Mass Spectrometry.ppt
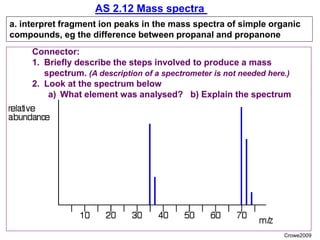
- 1. AS 2.12 Mass spectra a. interpret fragment ion peaks in the mass spectra of simple organic compounds, eg the difference between propanal and propanone Crowe2009 Connector: 1. Briefly describe the steps involved to produce a mass spectrum. (A description of a spectrometer is not needed here.) 2. Look at the spectrum below a) What element was analysed? b) Explain the spectrum
- 2. 1. Vaporisation of atoms or molecules; 2. Ionisation of atoms or molecules; 3. Acceleration of ions; 4. Deflection of ions; 5. Detection of ions. The steps involved to produce a mass spectrum:
- 3. Mass spectrum of chlorine, Cl2 Isotopes Cl-35 & Cl-37 Molecules Cl2-70 Cl2-72 Cl2-74
- 4. Mass Spectra of Organic Compounds 1. The molecular ion When an organic compound is bombarded by a stream of high energy electrons in the spectrometer: An electron is removed from the molecule, M, to form the molecular ion, M+. This ion will have the highest m/z value on the spectrum, and the value will correspond to the molecular weight of the compound.
- 5. Mass Spectra of Organic Compounds 2. Fragmentation patterns The molecular ions are energetically unstable, and some of them will break up into smaller pieces, e.g. The uncharged free radical (Y ) won't produce a line on the mass spectrum. Since only charged particles are detected by the mass spectrometer. All sorts of fragmentations of the original molecular ion are possible, and these help with the identification of the parent compound.
- 6. Mass Spectra of Organic Compounds 3. Fragmentation patterns The base peak in a mass spectrum is the tallest peak. The most abundant ion is responsible for the base peak. The base peak is given a value of 100. All other peaks are reported as percentages of the abundance of the base peak
- 7. Methanol The mass spectrum of methanol shows peaks at 32,31, 29, 28 and 15. Which line is the base peak? 32 represents the molecular ion, CH3OH+ Identify the other fragments. CH3O+ CHO+ CO+ CH3 +
- 8. Ethanol and methoxymethane have the same molecular formula, C2H6O, but their mass spectra are quite different: Identify them.
- 9. 45 29 15 17
- 10. 31 15 45
- 11. Work sheet – AS 2.12 mass spectra