Incorporating Cancer Immunotherapies Into the Oncology Treatment Arsenal in Managed Care Settings: Navigating the Complexities of Value Assessment & Cost Optimization in the Era of Immuno-Oncology
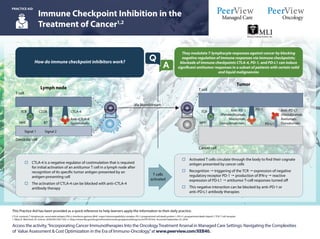
- 1. CTLA: cytotoxic T-lymphocyte–associated antigen; IFN-γ: interferon gamma; MHC: major histocompatibility complex; PD-1: programmed cell death protein 1; PD-L1: programmed death-ligand 1; TCR: T-cell receptor. 1. Ribas A, Wolchock JD. Science. 2018;359:1350-1355. 2. https://www.fda.gov/drugs/informationondrugs/approveddrugs/ucm279174.htm. Accessed September 25, 2018. Access the activity,“Incorporating Cancer Immunotherapies Into the OncologyTreatment Arsenal in Managed Care Settings: Navigating the Complexities of Value Assessment & Cost Optimization in the Era of Immuno-Oncology,”at www.peerview.com/XEB40. This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice. Immune Checkpoint Inhibition in the Treatment of Cancer1,2 PRACTICE AID They modulate T-lymphocyte responses against cancer by blocking negative regulation of immune responses via immune checkpoints; blockade of immune checkpoints CTLA-4, PD-1, and PD-L1 can induce significant antitumor responses in a subset of patients with certain solid and liquid malignancies How do immune checkpoint inhibitors work? o Activated T cells circulate through the body to find their cognate antigen presented by cancer cells o Recognition " triggering of the TCR " expression of negative regulatory receptor PD-1 " production of IFN-γ " reactive expression of PD-L1 " antitumor T-cell responses turned off o This negative interaction can be blocked by anti–PD-1 or anti–PD-L1 antibody therapies o CTLA-4 is a negative regulator of costimulation that is required for initial activation of an antitumor T cell in a lymph node after recognition of its specific tumor antigen presented by an antigen-presenting cell o The activation of CTLA-4 can be blocked with anti–CTLA-4 antibody therapy Lymph node T cell Dendritic cell TCR MHC Signal 1 Signal 2 CD28 B7 CTLA-4 Anti–CTLA-4 (Ipilimumab) Via bloodstream Tumor T cell Cancer cell TCR MHC Anti–PD-L1 (Atezolizumab, Avelumab, Durvalumab) Anti–PD-1 (Pembrolizumab, Nivolumab, Cemiplimab-rwlc) PD-L1 PD-1 T cells activated A Q
- 2. 1. Postow MA et al. N Engl J Med. 2018;378:158-168. 2. Brahmer JR et al. J Clin Oncol.2018;36:1714-1768. Immune-Related Adverse Events Associated With the Immune Checkpoint Inhibitors Used in Oncology1,2 PRACTICE AID Access the activity,“Incorporating Cancer Immunotherapies Into the OncologyTreatment Arsenal in Managed Care Settings: Navigating the Complexities of Value Assessment & Cost Optimization in the Era of Immuno-Oncology,”at www.peerview.com/XEB40. This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice. • In general, checkpoint inhibitor therapy should be continued with close monitoring, with the exception of some neurologic, hematologic, and cardiac toxicities (Minimalornosymptoms;diagnosticchangesonly) Grade 1 • Hold checkpoint inhibitor therapy for most grade 2 toxicities • Consider resuming immunotherapy when symptoms and/or lab values revert ≤ grade 1 • Corticosteroids (initial dose of 0.5-1 mg/kg/day of prednisone or equivalent) may be administered Grade 2 (Mild to moderate symptoms) (Severe or life-threatening symptoms) Grades 3/4 Grade 3 toxicities: • Hold checkpoint inhibitor therapy • Initiate high-dose corticosteroids (prednisone 1-2 mg/kg/day or methylprednisolone IV 1-2 mg/kg/day) • If symptoms do not improve with 48-72 hours of high-dose corticosteroid, infliximab may be offered for some toxicities • Taper corticosteroids over the course of at least 4-6 weeks • When symptoms and/or laboratory values revert to ≤ grade 1, rechallenging with immunotherapy may be offered; however caution is advised, especially in those patients with early-onset irAEs; dose adjustments are not recommended Grade 4 toxicities: • In general, permanent discontinuation of checkpoint inhibitor therapy is warranted, with the exception of endocrinopathies that have been controlled by hormone replacement Brahmer JR et al. Management of Immune-Related Adverse Events in PatientsTreatedWith Immune Checkpoint InhibitorTherapy: American Society of Clinical Oncology Clinical Practice Guideline. J Clin Oncol. 2018;36(17):1714-1768. For organ-specific assessment and management of irAEs, please see the ASCO guidelines: Additional resources available on the ASCO website: https://www.asco.org/practice-guidelines/quality-guidelines/guidelines/ supportive-care-and-treatment-related-issues#/29866 General recommendations and management principles include the following: IrAEs are often diagnosed by exclusion; other causes should be ruled out (including AEs of other therapies used), but immunotherapy-related toxicity should always be included in the differential There should be a high level of suspicion that new symptoms are treatment- related; early recognition, evaluation, and treatment of irAEs plus patient education are essential for best outcome Depending on severity of irAE, management may require corticosteroid or other immunosuppressive treatment and interruption or discontinuation of therapy If appropriate immunosuppressive treatment is used, patients generally recover from irAEs Use of immunosuppressive therapy to manage irAEs does not impact response to immunotherapy How should irAEs be diagnosed and managed?Q A What is the spectrum of potential irAEs?Why do irAEs occur? Any organ system can be affected; more commonly occurring are pulmonary (pneumonitis), dermatologic (rash, pruritus, blisters, ulcers, vitiligo), gastrointestinal (diarrhea, enterocolitis, transaminitis, hepatitis, pancreatitis), and endocrine (thyroiditis, hypophysitis, adrenal insufficiency) irAEs The precise pathophysiology is unknown, but translational studies have shown that T-cell, antibody, and cytokine responses may be involved Pancreatitis, autoimmune diabetes Colitis Enteritis Encephalitis, aseptic meningitis Thyroiditis, hypothyroidism, hyperthyroidism Dry mouth, mucositis Hypophysitis Uveitis Pneumonitis Thrombocytopenia, anemia Hepatitis Adrenal insufficiency Nephritits Vasculitis Arthralgia Neuropathy Rash, vitiligo Myocarditis Activated T cell Antithyroid antibodies Increasing levels of inflammatory cytokines Increasing levels of preexisting autoantibodies Enhancing complement- mediated inflammation due to direct binding of an anti–CTLA-4 antibody with CTLA-4 expressed on normal tissue Increasing T-cell activity against antigens that are present in tumors and healthy tissue Activated T cell Anti–CTLA-4 antibody CTLA-4 on pituitary Complement- mediated inflammation Cytokines Tumor with antigen and activated T cells Immune checkpoint inhibitors are associated with important clinical benefits, but general immunologic enhancement can also lead to a unique spectrum of immune-related adverse effects (irAEs) A A Q Q
- 3. CRC: colorectal cancer; CSCC: cutaneous squamous-cell carcinoma; CTLA: cytotoxic T-lymphocyte–associated antigen; dMMR: mismatch repair deficient; HCC: hepatocellular carcinoma; HNSCC: head and neck squamous cell carcinoma; MSI-H: microsatellite instability high; PD-1: programmed death 1; PD-L1: programmed death-ligand 1; RCC: renal cell carcinoma; SCLC: small cell lung cancer. 1. https://www.fda.gov/drugs/informationondrugs/approveddrugs/ucm279174.htm. Accessed October 19, 2018. Access the activity,“Incorporating Cancer Immunotherapies Into the OncologyTreatment Arsenal in Managed Care Settings: Navigating the Complexities of Value Assessment & Cost Optimization in the Era of Immuno-Oncology,”at www.peerview.com/XEB40. This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice. Rapidly Evolving Immuno-Oncology Landscape Immune Checkpoint Inhibitors and Biomarkers: Solid Tumors1 PRACTICE AID Drug Ipilimumab Nivolumab Pembrolizumab Cemiplimab-rwlc Atezolizumab Avelumab Durvalumab Mechanism Anti–CTLA-4 Anti–PD-1 Anti–PD-L1 Current Immune Checkpoint Inhibitors: Classes and Agents September Pembrolizumab (melanoma) December Nivolumab (melanoma) March Ipilimumab (melanoma) 2011 2014 2015 2016 2017 2018 May Atezolizumab (bladder) August Pembrolizumab (HNSCC) October Pembrolizumab (PD-L1+ NSCLC, first line) Atezolizumab (NSCLC, second line) November Nivolumab (HNSCC) March Nivolumab (squamous NSCLC, second line) October Nivolumab (nonsquamous NSCLC, second line) Pembrolizumab (PD-L1+ NSCLC, second line) Nivolumab + ipilimumab (melanoma, first line) Ipilimumab (melanoma, adjuvant) November Nivolumab (RCC) December Pembrolizumab (melanoma, first line) February Nivolumab (bladder) March Avelumab (Merkel cell carcinoma) May Durvalumab, avelumab, pembrolizumab (bladder) Pembrolizumab + chemotherapy (nonsquamous NSCLC, first line) Pembrolizumab (dMMR/MSI-H cancers) August Nivolumab (dMMR/MSI-H CRC) September Pembrolizumab (gastric) Nivolumab (HCC) December Nivolumab (melanoma, adjuvant) February Durvalumab (stage III NSCLC) April Nivolumab + ipilimumab (RCC, first line) June Pembrolizumab (PD-L1+ cervical) July Nivolumab + ipilimumab (dMMR/MSI-H CRC) August Nivolumab (SCLC, third line) Pembrolizumab + chemotherapy (nonsquamous NSCLC, first line—full approval) September Cemiplimab-rwlc (CSCC) October Pembrolizumab + carboplatin + paclitaxel or nab-paclitaxel (squamous NSCLC) November Pembrolizumab (HCC, 2nd line) December Atezolizumab + bevacizumab + chemotherapy (nonsquamous NSCLC) Current FDA-Approved Indications for Immune Checkpoint Inhibitors1 Clinically Relevant Predictive Biomarkers Currently used predictive biomarkers in clinical practice • PD-L1 expression • MSI-H or dMMR Next emerging predictive biomarker • TMB
- 4. ASCO: American Society of Clinical Oncology; ESMO-MCBS: European Society for Medical Oncology-Magnitude of Clinical Benefit Scale; HR: hazard ratio; ICER: Institute for Clinical and Economic Review; MSKCC: Memorial Sloan Kettering Cancer Center; NCCN: National Comprehensive Cancer Network; OS: overall survival; PFS: progression-free survival; RR: response rate. 1. Kaufman HL et al. J Immunother Cancer. 2017;5:38. Oncology Value Frameworks and Their Limitations in Assessing the Benefits and Costs of Cancer Immunotherapies1 PRACTICE AID Access the activity,“Incorporating Cancer Immunotherapies Into the OncologyTreatment Arsenal in Managed Care Settings: Navigating the Complexities of Value Assessment & Cost Optimization in the Era of Immuno-Oncology,”at www.peerview.com/XEB40. This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice. Current value models: £ Overestimate the treatment costs necessary to achieve maximum benefit – Eg, combinations of immunotherapy may be less expensive than monotherapy if they work faster (require less drug) and last longer £ Overestimate the impact of acute, but reversible toxicities – Toxicities are often short-lived (4 weeks) and manageable – Death from noncurative therapy is typically not counted as a toxicity in comparisons – Opportunities exist to reduce toxicity £ Underestimate the value of long-term survival and treatment-free survival – Time horizons need to account for long duration of benefits, including treatment-free survival » Benefits to patients, family, community, and colleagues £ Do not give adequate consideration to societal factors – Benefits of potential cures – Improvements in health and quality of life – Potential to use curative treatments as platforms on which to build Framework Factors Considered Purpose Costs Measured? Perspective ASCO Net health benefit: Clinical benefit (OS, PFS, RR), toxicity, extended survival Comparison of two regimens that have been reported in a randomized clinical trial No Patient ESMO-MCBS Magnitude of clinical benefit: Prognosis of condition, clinical benefit (OS, PFS), long-term survival (HR, RR), quality of life, toxicity Comparison of magnitude of benefit of regimens with reported comparative research outcome No Societal NCCN Evidence Blocks Efficacy, safety, quality of evidence, consistency of evidence, affordability Visual representation of key factors that provide information about the value of the recommendations within the guidelines Yes Patient MSKCC Drug Abacus Efficacy, cost toxicity, treatment novelty, costs of development, rarity of disease, population burden of condition Intended to provide information regarding the proper pricing of new drugs in the market Yes Societal ICER Value Incremental cost-effectiveness ratio: Cost new - Cost standard Effectiveness new - Effectiveness standard Comparison of two treatments based on efficacy and cost Yes Societal Comparison of Current Value Frameworks Used in Oncology Limitations of Current Oncology Value Frameworks Related to Immuno-Oncology
