Easl immuno poster
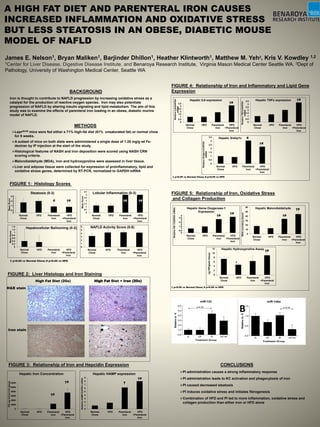
- 1. A HIGH FAT DIET AND PARENTERAL IRON CAUSES INCREASED INFLAMMATION AND OXIDATIVE STRESS BUT LESS STEATOSIS IN AN OBESE, DIABETIC MOUSE MODEL OF NAFLD James E. Nelson1, Bryan Maliken1, Barjinder Dhillon1, Heather Klintworth1, Matthew M. Yeh2, Kris V. Kowdley 1,2 1Center for Liver Disease, Digestive Disease Institute, and Benaroya Research Institute, Virginia Mason Medical Center Seattle WA. 2Dept of Pathology, University of Washington Medical Center, Seattle WA METHODS Leprdb/db mice were fed either a 71% high-fat diet (61% unsaturated fat) or normal chow for 8 weeks. A subset of mice on both diets were administered a single dose of 1.25 mg/g wt Fe- dextran by IP injection at the start of the study. Histological features of NASH and iron deposition were scored using NASH CRN scoring criteria. Malondialdehyde (MDA), iron and hydroxyproline were assessed in liver tissue. Liver and adipose tissue were collected for expression of proinflammatory, lipid and oxidative stress genes, determined by RT-PCR, normalized to GAPDH mRNA 0 0.5 1 1.5 2 2.5 3 3.5 Normal Chow HFD Parenteral Iron HFD +Parenteral Iron MeanScore Steatosis (0-3) # 0 0.5 1 1.5 2 2.5 3 3.5 Normal Chow HFD Parenteral Iron HFD +Parenteral Iron MeanScore Lobular Inflammation (0-3) †#†# 0 0.2 0.4 0.6 0.8 1 1.2 1.4 Normal Chow HFD Parenteral Iron HFD +Parenteral Iron MeanScore Hepatocellular Ballooning (0-2) 0 1 2 3 4 5 6 Normal Chow HFD Parenteral Iron HFD +Parenteral Iron NAFLD Activity Score (0-8) †# † p<0.05 vs Normal Chow; # p<0.05 vs HFD High Fat Diet (20x) High Fat Diet + Iron (20x) 0 0.5 1 1.5 2 2.5 3 3.5 Normal Chow HFD Parenteral Iron HFD +Parenteral Iron RelativeIL6/GAPDH mRNA Hepatic IL6 expression †# 0 0.5 1 1.5 2 2.5 3 3.5 Normal Chow HFD Parenteral Iron HFD +Parenteral Iron RelativeTNFα/GAPDH mRNA Hepatic TNFα expression †# † p<0.05 vs Normal Chow; # p<0.05 vs HFD 0 0.5 1 1.5 2 2.5 3 3.5 Normal Chow HFD Parenteral Iron HFD +Parenteral Iron RelativeSrebp1c/GAPDH mRNA Hepatic Srebp1c †# # 0 2000 4000 6000 8000 10000 12000 Normal Chow HFD Parenteral Iron HFD +Parenteral Iron HICµgiron/glivertissue Hepatic Iron Concentration †# †# 0 1 2 3 4 5 6 7 8 9 Normal Chow HFD Parenteral Iron HFD +Parenteral Iron RelativeHAMP/GAPDHmRNA Hepatic HAMP expression †# † BACKGROUND Iron is thought to contribute to NAFLD progression by increasing oxidative stress as a catalyst for the production of reactive oxygen species. Iron may also potentiate progression of NAFLD by altering insulin signaling and lipid metabolism. The aim of this study was to examine the effects of parenteral iron loading in an obese, diabetic murine model of NAFLD. FIGURE 1: Histology Scores FIGURE 2: Liver Histology and Iron Staining H&E stain Iron stain FIGURE 3: Relationship of Iron and Hepcidin Expression 0 0.5 1 1.5 2 2.5 3 3.5 4 Normal Chow HFD Parenteral Iron HFD +Parenteral Iron RelativeHO-1/GAPDHmRNA Hepatic Heme Oxygenase-1 Expression †# †# 0 10 20 30 40 50 60 Normal Chow HFD Parenteral Iron HFD +Parenteral IronMDA(umoles/gtissue) Hepatic Malondialdehyde †# †# 0 2 4 6 8 10 12 Normal Chow HFD Parenteral Iron HFD +Parenteral Iron ugHP/gramtissue Hepatic Hydroxyproline Assay †# † † p<0.05 vs Normal Chow; # p<0.05 vs HFD FIGURE 5: Relationship of Iron, Oxidative Stress and Collagen Production FIGURE 4: Relationship of Iron and Inflammatory and Lipid Gene Expression B CONCLUSIONS PI administration causes a strong inflammatory response PI administration leads to KC activation and phagocytosis of iron PI caused decreased steatosis PI induces oxidative stress and initiates fibrogenesis Combination of HFD and PI led to more inflammation, oxidative stress and collagen production than either iron or HFD alone