AQA Physics P6 [ Summary Answers ]
•
0 likes•753 views
Ans for AQA Physics P6 [ Summary Ans. ]
Report
Share
Report
Share
Download to read offline
Recommended
More Related Content
What's hot
What's hot (20)
Similar to AQA Physics P6 [ Summary Answers ]
Similar to AQA Physics P6 [ Summary Answers ] (20)
Serway, raymond a physics for scientists and engineers (6e) solutions

Serway, raymond a physics for scientists and engineers (6e) solutions
Engineering Fluid Mechanics 10th Edition Elger Solutions Manual

Engineering Fluid Mechanics 10th Edition Elger Solutions Manual
66.-Merle C. Potter, David C. Wiggert, Bassem H. Ramadan - Mechanics of Fluid...

66.-Merle C. Potter, David C. Wiggert, Bassem H. Ramadan - Mechanics of Fluid...
Recently uploaded
https://app.box.com/s/7hlvjxjalkrik7fb082xx3jk7xd7liz3TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...

TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...Nguyen Thanh Tu Collection
Recently uploaded (20)
21st_Century_Skills_Framework_Final_Presentation_2.pptx

21st_Century_Skills_Framework_Final_Presentation_2.pptx
HMCS Max Bernays Pre-Deployment Brief (May 2024).pptx

HMCS Max Bernays Pre-Deployment Brief (May 2024).pptx
HMCS Vancouver Pre-Deployment Brief - May 2024 (Web Version).pptx

HMCS Vancouver Pre-Deployment Brief - May 2024 (Web Version).pptx
Play hard learn harder: The Serious Business of Play

Play hard learn harder: The Serious Business of Play
On National Teacher Day, meet the 2024-25 Kenan Fellows

On National Teacher Day, meet the 2024-25 Kenan Fellows
TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...

TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...
dusjagr & nano talk on open tools for agriculture research and learning

dusjagr & nano talk on open tools for agriculture research and learning
Simple, Complex, and Compound Sentences Exercises.pdf

Simple, Complex, and Compound Sentences Exercises.pdf
AQA Physics P6 [ Summary Answers ]
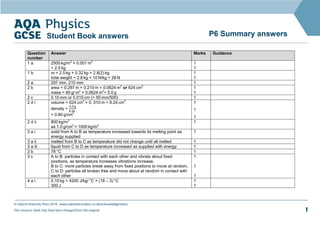
- 1. © Oxford University Press 2016 www.oxfordsecondary.co.uk/acknowledgements This resource sheet may have been changed from the original. 1 P6 Summary answers Student Book answers Question number Answer Marks Guidance 1 a 2500 kg/m3 × 0.001 m3 = 2.5 kg 1 1 1 b m = 2.5 kg + 0.32 kg = 2.8(2) kg total weight = 2.8 kg × 10 N/kg = 28 N 1 1 2 a 297 mm, 210 mm 1 2 b area = 0.297 m × 0.210 m = 0.0624 m2 or 624 cm2 mass = 80 g/ m2 × 0.0624 m2 = 5.0 g 1 1 2 c 0.10 mm or 0.010 cm (= 50 mm/500) 1 2 d i volume = 624 cm2 × 0. 010 m = 6.24 cm3 density = 5.0 g 8.24 = 0.80 g/cm3 1 1 1 2 d ii 800 kg/m3 as 1.0 g/cm3 = 1000 kg/m3 1 3 a i solid from A to B as temperature increased towards its melting point as energy supplied 1 3 a ii melted from B to C as temperature did not change until all melted 1 3 a iii liquid from C to D as temperature increased as supplied with energy 1 3 b 78 °C 1 3 c A to B: particles in contact with each other and vibrate about fixed positions, as temperature increases vibrations increase, B to C: more particles break away from fixed positions to move at random, C to D: particles all broken free and move about at random in contact with each other 1 1 1 4 a i 0.10 kg × 4200 J/kg/ °C × (18 – 3) °C 300 J 1 1
- 2. © Oxford University Press 2016 www.oxfordsecondary.co.uk/acknowledgements This resource sheet may have been changed from the original. 2 P6 Summary answers Student Book answers Question number Answer Marks Guidance 4 a ii 6300 J 450 s = 14 J/s 1 1 4 b i from 3 ºC to 0 ºC = 0.10 kg x 4 200 J/kg x (3 – 0) ºC = 1 260J or 1.26 kJ to freeze ΔE = 0.10 kg x 340 kJ/kg = 34 kJ or 34 000 J total = 34 000 J + 1260 J = 35 260 J or 35.3 kJ 1 1 1 1 4 b ii time taken at energy transfer rate of 14 J/s = 35 260 J 14 J/s = 2520 s or 42 minutes 1 1 5 a 3 000 W x 30 s = 90 000 J 1 1 5 b 90 000 J 2300 000 J/kg = 0.039 kg 1 1 GCSE Physics only 6 a 120 000 Pa × 25 cm3 100 000 Pa = 30 cm3 1 1
- 3. © Oxford University Press 2016 www.oxfordsecondary.co.uk/acknowledgements This resource sheet may have been changed from the original. 3 P6 Summary answers Student Book answers Question number Answer Marks Guidance 6 b pressure falls because internal energy of molecules decreases and average speed decreases, so collide less often with surface of container, when they collide with surfaces , individual impacts exert less force on surface, so total force per unit area molecules exert on surface (i.e., gas pressure) reduced 1 1 1 1 7 a particles in gas move at random and average separation much greater than in liquid or solid state, for given number of particles, mass is same in each state whereas volume occupied as gas much greater than in solid or liquid, since density = mass/volume, density of a gas much less than density of solid or liquid 1 1 1 7 b particles in liquid move at random in contact with each other, particles in solid hold each other together in fixed shape and vibrate about mean positions, when liquid cools and temperature falls, particles move slower, until slow enough for forces of attraction between them to bond them together in a fixed shape 1 1 1 1