Introduction to Extractive Metallurgy Course and Methods
- 1. MME-213 Extractive Metallurgy Week 1 Introduction to the course, some basic terms
- 2. Text books
- 4. Tentative Lecture Plan Week Contents CLOs Week 1 Introduction to the course, the importance of extractive metallurgy, and sources of non- ferrous metals. CLO1 Week 2 Methods of beneficiation, crushing, grinding, sizing, magnetic separation, electrostatic separation. CLO1, CLO3 Week 3 Methods of mineral beneficiation: gravity concentration and froth floatation General methods of extraction: pyrometallurgy, Calcination, roasting and its types CLO1, CLO2, CLO3, CLO4 Week 4 Pyrmetallurgy: Smelting, the role of vacuum in pyrometallurgy Hydrometallurgy leaching: solutions, operation, kinetics, and mechanisms of leaching CLO 1, CLO2, CLO3, CLO4 Week 5 Comparison of hydrometallurgy and pyrometallurgy Introduction to electrometallurgy, Quiz 1 CLO3 Week 6 Electrowinning and electrorefining CLO1, CLO2, CLO3, CLO4 Week 7 General methods of refining CLO1 CLO3 Week 8 Extraction of metals from oxides: extraction of Mg CLO1, CLO2
- 5. Tentative Lecture Plan Week Contents CLOs Week 10 Extraction of metals from oxides: extraction of Aluminum CLO1, CLO2, CLO3, CLO4 Week 11 Extraction of ferroalloys, Extraction of metals from sulfides: extraction of Cu CLO1, CLO2, CLO3, CLO4 Week 12 Extraction of Copper cont...., extraction of Zinc CLO1, CLO2, CLO3, CLO4 Week 13 Extraction of reactor metals CLO 1, CLO2, CLO3, CLO4 Week 14 Extraction of metals from halides: extraction of reactor metals, quiz 2 CLO1, CLO2, CLO3, CLO4 Week 15 Extraction of metals from halides: extraction of reactor metals cont. Extraction of metals from halides: extraction of reactor metals, quiz 2 CLO1, CLO2, CLO3, CLO4 Energy, cost, and environment-related problems in industry exercising non-ferrous extractive metallurgy. . CLO3, CLO4 Week 16
- 6. A brief overview of modern day extractive metallurgy • Metals exist in nature in the combined form, i.e., in minerals. Mineral Ore Naturally occurring inorganic compound of one or more metals in association with nonmetals such as oxygen, sulphur, and the halogens. All minerals are not ores. Clay is the mineral of Al. An ore is a naturally occurring aggregate or a combination of minerals from which one or more metals or minerals may be economically extracted. Bauxite and cryolite are the ores of Al.
- 7. Factors effecting economy of extraction • Percentage of metal in the ore • The percentage of impurities in the ore (which are difficult to remove) • The form in which the metal occurs, i.e., nature of mineral in the ore • The physical condition of the ore • The location and magnitude of the ore • The proximity to transport facilities • The market value of the metal
- 8. Gold Quartz A gold ore What is gangue in this ore? How will we separate it out?
- 9. Comminution • Ores → available in big lumps → unfit for extraction processes • Ores → valuable + waste frac on (gangue) • Comminu on → par cle size of ore is reduced. • Comminu on → mineral of interest is separated from gangue • Crushing → reduces par cles size to 6mm • Grinding → reduces par cle size below 6 mm
- 10. Mineral Dressing • Ore dressing is almost always needed before extraction operation. • Ores must be concentrated → gangue must be separated. • Gravity concentration methods (Jigging, tabling, heavy media separation) • Magnetic separation • Electrostatic separation • Froth flotation
- 11. Extractive Metallurgy • Pyrometallurgy - deals with the methods of extraction and refining of metals by processes involving high temperatures • Calcination CaCO3 → CaO + CO2 850 °C
- 13. Extractive Metallurgy • Pyrometallurgy - deals with the methods of extraction and refining of metals by processes involving high temperatures • Calcination: thermal treatment of an ore decomposition and release of volatile products products usually are CO2 and H2O Why calcination requires high temperatures and how can those temperatures be determined?
- 14. Extractive Metallurgy • Pyrometallurgy - deals with the methods of extraction and refining of metals by processes involving high temperatures • Roasting: precedes smelting in pyrometallurgy and leaching in hydrometallurgy. mineral constituent of an ore is converted into another chemical form. calcina on → decomposi on of an ore roas ng → chemical conversion of an ore Example ZnS + (g) → ZnO + S (g) Types of roasting: • Oxidizing roasting • Volatilizing roasting • Chloridizing roasting • Magnetic roasting
- 15. Extractive Metallurgy • Pyrometallurgy - deals with the methods of extraction and refining of metals by processes involving high temperatures • Smelting: heating process for the production of metal or matte. process of melting and separating charge into two immiscible liquid layers, i.e. liquid slag and liquid matte or liquid metal. What is a matte? Mattes are mixed molten sulphides of heavy metals
- 16. Extractive Metallurgy • Hydrometallurgy – refers to the application of aqueous solutions for metal recovery from ores. • Leaching: metallic values in an ore ae selectively dissolved using a suitable liquid reagent. selectivity in dissolution depends on the nature of the reagent. metal rich solution is later processed for metal recovery. highly effective for lean ores. What is leaching?
- 17. Extractive Metallurgy • Hydrometallurgy – refers to the application of aqueous solutions for metal recovery from ores. • Leaching Photographs of raw ore (a), sulfuric acid (H2SO4, 0.15 M)- treated ore (b) (after a weeklong, 7 days experiment), and cross-section of leached ore (c) (1, 2: ore containing malachite before reaction; 1′, 2′: ore after malachite leaching reaction). Taken from https://doi.org/10.3390/min11091020
- 18. Extractive Metallurgy • Hydrometallurgy – refers to the application of aqueous solutions for metal recovery from ores. • Leaching Taken from https://doi.org/10.3390/min11091020 The next process is recovering of the metallic values from leach liquor Processes include: • Precipitation • Cementation • Electrolysis • Ion Exchange • Solvent extraction will go through them in the upcoming classes
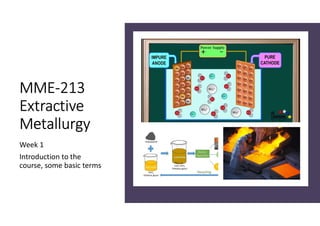
- 19. Extractive Metallurgy • Electrometallurgy – refers to all metallurgical processes which utilize electricity and electrical effects. • uses electrolysis to extract the metal. • Electrowinning extraction of metal from the electrolyte. used extensively for the extraction of reactive metals like Al and Mg. Alternative to pyrometallurgical extraction for metals like Cu and Zn Inert anode Metal ions in solutions Metal from solution is deposited on the cathode
- 20. Extractive Metallurgy • Electrometallurgy – refers to all metallurgical processes which utilize electricity and electrical effects. • uses electrolysis to extract the metal. • Electrorefining refining of the impure metal which is in the form of anode. crude metal goes into the electrolyte pure metals from solution deposits on the cathode Animation taken from https://www.ausetute.com.au/electrorefine.html
- 21. Unit operation and unit process Unit operation → a step which is characterised by certain physical features (no chemical changes takes place) Crushing, grinding, sizing, mixing etc are unit operations Unit processes → refers to the steps involving chemical reactions Roasting, calcination, smelting, leaching, etc are unit processes