5. Avogardos.pptx
•Download as PPTX, PDF•
0 likes•880 views
AVOGADROS
Report
Share
Report
Share
More Related Content
What's hot
What's hot (20)
Papel ng mamamayan sa pagkakaroon ng mabuting pamamahala

Papel ng mamamayan sa pagkakaroon ng mabuting pamamahala
Hudyat sa pagkasunod sunod ng mga pangayayari grade7

Hudyat sa pagkasunod sunod ng mga pangayayari grade7
Similar to 5. Avogardos.pptx
Similar to 5. Avogardos.pptx (20)
Combined-Avogadros-and-Ideal-Gas-Laws [Autosaved].pptx![Combined-Avogadros-and-Ideal-Gas-Laws [Autosaved].pptx](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Combined-Avogadros-and-Ideal-Gas-Laws [Autosaved].pptx](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
Combined-Avogadros-and-Ideal-Gas-Laws [Autosaved].pptx
Recently uploaded
young call girls in Pandav nagar 🔝 9953056974 🔝 Delhi escort Service

young call girls in Pandav nagar 🔝 9953056974 🔝 Delhi escort Service9953056974 Low Rate Call Girls In Saket, Delhi NCR
Captivating Charm: Exploring Marseille's Hillside Villas with Our 3D Architec...

Captivating Charm: Exploring Marseille's Hillside Villas with Our 3D Architec...Yantram Animation Studio Corporation
Call Girls Service Mukherjee Nagar @9999965857 Delhi 🫦 No Advance VVIP 🍎 SER...

Call Girls Service Mukherjee Nagar @9999965857 Delhi 🫦 No Advance VVIP 🍎 SER...Call Girls In Delhi Whatsup 9873940964 Enjoy Unlimited Pleasure
Recently uploaded (20)
VIP College Call Girls Gorakhpur Bhavna 8250192130 Independent Escort Service...

VIP College Call Girls Gorakhpur Bhavna 8250192130 Independent Escort Service...
CALL ON ➥8923113531 🔝Call Girls Aminabad Lucknow best Night Fun service

CALL ON ➥8923113531 🔝Call Girls Aminabad Lucknow best Night Fun service
Cheap Rate Call girls Malviya Nagar 9205541914 shot 1500 night

Cheap Rate Call girls Malviya Nagar 9205541914 shot 1500 night
NATA 2024 SYLLABUS, full syllabus explained in detail

NATA 2024 SYLLABUS, full syllabus explained in detail
VIP Call Girls Service Bhagyanagar Hyderabad Call +91-8250192130

VIP Call Girls Service Bhagyanagar Hyderabad Call +91-8250192130
young call girls in Pandav nagar 🔝 9953056974 🔝 Delhi escort Service

young call girls in Pandav nagar 🔝 9953056974 🔝 Delhi escort Service
Raj Nagar Extension Call Girls 9711199012 WhatsApp No, Delhi Escorts in Raj N...

Raj Nagar Extension Call Girls 9711199012 WhatsApp No, Delhi Escorts in Raj N...
Captivating Charm: Exploring Marseille's Hillside Villas with Our 3D Architec...

Captivating Charm: Exploring Marseille's Hillside Villas with Our 3D Architec...
VIP Russian Call Girls in Saharanpur Deepika 8250192130 Independent Escort Se...

VIP Russian Call Girls in Saharanpur Deepika 8250192130 Independent Escort Se...
Abu Dhabi Call Girls O58993O4O2 Call Girls in Abu Dhabi`

Abu Dhabi Call Girls O58993O4O2 Call Girls in Abu Dhabi`
VIP Call Girl Amravati Aashi 8250192130 Independent Escort Service Amravati

VIP Call Girl Amravati Aashi 8250192130 Independent Escort Service Amravati
Call Girls Service Mukherjee Nagar @9999965857 Delhi 🫦 No Advance VVIP 🍎 SER...

Call Girls Service Mukherjee Nagar @9999965857 Delhi 🫦 No Advance VVIP 🍎 SER...
Best VIP Call Girls Noida Sector 44 Call Me: 8448380779

Best VIP Call Girls Noida Sector 44 Call Me: 8448380779
The_Canvas_of_Creative_Mastery_Newsletter_April_2024_Version.pdf

The_Canvas_of_Creative_Mastery_Newsletter_April_2024_Version.pdf
5. Avogardos.pptx
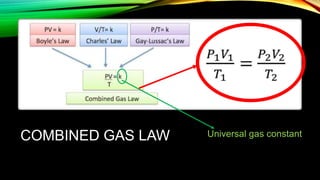
- 1. COMBINED GAS LAW Universal gas constant
- 2. SAMPLE PROBLEM • The oxygen tank manufacturer used to produce 5.0L oxygen tanks at 2000 psi and 25°C . Statistics suggest that the 3.0 L oxygen tank at 1500 psi more marketable. What temperature requirement is needed to produce a 3 L oxygen tank at 1500 psi? Initial Conditions Final Conditions V1=5.0 L V2= 3.0 L T1= 25° C T2= ? P1= 2000 psi P2=1500psi
- 3. Initial Conditions Final Conditions V1=5.0 L V2= 3.0 L T1= 25° C T2= ? P1= 2000 psi P2=1500psi T2 = T1P2V2 P1V1 T2 =(298 K) (1500psi)(3.0L) (2000 psi) (5.0 L) T2 = 134.1 K
- 4. SAMPLE PROBLEM A gas balloon has a volume of 106.0 liters when the temperature is 45.0 °C and the pressure is 740.0 mm of mercury. What will its volume be at 20.0 °C and 780 .0 mm of mercury pressure?
- 5. SEATWORK 1 •Helium gas has a volume of 250 mL at 0°C at 1.0 atm. What will be the final pressure if the volume is reduced to 100mL at 45° C? 2.91 atm
- 6. SEATWORK 2 •The volume of the gas at 27°C and 700.0 mmHg is 600.0 mL. What is the volume of the gas at -20.0°C and 500.0 mmHg? 708.4ml
- 7. LORENZO ROMANO AMADEO CARLO AVOGADRO The volume of gas id directly proportional to the number of moles of gas present at a given temperature and pressure. V∞ n at constant T and P V ∞ n K 𝑉 𝑛 = K
- 8. BOYLES/CHARLES/AVOGADRO PV= nRT V=volume in liters P= pressure in atm n= moles T=temperature in Kelvin R=universal gas constant (0.0821 L. atm mol K)
- 9. APPLICATION USING GAS LAW The exposure to high temperature increases the kinetic energy of the gas causing an increase in the pressure due to the increased collision. An increase in pressure would result in expansion in volume.
- 12. p= 𝑚 𝑎
- 13. d= 𝑚 𝑣
- 14. Boyle’s Law Combined Gas Law Ideal gas law