Final mab DI question presentation.docx
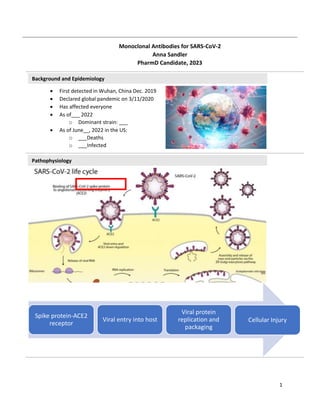
- 1. 1 Spike protein-ACE2 receptor Viral entry into host Viral protein replication and packaging Cellular Injury Background and Epidemiology Pathophysiology First detected in Wuhan, China Dec. 2019 Declared global pandemic on 3/11/2020 Has affected everyone As of___ 2022 o Dominant strain: ___ As of June__, 2022 in the US: o ___Deaths o ___Infected Monoclonal Antibodies for SARS-CoV-2 Anna Sandler PharmD Candidate, 2023
- 2. 2 Cellular injury, Cytokine release RAAS dysfunction Pro-inflammatory Clots, Multi-system inflammatory syndrome Diagnosis and Presentation Monoclonal Antibodies Tests: o PCR to detect genetic material (RNA) o Antigen tests to detect viral proteins Presentation (~4-5 days following exposure): o Fever, cough, myalgias o N/V o Smell and taste abnormalities Complications: o ARDS o Thromboembolic o Neurologic o Long-COVID Transmission: Respiratory Droplets Received EUA from FDA Bind to spike protein to block attachment to ACE2 receptor Use: Immunocompromised patients Prophylaxis or Treatment Adults Children ≥ 12 y/o and ≥ 40 kg* Bebtelovimab Etesevimab Casirivinmab Sotrovimab Imdevimab Tixagevimab/cilgavimab Bamlanivimab/etesevimab ARDS: Acute Respiratory Distress Syndrome; RAAS: Renin-Angiotensin-Aldosterone System; EUA: Emergency Use Authorization * With the one exception being bamlanivimab/etesevimab
- 3. 3 Prophylaxis: As of May 23, 2022 Treatment: As of May 23, 2022 Pre-exposure Who: Moderately or severely immunocompromised patients at increased risk of inadequate immune response, or those who cannot get the vaccine Post-exposure Who: Persons exposed to COVID-19 at high risk of progressing to severe disease tixagevimab/cilgavimab casirivimab/imdevimab balanivimab/etesevimab Indication: Non-hospitalized patients without supplementary O2 requirements Mild-to-moderate disease at risk to progress to severe disease Unable to receive ritonavir-boosted nirmatrelvir (PaxlovidTM ) or remdesivir bebtelovimab sotrovimab casirivimab/imdevimab balanivimab/etesevimab Immunosuppressive treatments or diseases >/= 65 y/o Chronic organ diseases: CV, Lung, Kidney Pregnancy Immunocompromising conditions Non-fully vaccinated individuals Nursing home or prison residents
- 4. 4 Brand/generic Date EUA Indication Dosing Adverse Drug Reactions Pearls Tixagevimab/Cilgavimab (EvusheldTM ) Dec 2021 Pre-exposure prophylaxis 300 mg/300 mg IM as a single dose administered in 2 separate syringes consecutively Dizziness, fatigue, headache, cough, hypersensitivity, anaphylaxis Warnings and Precautions: CV events Higher reporting of MI and cardiac failure Can be used in hospitalized or non- hospitalized patients Ineffective against Omicron BA.1 and BA.1.1 Bebtelovimab Feb 2022 COVID-19 Treatment 175 mg IV X 1 within 7 days of symptom onset Pruritis, infusion-related reactions, N/V Monitor patients after administration Consider slowing or stopping infusion if reaction occurs Effective against all strains Casirivimab/imdevimab (REGEN-COVTM ) Nov 2020 COVID-19 treatment Post- exposure prophylaxis 600 mg/600mg IV or SubQ once Within 10 days of symptom onset Erythema at injection site, pruritis, N/V, hypersensitivity Post-exposure prophylaxis for use in Any patient DISTRIBUTION PAUSED Sotrovimab (XevudyTM ) May, 2021 COVID-19 treatment 500 mg IV as single dose as soon as possible after positive result and within 7 days of symptom onset Skin rash, diarrhea, hypersensitivity reaction, anaphylaxis Warnings and precautions: Hypersensitivity, infusion-related reactions, resistance DISTRIBUTION PAUSED Retains activity against Omicron BA.1 and BA.1.1 subvariants, decreased activity against BA.2 Balanivimab/etesevimab Feb 2021 COVID-19 treatment Post- exposure prophylaxis 700 mg/1.4g as a single dose administered together as a single IV infusion, within 10 days of symptom onset Pruritis, nausea, anaphylaxis, dizziness Can be used in children and in neonates Post-exposure prophylaxis for use in Any patient Ineffective against Omicron BA.1 and BA.1.1 LIMITED USE to susceptible regional variants Drug Table
- 5. 5 References NIH COVID-19 Treatment Guidelines IDSA COVID-19 Treatment Guidelines UpToDate CDC website Sriram K, Insel PA. Physiol Rev. 2021;101(2):545-567. doi:10.1152/physrev.00035.2020 Image Links Globe COVID life cycle RNA Rapid Test COVID and Organ Systems Evusheld N/V: Nausea and Vomiting; CV: Cardiovascular
- 6. 6