SWAS FAMILIARIZATION.pptx
- 1. SWAS FAMILIARIZATION Presented By : Nihar Behera, CPP Operations, Paradeep
- 2. CONTENTS • SWAS requirement ? • Sampling Conditioning / Wet Panel • Schematic diagram of Sample inlet • Important equipment of Sample System • Sample Analysis • Do’s & Don'ts • Conclusion
- 3. Need for Boiler water treatment : • Inhibits corrosion. • Prevent freezing of water in the system. • Increase the boiling point of water in the system. • Inhibits the growth of mold & bacteria. • Allow improved leak detection.
- 4. SWAS Requirement ? • Steam and water analysis system (SWAS ) : It is a system dedicated to the analysis of steam or water. In power stations, it is usually used to analyze boiler steam and water to ensure the water used to generate electricity is clean from impurities which can cause corrosion to any metallic surface, such as in boiler and turbine. • In any power plant running on steam, the purity of boiler feed water and steam is absolutely crucial; especially to steam turbine, steam boiler, super heater, condenser and other steam equipment. • To prevent damage of steam turbine, steam boiler and other apparatus due to scaling and corrosion, online steam and water analysis of critical parameters such as pH, Conductivity, Dissolved Oxygen, Silica, Sodium, Phosphate etc. is a must.
- 5. SWAS Requirement ? • To keep the power plant up and running, with minimum erosion and corrosion of steam turbine, steam boiler and condenser SWAS works in two stages: 1. Sample Conditioning 2. Sample Analysis
- 6. Sample Conditioning or Wet Panel
- 7. Sample Conditioning or Wet Panel • The Sample is First cooled in Sample Coolers, depressurized in pressure regulators and then fed to various analyzers while keeping the flow characteristics constant by means of a Back Pressure Regulating Device. • There are a lots of safety equipment provided in wet panels, so that the operators feel safe while working with these systems.
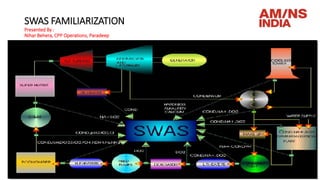
- 8. Schematic Diagram of Sample Flow (P&ID discussion)
- 9. Important Components of Sampling System(Wet Panel) 1. SAMPLE COOLER • Sample Coolers are COIL-IN-SHELL type CONTRA-FLOW heat exchangers. • Coil Materials are available such as Stainless Steel AISI 316, Monel 400 & Inconel 62.5 and so on.
- 10. Important Components of Sampling System(Wet Panel) 2. HIGH PRESSURE REGULATOR • Piston Type High Pressure Regulators. • These are used in primary conditioning where sample pressures are higher than 100 Kgicm2. • As these are piston type Pressure Regulators , there is no fear of diaphragm rupture etc.
- 11. Important Components of Sampling System(Wet Panel) 3. BACK PRESSURE REGULATOR • Back Pressure Regulator(BRP) to avoid low flow (or fluctuating flow) conditions to analyzers in the event grab sample valve operation. In the absence of such a device ,the sample would flow to grab sample line when the grab sample valve is opened. This can create low flow alarm conditions in sample going to analyzers. • A pressure Regulator and Back Pressure Regulator combination provides very stable pressure &. flow conditions, thereby ensuring reliable, efficient and accurate analysis.
- 12. Important Components of Sampling System(Wet Panel) 4. SIGHT GLASS • Sample flow Indicator (sight Glass) to view the sample flow inside the sample line. • A rotating wheel indicates presence of cooling water. • The sight glass is made of high grade stainless steel.
- 13. Important Components of Sampling System(Wet Panel) 6. PRESSURE RELIEF VALVE • Pressure relief Valve comes fitted with Sample Cooler. • Pressure Relief Valve is important as it protects the Sample Cooler in case the coil fails. • This is also important for human safety as pressurized cooler may burst due to full sample pressure under coil failure conditions.
- 14. Important Components of Sampling System(Wet Panel) 7. HIGH- TEMP ISOLATION VALVE • This valve is an easy to operate & can be used for most high pressure and temperature applications. • Its unique plug/seat geometry and stuffing box design allow these valves to operate for extended period of time without gland leakage and passing.
- 15. Important Components of Sampling System(Wet Panel) 8. CATION COLUMN • The Duplex type. Cation Conductivity Column is a field proven design. • The Cation Conductivity measurements are considered to be more reliable than ordinary conductivity measurements. • This ensures Elimination Of Masking Effect of desired chemicals used in treating the water.
- 16. Sample Analysis : • 1. Feed Water : pH, Specific Conductivity, DO, Silica, Sodium, Phosphate, Hydrazine. • 2. CBD : pH, Specific Conductivity. • 3. Main Steam : pH, Cation Conductivity. • 4. CEP Discharge : pH, Specific Conductivity, Cation Conductivity.
- 17. pH Analyzer : • pH is a measure of the acidity and alkalinity properties of a water solution, which are determined by the concentration of hydrogen ions (H+) present in the solution. • pH is defined as pH = -log10 H+ • Acids : HA = H+ (hydrogen ion) + A- (anion) • Bases : MOH = OH- (hydrogen ion) + M+ (cation)
- 18. Why pH measurement required ? • The steam which goes to the turbine has to be ultra pure. • The pH value of the feed water gives direct indication of alkalinity or acidity of the water. The ultra pure water has pH value of 7. • In a steam circuit, to keep the pH value of feed water at slight alkaline levels. • It helps in preventing the corrosion of pipe work and other equipment. • pH Analyzers are recommended at following location in a steam circuit : high pressure heaters, DM Makeup, CEP discharge.
- 19. Conductivity Analyzer : • Conductivity is the ability of a solution, a metal or a gas- in brief all materials - to pass an electric current. • In solutions the current is carried by cation and anions whereas in metals it is carried by electrons. • How well a solution conducts electricity depends on a number of factors : 1. Concentration 2. Mobility of ions 3. Valence of ions 4. Temperature
- 20. Conductivity : • Electricity is the flow of electrons. This indicates that ions in solution will conduct electricity. Conductivity is the ability of a solution to pass current. The conductivity reading of a sample will change with temperature. • k = G x K • k = conductivity (S/cm) • G = conductance (S) , where G = 1/R • K = Cell Constant (cm-1)
- 21. Conductance : • Conductance (G) is defined as the reciprocal of the electrical resistance (R) of a solution between two electrodes. • G = 1/R (S) • The conductivity meter in fact measures the conductance and displays the reading converted into conductivity.
- 22. Cell Constant : • This is the ratio of the distance (d) between the electrodes to the area (a) of the electrodes. • K = d / a K = Cell constant (cm-1) a = effective area of the electrodes (cm2) d = distance between the electrodes (cm)
- 23. Conductivity Solution : • Conductivity is typically measured in aqueous solutions of electrolytes. • Electrolytes are substances containing ions, i.e. solutions of ionic salts or of compounds that ionize in solution. • The ions formed in solution are responsible for carrying the electric current. • Electrolytes include acids, bases and salts and can be either strong or weak.
- 24. Why Conductivity Measured ? • Conductivity is an important parameter for detecting any contamination of steam in the boiler circuit. • Conductivity of pure water is almost zero. • Ingress of any kind of dissolved impurity will raise conductivity instantly. • Thus conductivity is an important parameter for the detection of leakages.
- 25. Indications of the problem 1. Conductivity decreasing before the cation column. • Probable Reasons: • Boiler water tube leakage. • Boiler blowdown is in progress. • Boiler drain valves or blowdown valves passing. • Instrument reading wrongly due to fault or loss of sample flow. • Corrective Action: • Confirm tube leak and dose caustic soda through HP dosing system & maintain conductivity. • Check drain valves & blowdown valves for any leak & attend. • Check for free flow of sample or rectify instrument if needed.
- 26. Indications of the problem 2. Conductivity increasing before the cation column • Probable Reasons: • Condenser tube leakage. • Boiler HP dosing is in progress. • Instrument reading wrongly due to fault or loss of sample flow. • Corrective Action: • Confirm condenser tube leak and take action to maintain conductivity till Unit shutdown. • Check for free flow of sample or rectify instrument if needed.
- 27. Indications of the problem 3. Conductivity decreasing after the cation column • Probable Reasons: • Boiler water tube leakage. • Boiler blowdown is in progress. • Boiler drain valves or blowdown valves passing. • Corrective Action: • Confirm tube leak, check conductivity before cation column and dose caustic soda through HP dosing system & maintain conductivity. • Check drain valves & blowdown valves for any leak & attend.
- 28. Indications of the problem 4. Conductivity increasing after the cation column • Probable Reasons: • Condenser tube leakage. • Cation column getting exhausted. • Contamination of make up water from WTP. • Instrument reading wrongly due to fault or loss of sample flow. • Corrective Action: • Confirm condenser tube leak and take action to maintain conductivity till Unit shutdown. • Inform chemist regarding increase in conductivity after checking other possible causes. • Ensure final treated water outlet conductivity. • Check for free flow of sample or rectify instrument if needed.
- 29. Silica Analyzer • The Presence of silica in the steam and water circuits of a power generation plant is associated with a number of problems both in the Super Heater and Turbine sections. • The solubility of silica in steam increases with pressure. Hence there are chances of silica carryover. • The Presence of Silica in the steam can lead to deposition in Superheated tubes and on Turbine Blades which may lead to loss of efficiency and Turbine blade Failure • Silica in the steam cycle can result in deposition of a "glass“ layer on surfaces, resulting in a loss of thermal process efficiency. • Deposition of silica on the turbine blades can result in the turbine becoming imbalanced, reducing efficiency and, in extreme cases, causing extensive damage to the turbine.
- 30. Dissolved Oxygen • The DO determination measures the amount of dissolved (or free) oxygen present in water or wastewater. • At elevated temperature dissolved oxygen causes corrosion which may cause puncture and failure of piping and components respectively. • Dissolved oxygen also promotes electrolytic action between dissimilar metals causing corrosion and leakage at joints and gaskets. • Mechanical Deaeration and chemicals scavengers additives are used top remove the DO. • DO monitoring is imperative in power stations using neutral or combined operating conditions (pH 7.0-7.5 or 8.0-8.5) • In steam Circuit where DO monitoring is required are Deaerator Inlet and Outlet (Feed water, Condenser & Deaerator Outlet) .
- 31. Dissolved Oxygen Working Principle • The measurement of dissolved oxygen is based on the well known Clark cell principle. • An oxygen-permeable membrane isolates the electrodes from the sample water, thus obviating the need for sample conditioning. • A gold working electrode (cathode) reduces the dissolved oxygen to hydroxyl ions: O2 + 2H20 + 4e- = 4OHA • A large silver counter electrode (anode) provides the oxidation reaction which occurs on its surface: 4Ag+ + 4Br = 4AgBr + 4e- • The reduction of oxygen is the current limiting reaction, thus making the cell current linearly proportional to the dissolved oxygen concentration • Electrochemical reactions and diffusion rates are temperature-sensitive.
- 32. Phosphate Analyzer • This treatment is used to precipitate the hardness constituents of water and provide alkaline pH control, which will reduce boiler corrosion. • Maintains the sodium-to- phosphate molar ratio — (2.1 to 2.9) • This ratio must be maintained to prevent formation of phosphoric acid (ratio below 2.1) or free sodium hydroxide (ratio above. 2.9) • The use of phosphate analyzer is to provide a safe alkaline environment in the boiler.
- 33. Phosphate Analyzer • Phosphate treatment serves two basic purposes : 1. Phosphate controls the pH in the range that is least corrosive to carbon steel. 2. The event of a condenser leakage or other process upset, phosphate or the alkalinity produced by it, reacts with Ca, Mg, Si and other minerals to produce soft sludge that can be blown down.
- 34. Parameters Analyzing Abnormal Data Examples Expected Symptoms Serious Problems Feed water pH high Excessive chemical dosing Cu-alloy corrosion Feed water pH low Impurity contamination & see water leakage Leakage from boiler tubes Boiler water pH high Excessive chemical dosing Cu-alloy corrosion Boiler water pH low Impurity contamination & see water leakage Leakage from boiler tubes Feed water O2 high Deaerator performance degradation. Low condenser vacuum. Cu-alloy corrosion Boiler water silica high Boiler blowdown rate insufficient Turbine performance degradation Feed water conductivity high Impurity contamination & see water leakage Leakage from boiler tubes Steam conductivity high Impurity contamination & see water leakage Leakage from boiler tubes
- 35. SWAS Do’s & Don’ts Do’s : • Always keep the coolants flowing even if sample is stopped. • Check the loose wiring connections. • Always flush all the sample & coolant lines before operating. • Check for leaks in both sample & coolant lines. • Check whether the connections to valves, pressure regulator, flow indicators etc. are in right direction. • Always switch off the main power supply while carrying out any maintenance on the system.
- 36. SWAS Do’s & Don’ts Don’ts : • Don’t disturb the settings of pressure regulator, temperature switch, pressure switch and other valves without consulting the Service Engineer. • Don’t stop the coolant supply, before isolating the sample supply. • Don’t carry any maintenance function without isolating the sample supply.
- 37. Conclusion • Water treatment technologies for Thermal Power Plants have been examined and improved as a counter measure against damage due to factors such as corrosion and scale deposition. • As discussed herewith abnormalities in water quality can be a precursor of problems and therefore serious problems can be prevented by analyzing the data and taking necessary measures. • Thank You All !!!!!!!!!