therapy. Therapeutic anticoagulation is recommended for those at high risk for thrombosis
- 2. Access the activity, “Driving Change in Multiple Myeloma: Updates on Novel Agent Classes and Next-Generation Therapeutics,” at PeerView.com/TXD40. The Multiple Myeloma Treatment Pyramid: Recommendations for Non–ASCT-Eligible Patients1 PRACTICE AID a Triplet regimens should be used as the standard therapy for patients with multiple myeloma; however, elderly or frail patients may be treated with doublet regimens. b Continuously until progression. Benboubker L et al. N Engl J Med. 2014;371:906-917. c Preferred initial treatment in patients with acute renal insufficiency. Consider switching to bortezomib/lenalidomide/dexamethasone after renal function improves. d May interfere with serological testing and cause false-positive indirect Coombs test. e Can potentially cause cardiac and pulmonary toxicity, especially in elderly patients. ASCT: autologous stem cell transplant. 1. NCCN Clinical Practice Guidelines in Oncology: Multiple Myeloma. V.3.2019. https://www.nccn.org/professionals/physician_gls/pdf/myeloma.pdf. Accessed July 1, 2019. 2. https://www.fda.gov/drugs/ resources-information-approved-drugs/fda-approves-daratumumab-multiple-myeloma-ineligible-autologous-stem-cell-transplant. Accessed July 5, 2019. Please consult NCCN guidelines for additional information regarding the regimens listed here, including summary of indications for use. Other recommended regimens Preferred regimens Useful in certain circumstances Newly Diagnosed Transplant-Ineligible Patients Category 2A Bortezomib/cyclophosphamide/ dexamethasonec Category 1 Category 2A • Bortezomib/dexamethasonea • Cyclophosphamide/ lenalidomide/dexamethasone Category 2A • Carfilzomibe /lenalidomide/ dexamethasone • Carflizomibe /cyclophosphamide/ dexamethasone • Ixazomib/lenalidomide/ dexamethasone • Bortezomib/lenalidomide/ dexamethasone • Lenolidomide/low-dose dexamethasonea,b • Daratumumabd /bortezomib/ melphalan/prednisone Clinical Notes • Selected, but not inclusive of all regimens • Herpes zoster prophylaxis for patients treated with proteasome inhibitors or daratumumab • Subcutaneous bortezomib is the preferred method of administration • Aspirin (81-325 mg) is recommended with immunomodulator-based therapy; therapeutic anticoagulation is recommended for those at high risk for thrombosis Daratumumab/lenalidomide/ dexamethasone recently approved for use in transplant-ineligible patients2
- 3. Access the activity, “Driving Change in Multiple Myeloma: Updates on Novel Agent Classes and Next-Generation Therapeutics,” at PeerView.com/TXD40. The Multiple Myeloma Treatment Pyramid: Recommendations for Patients With Relapsed/Refractory Disease1 a Can potentially cause cardiac and pulmonary toxicity, especially in elderly patients. b Triplet regimens should be used as the standard therapy for patients with multiple myeloma; however, elderly or frail patients may be treated with doublet regimens. c Clinical trials with these regimens primarily included patients who were lenalidomide-naïve or with lenalidomide-sensitive multiple myeloma. d May interfere with serological testing and cause false-positive indirect Coombs test. e Consider single-agent lenalidomide or pomalidomide for steroid-intolerant individuals. ASCT: autologous stem cell transplant; IMiD: immunomodulatory drug; MM: multiple myeloma; NCCN: National Comprehensive Cancer Network; R/R: relapsed/refractory; XPO1: exportin 1. 1. NCCN Clinical Practice Guidelines in Oncology: Multiple Myeloma. V.3.2019. https://www.nccn.org/professionals/physician_gls/pdf/myeloma.pdf. Accessed July 1, 2019. 2. Richardson PG et al. 2019 American Society of Clinical Oncology Annual Meeting (ASCO 2019). Abstract 8004. 3. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-accelerated-approval-selinexor- multiple-myeloma. Accessed July 1, 2019. Please consult NCCN guidelines for additional information regarding the regimens listed here, including summary of indications for use. PRACTICE AID Other recommended regimens Preferred regimens Useful in certain circumstances Therapy for Previously Treated Myeloma (If a regimen listed here was used as a primary induction therapy and relapse is >6 mo, the same regimen may be repeated) Category 2A • Bendamustine/bortezomib/ dexamethasone • Bendamustine/lenalidomide/ dexamethasone • Bortezomib/cyclophosphamide/ dexamethasone • Carfilzomiba /cyclophosphamide/ dexamethasone • Cyclophosphamide/lenalidomide/ dexamethasone • Daratumumabd • Daratumumabd /pomalidomide/ dexamethasone • Elotuzumab/bortezomib/ dexamethasone • Elotuzumab/pomalidomide/ dexamethasone • Ixazomib/dexamethasoneb • Ixazomib/pomalidomide/ dexamethasone • Panobinostat/carfilzomiba,b • Panobinostat/lenalidomide/ dexamethasone • Pomalidomide/cyclophosphamide/ dexamethasone • Pomalidomide/bortezomib/ dexamethasone • Pomalidomide/carfilzomiba / dexamethasone Category 2A • Bendamustine • High-dose cyclophosphamide Generally reserved for aggressive disease • Dexamethasone/ cyclophosphamide/etoposide/ cisplatin (DCEP) • Dexamethasone/thalidomide/ cisplatin/doxorubicin/ cyclophosphamide/etoposide (DT-PACE) ± bortezomib (VTD-PACE) • In phase 3 ICARIA-MM trial, the addition of the next-generation anti-CD38 antibody isatuximab to pomalidomide and dexamethasone substantially improved PFS vs doublet therapy in patients with R/R MM2 • XPO1 inhibitor selinexor recently approved for use in patients with R/R MM who have received ≥4 prior therapies (including patients refractory to ≥2 proteasome inhibitors or IMiDs and a CD38 antibody)3 Category 2A • Bortezomib/lenalidomide/dexamethasone • Carfilzomib (weekly)a /dexamethasoneb Category 1 • Bortezomib/liposomal doxorubicin/dexamethasone • Bortezomib/dexamethasoneb • Lenalidomide/ dexamethasoneb,e • Panobinostat/bortezomib/ dexamethasone • Pomalidomide/ dexamethasoneb,e Category 1 • Carfilzomib (2 x wk)a / dexamethasoneb • Carfilzomiba /lenalidomide/ dexamethasonec • Daratumumabd /bortezomib/ dexamethasone • Daratumumabd /lenalidomide/ dexamethasone • Elotuzumab/lenalidomide/ dexamethasonec • Ixazomib/lenalidomide/ dexamethasonec Clinical Notes • Selected, but not inclusive of all regimens • Herpes zoster prophylaxis for patients treated with proteasome inhibitors or daratumumab • Subcutaneous bortezomib is the preferred method of administration • Aspirin (81-325 mg) is recommended with immunomodulator-based therapy. Therapeutic anticoagulation is recommended for those at high risk for thrombosis • Consideration for appropriate regimen is based on the context of clinical relapse Recent updates in R/R MM
