Proteins and their biological structures
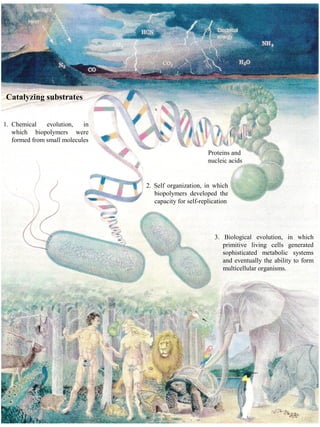
- 1. Catalyzing substrates 1. Chemical evolution, in which biopolymers were formed from small molecules Proteins and nucleic acids 2. Self organization, in which biopolymers developed the capacity for self-replication 3. Biological evolution, in which primitive living cells generated sophisticated metabolic systems and eventually the ability to form multicellular organisms.
- 4. 1. Enzymes (more 2000 proteins) - very specific catalysis up to 1020 times 2. Regulatory proteins (hormones, gene expression) 3. Transport proteins (Hb, HSA, membrane proteins) 4. Storage proteins (ovalbumin, casein, ferritin - 460kD -4500 atoms of iron) 5. Contractile and motile proteins (actin, myosin, tubulin, dynein, kinesin) 6. Structural proteins (keratins, collagen -1/3 of total) 7. Protective and exploitive proteins (immunoglobulins, thrombin, fibrinogen) 8. Exotic proteins (antifreeze proteins, monellin – sweetener, resilin – elastics, glue proteins) BIOLOGICAL FUNCTIONS OF PROTEINS
- 5. Although more than 300 different amino acids have been described in nature, only 20 are commonly found as constituents of mammalian proteins. These are the only amino acids that are coded for by DNA, the genetic material in the cell. At physiologic pH, the carboxyl group is dissociated, forming the negatively charged carboxylate ion (–COO- ), and the amino group is protonated (–NH3 + ). In proteins, almost all carboxyl and amino groups are combined through peptide linkage and, in general, are not available for chemical reaction except for hydrogen bond formation.
- 7. UNCHARGED POLAR SIDE CHAINS ACIDIC SIDE CHAINS BASIC SIDE CHAINS
- 8. Protein function can be understood only in terms of protein structure 1. Primary structure is the amino acid sequence of its polypeptide chain 2. Secondary structure in the local spatial arrangement of a polypeptide’s backbone atoms without regards to the conformations f its side chain 3. Tertiary structure refers to the three-dimensional structure of an entire polypeptide 4. Quaternary structure refers to the spatial arrangement of the subunits of the protein associated through noncovalent interactions and disulfide bonds. (a) – Lys – Ala – His – Gly – Lys – Lys – Val – Leu – Gly – Ala – Primary structure (amino acid sequence in a polypeptide chain) (b) Secondar y structure (helix) (c) (d) β2 β Tertiary structure: one complete protein chain (β chain of hemoglobin) β1 α2 α1 Quatemary structure: the four separate chains of hemoglobin assembled into an oligomeric protein The structural hierarchy in proteins: (a) primary structure, (b) secondary structure, (c) tertiary structure, and (d) quaternary structure.
- 9. Interactions between the side chains of amino acid residues in proteins. 1- electrostatic interactions; 2 – hydrogen bonds; 3 – hydrophobic interactions; 4 – disulfide bonds.
- 10. SECONDARY STRUCTURE OF PROTEINS The polypeptide backbone forms regular arrangements of amino acids that are located near to each other in the linear sequence. These arrangements are termed the secondary structure of the polypeptide. The α-helix, β- sheet, and β-bend (β-turn) are examples of secondary structures frequently encountered in proteins.
- 11. The 20 amino acids commonly found in proteins are joined together by peptide bonds. The linear sequence of the linked amino acids contains the information necessary to generate a protein molecule with a unique three- dimensional shape. The complexity of protein structure is best analyzed by considering the molecule in terms of four organizational levels, namely, primary, secondary, tertiary, and quaternary.
- 12. Interactions stabilizing tertiary structure The unique three-dimensional structure of each polypeptide is determined by its amino acid sequence. Interactions between the amino acid side chains guide the folding of the polypeptide to form a compact structure. 1. Disulfide bonds 2. Hydrophobic interactions 3. Hydrogen bonds 4. Ionic interactions
- 13. Size of Protein Molecules Protein Mr Number of Residues per Chain Subunit Organization Insulin (bovine) 5,733 21 (A) 30 (B) αβ Cytochrome c (equine) 12,500 104 α1 Ribonuclease A (bovine pancreas) 12,640 124 α1 Lysozyme (egg white) 13,930 129 α1 Myoglobin (horse) 16,980 153 α1 Chymotrypsin (bovine pancreas) 22,600 13 (α) 132 (β) 97 (γ) αβγ Hemoglobin (human) 64,500 141 (α) 146 (β) α2β2 Serum albumin (human) 68,500 550 α1 Hexokinase (yeast) 96,000 200 α4 γ-Glubulin (horse) 149,900 214 (α) 446 (β) α2β2 Glutamate dehydrogenase (liver) 332,694 500 α6 Myosin (rabbit) 470,000 1800 (heavy, h) 190 (α) 149 (α’) 160 (β) h2α1α’2 β2 Ribulose bisphosphate carboxylase (spinach) 560,000 475 (α) 123 (β) α8 β8 Glutamine synthetase (E. coli) 600,000 468 α12 Insulin Cytochrome c Ribonuclease Lysozyme Myoglobin Hemoglobin Immunoglobulin Glutamine synthetase
- 14. PROTEIN FOLDING Protein folding, which occurs within the cell in seconds to minutes, employs a shortcut through the maze of all folding possibilities. For example, positively and negatively charged side chains attract each other, conversely, similarly charged side chains repel each other. In addition, interactions involving hydrogen bonds, hydrophobic interactions, and disulfide bonds all exert an influence on the folding process. This results in a correctly folded protein with a low-energy state. DENATURATION OF PROTEINS Protein denaturation results in the unfolding and disorganization of the protein’s secondary and tertiary structures, which are not accompanied by hydrolysis of peptide bonds. Denaturing agents include heat, organic solvents, mechanical mixing, strong acids or bases, detergents, and ions of heavy metals such as leas and mercury. Most proteins, once denatured, remain permanently disordered. Denatured proteins are often insoluble and, therefore, precipitate from solution.
- 15. Structure and function of hemoglobin Hemoglobin is found exclusively in red blood cells (RBCs), where its main function is to transport oxygen (O2) from the lungs to the capillaries of the tissues. Hemoglobin A, the major hemoglobin in adults, is composed of four polypeptide chains – two α chains and two β chains – held together by noncovalent interactions. Each subunit has stretches of α-helical structure, and a heme-binding pocket.
- 16. Amino acid substitution in Hb S β chains: A molecule of Hb S contains two normal α- globin chains and two mutant β-globin chains (βS ), in which glutamate at position six has been replaced with valine. The replacement of the charged glutamate with the nonpolar valine forms a protrusion on the β-globin that fits into a complementary site on the β chain of another hemoglobin molecule in the cell. At low oxygen tension, deoxyhemoglobin S polymerizes inside the RBC, forming a network of fibrous polymers that stiffen and distort the cell, producing rigid, misshapen erythrocytes. Such sickled cells frequently block the flow of blood in the narrow capillaries. This interruption in the supply of oxygen leads to localized anoxia (oxygen deprivation) in the tissue, causing pain and eventually death (infarction) of cells in the vicinity of the blockage.
- 17. 1. Amino acid substitution in Hb S β chains: A molecule of Hb S contains two normal α-globin chains and two mutant β globin chains (βS ), in which glutamate at position six has been replaced with valine. 2. Sickling and tissue anoxia: The replacement of the charged glutamate with the nonpolar valine forms a protrusion on the β-globin that fits into a complementary site on the β chain of another hemoglobin molecule in the cell. At low oxygen tension, deoxyhemoglobin S polymerizes inside the RBC, forming a network of fibrous polymers that stiffen and distort the cell, producing rigid, misshapen erythrocytes. Such sickled cells frequently block the flow of blood in the narrow capillaries. This interruption in the supply of oxygen leads to localized anoxia (oxygen deprivation) in the tissue, causing pain and eventually death (infarction) of cells in the vicinity of the blockage.
- 19. Hemeproteins are a group of specialized proteins that contain heme as a tightly bound prosthetic group. The role of the heme group is dictated by the environment created by the three- dimensional structure of the protein. In hemoglobin and myoglobin, the two most abundant heme-proteins in humans, the heme group serves to reversibly bind oxygen. Heme is a complex of protoporphyrin IX and ferrous iron (Fe2+ ). The iron is held in the center of the heme molecule by bonds to the four nitrogens of the porphyrin ring. The heme Fe2+ can form two additional bonds, one on each side of the planar porphyrin ring. In myoglobin and hemoglobin, one of these positions is coordinated to the side chain of a histidine residue of the globin molecule, whereas the other position is available to bind oxygen.
- 20. Structure and function of myoglobin Myoglobin, a hemeprotein present in heart and skeletal muscle, functions both as a reservoir for oxygen, and as an oxygen carrier. Myoglobin consists of a single polypeptide chain that is structurally similar to the individual subunit polypeptide chains of the hemoglobin molecule. Myoglobin is a compact molecule, with approximately 80 % of its polypeptide chain folded into eight stretches of α- helix.
- 21. An artist's rendering of sperm whale myoglobin. One of the heme group's propionic acid side chains has been displaced for clarity. The amino acid residues are consecutively numbered, starting from the N-terminus, and the eight helices are likewise designated A through H.
- 22. Sickle cell hemoglobin (HbS) Glu (β-chain) replaced by Val DeoxyHbS undergoes self association and forms a liquid crystalline phase, which distorts the erythrocytes into a sickle shape. Sickling: leads to their aggregation and to a decrease in blood circulation (anemia, tissue infarction and chronic failure of organ function) HEMOGLOBIN: colored component of the blood 34% solution in erythrocytes Mr 64500 - tetramer composed of 2 polypeptide chain pairs With a Fe2+ content of 0.334%, the total of 950g Hb in a human represent 3.5 g or 80% of the total body iron Several forms of human Hb (α, β, γ, ε, δ) Presently known 153 abnormal Hb (87 are variations of β-chain) β-Thalassemia – no β-chain – patients contains HbF instead HbA1
- 23. Quaternary structure of hemoglobin: The hemoglobin tetramer can be envisioned as being composed of two identical dimers. The two polypeptide chains within each dimer are held tightly together, primarily by hydrophobic interactions. Ionic and hydrogen bonds also occur between the members of the dimer. • T form: The deoxy form of hemoglobin is called the “T” (tense) form. In the T form, the two αβ dimers interact through a network of ionic bonds and hydrogen bonds that constrain the movement of the polypeptide chains. The T form is the low-oxygen-affinity form of hemoglobin. • R form: The binding of oxygen to hemoglobin causes the rupture of some of the ionic bonds and hydrogen bonds between the αβ dimers. This leads to a structure called the “R”, or relaxed form, in which the polypeptide chains have more freedom of movement. The R form is the high-oxygen-affinity form of hemoglobin.
- 24. Aggregation or association of two or more identical or different polypeptide chains by noncovalent interaction leading to stable oligomeric (or multimeric) structure. 650 proteins (including 500 enzymes) with subunit structure had been described by 1974. Most of the known multimeric proteins contain either 2 or 4 similar sized subunits. Far less common are proteins with uneven numbers of subunits (by 1973, 28 trimeric proteins, including 23 enzymes; 5 pentameric, 2 heptameric and 1 nonameric). Possession of quarternary structure appear to confer a flexibility of shape and activity, which is necessary for the physiological role of the proteins. Monomers derived from multimeric enzymes are usually inactive. The subunit composition can be determined by dissociation of aggregate and investigation of the separate subunits by:ultracentrifugation, polyaerylamide disc electrophoresis, gel-filtration, ion-exchange chromatography The structure of the intact aggregate can be studied by: electron microscopy, low angle X-ray or neutron diffraction. QUARTERNARY STRUCTURE OF PROTEINS
- 25. The binding of a hypothetical pair of proteins by two ionic bonds, one hydrogen bond, and one large combination of hydrophobic and van der Waals interactions. The structural complementarity of the surfaces of surfaces of the two molecules gives rise to this particular combination of weak bonds and hence to the specificity of binding between the molecules.
- 26. Binding of oxygen to myoglobin and hemoglobin Myoglobin can bind only one molecule of oxygen. Hemoglobin can bind four oxygen molecules – one at each of its four heme groups. Oxygen dissociation curve: A plot of Y measured at different partial pressures of oxygen (pO2) is called the oxygen dissociation curve. This graph illustrates that myoglobin has a higher oxygen affinity at all pO2 values than does hemoglobin. • Myoglobin (Mb): The oxygen dissociation curve for myoglobin has a hyperbolic shape. The equilibrium is shifted to the right or to the left as oxygen is added to or removed from the system. Hemoglobin (Hb): The oxygen dissociation curve for hemoglobin is sigmoidal in shape, indicating that the subunits cooperate in binding oxygen. Cooperative binding of oxygen by the four subunits of hemoglobin means that the binding of an oxygen molecule at one heme group increases the oxygen affinity of the remaining heme groups in the same hemoglobin molecule. Allosteric effects The ability of hemoglobin to reversibly bind oxygen is affected by the pO2, the pH of the environment, the partial pressure of carbon dioxide, pCO2, and the availability of 2,3-bisphosphoglycerate. These are collectively called allosteric (“other site”) effectors.
- 27. Heme-heme interactions The net effect is that the affinity of hemoglobin for the last oxygen bound is approximately 300 times greater than its affinity for the first oxygen bound. Loading and unloading oxygen For example, in the lung, the concentration of oxygen is high and hemoglobin becomes virtually saturated (or “loaded”) with oxygen. In contrast, in the peripheral tissues, oxyhemoglobin releases (or “unloads”) much of its oxygen for use in the oxidative metabolism of the tissues. The concentration of both CO2 and H+ in the capillaries of metabolically active tissues is higher than that observed in alveolar capillaries of the lungs. In the tissues, CO2 is converted by carbonic anhydrase to carbonic acid: CO2 + H2O H2CO3 H2CO3 HCO3 – + H+ which spontaneously loses a proton, becoming bicarbonate: The H+ contributes to the lowering of pH. This differential pH gradient (lungs having a higher pH, tissues a lower pH) favors the unloading of oxygen in the peripheral tissues, and the loading of oxygen in the lung. Thus, the oxygen affinity of the hemoglobin molecule responds to small shifts in pH between the lungs and oxygen- consuming tissues, making hemoglobin a more efficient transporter of oxygen. Source of the protons that lower the pH
- 28. Effect of 2,3-bisphosphoglycerate on oxygen affinity It is the most abundant organic phosphate in the RBC, where its concentration is approximately that of hemoglobin. 2,3-BPG is synthesized from an intermediate of the glycolytic pathway. • Binding of 2,3-BPG to deoxyhemoglobin: 2,3-BPG decreases the oxygen affinity of hemoglobin by binding to deoxyhemoglobin. This binding stabilizes the taut conformation of deoxyhemoglobin. HbO2 + 2,3-BPG Hb–2,3-BPG + O oxyhemoglobin deoxyhemoglobin • One molecule of 2,3-BPG binds to a pocket, formed by the two β-globin chains, in the center of the deoxyhemoglobin tetramer. This pocket contains several positively charged amino acids that form ionic bonds with the negatively charged phosphate groups of 2,3-BPG. • Shift of the oxygen dissociation curve: The presence of 2,3-BPG significantly reduces the affinity of hemoglobin for oxygen, shifting the oxygen dissociation curve to the right. This reduced affinity enables hemoglobin to release oxygen efficiently found in the tissues. • Response of 2,3-BPG levels to chronic hypoxia or anemia: The concentration of 2,3-BPG in the RBC increases in response to chronic hypoxia, such as that observed in chronic obstructive pulmonary disease (COPD) like emphysema, or at high altitudes, where circulating hemoglobin may have difficulty receiving sufficient oxygen.
- 29. 1. Amino acid substitution in Hb S β chains: A molecule of Hb S contains two normal α-globin chains and two mutant β globin chains (βS ), in which glutamate at position six has been replaced with valine. 2. Sickling and tissue anoxia: The replacement of the charged glutamate with the nonpolar valine forms a protrusion on the β-globin that fits into a complementary site on the β chain of another hemoglobin molecule in the cell. At low oxygen tension, deoxyhemoglobin S polymerizes inside the RBC, forming a network of fibrous polymers that stiffen and distort the cell, producing rigid, misshapen erythrocytes. Such sickled cells frequently block the flow of blood in the narrow capillaries. This interruption in the supply of oxygen leads to localized anoxia (oxygen deprivation) in the tissue, causing pain and eventually death (infarction) of cells in the vicinity of the blockage.
- 31. 1. H+ decreases affinity to O2 2. Blood to tissues → acidification 3. Binding of H+ to Hb conformation 4. Lung O2 to Hb, H+ release 5. Enhancement of pH 6. Blood – O2 binding, tissue – O2 release BOHR'S EFFECT Effect of H+ on oxygen binding by hemoglobin (Hb). A. In the tissues, CO2 is released. In the red blood cell, this CO2 forms carbonic acid, which releases protons. The protons bind to Hb, causing it to release O2 to the tissues. B. In the lungs, the reactions are reversed. O2 binds to protonated Hb, causing the release of protons. They bind to bicarbonate (HCO3 - ), forming carbonic acid which is cleaved to H2O and CO2, which is exhaled.
- 32. 1. Selection of protein source (tissues, cells, E.coli, yeasts, cloning) 2. Solubilization a. cytosol - osmotic lysis of cells by hypotonic solution + centrifugation b. membrane - homogenization + centrifugation 3. Stabilization - isolation in buffers, temperature, inhibitors of proteases PROTEIN ISOLATION PROTEIN PURIFICATION CHARACTERISTIC PROCEDURE 1. CHARGE 1.Ion-exchange chromatography 2. Electrophoresis 3. Isoelectric focusing 2. POLARITY 1. Adsorption chromatography 2. Paper chromatography 3.Reverse-phase chromatography 4.Hydrophobic chromatography 3. SIZE 1. Dialysis and ultrafiltration 2. Gel electrophoresis 3. Gel filtration chromatography 4. Ultracentrifugation 4. SPECIFICITY 1. Affinity chromatography
- 33. In the process of ion exchange, ions that electrostatically bound to an insoluble and chemically inert matrix are reversibly replaced by ions in solution: R + A- + = R + B- + A- A schematic diagram illustrating the separation of several proteins by ion exchange chromatography using stepwise elution. A device for generating a linear concentration gradient. Two connected open chambers, which have identical cross-sectional areas, are initially filled with equal volumes of solutions of different concentrations. As the column is washed, a process known as elution, those proteins with relatively low affinities to ion exchanger move through the column faster than the proteins that bind to the ion exchanger with higher affinities. ION-EXCHANGE CHROMATOGRAPHY
- 34. The molecules are separated according to their size and shape Molecules with molecular masses ranging below the exclusion limit (the molecular mass of the smallest molecule unable to penetrate the pores) of a gel will elute from the gel in the order of their molecular masses, with the largest eluting first. Dialysis is a process that separates molecules according to size through the use of semipermeable membranes containing pores of less then macromolecular dimensions GEL FILTRATION CHROMATOGRAPHY At start of dialysis (a) (b) At equilibrium Dialysis bad Buffer Concentrated solution
- 35. Molecule known as a ligand, specifically binds to the protein of interest is covalently attached to an inert and porous matrix The separation of macromolecules by affinity chromatography. The formation of cyanogen bromide- activated agarose (top) and its reaction with a primary amine to form a covalently attached ligand for affinity -chromatography (bottom). Examples of the various types of nucleophilic groups that can be covalently attached to epoxy-activated agarose via reation with its epoxide groups. AFFINITY CHROMATOGRAPHY
- 36. ELECTROPHORESIS Migration of ions in an electric field Polyacrylamide and agarose Paper electrophoresis, (a) A schematic diagram of the apparatus used. The sample is applied to a point on the buffer-moistened paper. The ends of the paper are dipped into reservoirs of buffer in which the electrodes are immersed and an electric field is applied. Uncharged molecules remain the point of sample application. Visualization with amido black, Coomassie brilliant blue, silver stain, radioactivity – + – + Cathode Cathode Plastic frame Plastic frame Anode Anode Buffer Buffer Sample wells Sample wells Sample Stacking gel Running gel
- 37. [CH3-(CH2)10-CH2-O-SO3 - ]Na+ – sodium dodecyl sulfate SDS-treated protein stend to have identical charge-to-mass ratios and similar shapes. SDS-PAG ELECTROPHORESIS The SDS – polyacrylamide electrophoresis pattern of the supernatant (left) and membrane fractions (right) of various strains of the Salmonella typhimurium. Samples of 200-µg of protein each were run in parallel lanes on a 35-cm - long x 0.8- mm-thick slab gel containing 10% polyacrylamide. The lane marked MW contains molecular weight standards. A logarithmic plot of the molecular masses of 37 different polypeptide chains ranging from 11 to 70 kD versus their relative electrophoretic mobilities on an SDS-polyacrylamide gel. Many proteins contain more than one polypeptide chain → SDS treatment disrupt the noncovalent interactions between these subunits
- 38. If a mixture of proteins is electrophoresed through a solution having stable pH gradient in which the pH smoothly increases from anode to catode, each protein will migrate to the position in the pH gradient corresponding to its isoelectric point. IMMUNOBLOTTING ISOELECTRIC FOCUSING The detection of proteins by immunoblotting
