ASandler_PD_Topic Discussion_8_30.docx
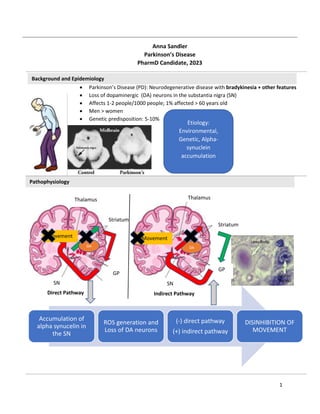
- 1. 1 Accumulation of alpha synucelin in the SN ROS generation and Loss of DA neurons (-) direct pathway (+) indirect pathway DISINHIBITION OF MOVEMENT Background and Epidemiology Pathophysiology Anna Sandler Parkinson’s Disease PharmD Candidate, 2023 Parkinson’s Disease (PD): Neurodegenerative disease with bradykinesia + other features Loss of dopaminergic (DA) neurons in the substantia nigra (SN) Affects 1-2 people/1000 people; 1% affected > 60 years old Men > women Genetic predisposition: 5-10% Etiology: Environmental, Genetic, Alpha- synuclein accumulation Thalamus Striatum GP SN DA Movement Striatum Thalamus GP SN Movement DA Direct Pathway Indirect Pathway
- 2. 2 Psychtoic Mood, Cognition Non- motor sx Sensory Sleep Autonomic 1: Exclusion can also be done by seeing whether a patient has a clear response to Levodopa treatment, which is not well seen with overlapping syndromes. Imaging studies can also help exclude bleeding, stroke, or massive lesions. 2: Typical /first generation antipsychotics are most implicated such as haloperidol and fluphenazine. Prokinetics drugs include metoclopramide, and valproic acid has been implicated in inducing parkinsonism with long-term use. No treatments have been shown to be disease-modifying or to be neuroprotective Diagnosis based on clinical symptoms (sx) and exclusion1 o Other conditions: Lew body disease, multiple system atrophy o Iatrogenic2 : Antipsychotics, CCBs, antiepileptics, and Li Cardinal features: Bradykinesia + Tremor and/or rigidity o Late manifestation: Gait disturbance Other features due to deposition of alpha-synuclein in other brain areas Treatment overview Hallucinations Impulse control Depression Anxiety Impaired judgement GI disturbances Sexual dysfunction Restless legs syndrome REM behavior sleep disorder Non-pharmacologic Non-motor symptoms Motor symptoms Managing side effects/resistance Diagnosis and Clinical Presentation Individualize
- 3. 3 Drug/Class Dosing Pearls, Adverse Effects (AEs), Monitoring Dopamine precursor Carbidopa- levodopa (Sinemet, Parcopa) Initial: 25/100 mg IR ½ tab BID-TID Titrate over several weeks to full tab TID as tolerated Take with meals or snack to avoid nausea Superior effects on PD motor sx and qol > other drugs Higher rate of adverse effects: dyskinesias Should not be abruptly stopped d/t withdrawl concern AEs/Monitoring: Nausea, somnolence,orthostatic hypotension (OH) headache (HA) (early, non-serious), delusions, agitation (older patients, serious), impulse- control disorders, LFTs, BUN, SCr, CBC, s/sx of peripheral neuropathy MAOBis: Selegiline Rasagiline Safinamide Selegiline: 5 mg PO BID Rasagiline: 0.5 mg PO once daily increased to 1 mg daily as tolerated Safinamide: 50 mg once daily titrated to 100 mg once daily AEs/monitoring: Nausea, HA, confusion, hallucinations, falls, insomnia dyskinesia, hypertensive crisis4 Avoid with TCAs or SSRIs, washout period of 14 days required, Safinamide usually as adjunct to levodopa to help w/ motor fluctuations Amantadine IR: 100 mg PO BID or TID ER: 100 mg daily Can be used as monotherapy (if tremor present)5 AEs/monitoring: Livedo reticularis and ankle edema, rarely tx-limiting, renal function, mental status, BP Managing motor symptoms Therapy may not be needed Pharmacological options: o Monoamine Oxidase type B inhibitor (MAOBi) o Amantadine o Levodopa (L-dopa) o Anticholinergic Mild </= 65 y/o Dopamine agonist (DA) L-dopa Maintaining Independence in Everyday Life with Non-Pharmacological Treatments Nutrition o At risk of malnutrition due to increased metabolic demands and taste alterations o No consensus on a specific diet but certain recommendations: high fiber, hydration, protein restriction?3 >65 y/o L-dopa> DA 3: Dietary protein restriction may be needed in advanced disease with motor fluctuations; competition with other amino acids may interfere with the absorption of Levodopa. 4: Doses of selegiline > 10 mg/day place patients at higher risk of a hypertensive crisis, impulse control disorders are more common in older patients with advanced , the serious AEs 5: Can be used as monotherapy in younger patients in PD, particularly when tremor is prominent, also useful in managing dyskinesias and off-time Exercise and Physical Therapy o Regular aerobic exercise: Walking, swimming, tai chi o Resistance training o Balance training o May slow motor progression and improve non- motor symptoms Moderate-severe L-dopa preferred at any age Mild-moderate Age Motor sx management: Drug Table
- 4. 4 Drug/Class Dosing Pearls, Clinical Trials, Adverse effects (AEs) DAs: Pramipexole, ropinirole, rotigotine Pramipexole: 0.125 mg TID (IR), increase by 0.125 mg every 5-7 days or 0.375 mg daily at bedtime (ER), titrated by 0.375 mg increments to max 1.5- 4.5 mg Ropinirole: 0.25 mg TID (IR), gradually increase by 0.25 mg/week for 4 weeks (max: 3 mg/day), or 2 mg daily at bedtime (ER), titrate by 2 mg increments to max of 24 mg Rotigotine: Once daily TD patch, 2mg/24 hours and titrated weekly by 2mg/24 hour increments to max of 6 mg/24 hours AEs/Monitoring: nausea, vomiting, renal function, OH, confusion, hallucinations, peripheral edema, impulse control disorders Start low and go slow, do not abruptly stop treatment Anticholinergics6 : Trihexyphenidyl Benztropine Trihexylphenidyl: 0.5mg-1 mg PO BID, gradully increased to 2mg daily Benztropine: 0.5-2 mg BID AEs/monitoring: dry mouth, blurred vision, constipation, tachycardia, intraocular pressure Sometimes useful as monotherapy for patients </= 65 y/o who have tremor, w/h significant bradykinesiaor gait disturbance. Can also be added in those unresponsive to L-dopa or DAs. Gradually d/c to avoid exacerbation of PD Side effect Management Other notes Nausea Additional carbidopa Add domperidone or PRN Ondansetron or trimethobenzamide Orthostasis May need to d/c or lower reduce anti- hypertensive medication Taper or withdraw the less-effective therapies prior to reducing L-dopa Impulse control disorders (ICDs) Persistent: Cognitive behavioral therapy? Education’ Reduce dose until ICD resolves Most commonly associated with DAs Risk factors: younger age, male, comorbid anxiety and depression Managing side effects/resistance to treatment Motor sx management: Drug Table continued No benefit of catechol-O- methyltransferase (COMT) inhibitors as initial therapy!7 Rescue DA: Apomorphine 6: DA depletion can lead to cholinergic sensitivity, resulting in exacerbation of PD sx by cholinergic drugs. Thus, anticholinergic drugs may be useful in PD, especially younger patients who have disturbing tremors. 7: The STRIDE-PD trial demonstrated that the combination of levodopa-carbidopa with entacapone (LCE) resulted in an increased frequency of dyskinesias compared to levodopa-carbidopa monotherapy (LC); frequency at week 134: 42% versus 32%; p=0.02
- 5. 5 Side effect Management Other notes Wearing off Initial increases in levodopa dose Smaller but more frequent doses Add COMT inhibitors, DA or MAOBi Rescue therapy with apomorphine Longer-acting formulations Protein avoidance and/or taking med on empty stomach 40-60 min before or 60-90 min after a meal Save greater protein content for the evening Dose increases may lead to or worsen dyskinesias Doses in the evening may fail d/t failure to reach therapeutic threshold Use of a MAO B inhibitor may help prolong the half-life of DA in the brain Obtain baseline EKG before starting apomorphine Peak-dose dyskinesia Reduce levodopa dose Administer more frequently Use amantadine Clozapine in refractory dyskinesia Extensive monitoring required with clozapine: ANC, WBC, Unpredictable off periods Document and diaries Avoid protein around drug administration Failure of on response Avoid protein around drug administration Add a dose of levodopa one hour before the usual failure of “on” time Avoid Metoclopramide due to dopamine receptor block! Off period dystonia Take CR levodopa before bed time if occurs in the morning Take levodopa or DA during the night or first thing in the morning Use apomorphine rescue injection Monitor supine and standing BP and pulse, s/sx hemolytic anemia, OH, and mental status changes with apomorphine Akathisia Take CR levodopa or DA before bed Persistent tremor Add on amantadine or anticholinergic Deep brain stimulation if persists despite maximal therapy Managing Motor fluctuations and dyskinesias8 of levodopa Motor fluctuations: Motor fluctuations consist of on and off periods Wearing off: Emergence of PD sx as levodopa diminishes Unpredictable Off: No relationship between time of dose and appearance of off episode Failed response: Lack of a response following dose of levodopa 8: Due to the progressive nature of PD, presynaptic neurons actually start failing to store and release levodopa after enzymatic conversion to dopamine, resulting in a shorter half-life. Pulsatile stimulation of dopamine receptors may also be involved in the narrow therapeutic window. Other contributing factors include: reduced intestinal absorption due to poor gastric emptying, competition from dietary protein, and slow intestinal transit times. Avoid CR formulations in severe/complex dyskinesia patterns
- 6. 6 Symptom Management Other notes Erectile dysfunction Sildenafil Depression SNRI or SSRI, TCA if patients do not improve Sertraline, Paroxetine, venlafaxine Monitor: QTc especially if risk factors, exacerbation of motor symptoms or comorbid sleep disorders Caution SSRIs and exacerbation of motor symptoms Psychosis Potentially reducing doses of anti-PD drugs Quetiapine or clozapine in low doses > other antipsychotics Counsel on risk of stroke, MI, death Dementia Cholinesterase inhibitors Rivastigmine Donepezil Memantine Anxiety SSRIs, SNRIs, buspirone Citalopram, sertraline Psychotherapy Managing the non-motor symptoms of PD
- 7. 7 References: 1. Shin HW, Chung SJ. Drug-Induced Parkinsonism. J Clin Neurol. 2012;8(1):15. doi:10.3988/jcn.2012.8.1.15 2. Kwok JYY, Kwan JCY, Auyeung M, et al. Effects of Mindfulness Yoga vs Stretching and Resistance Training Exercises on Anxiety and Depression for People With Parkinson Disease: A Randomized Clinical Trial. JAMA Neurol. 2019;76(7):755-763. doi:10.1001/jamaneurol.2019.0534 3. Stocchi F, Rascol O, Kieburtz K, et al. Initiating levodopa/carbidopa therapy with and without entacapone in early Parkinson disease: The STRIDE-PD study. Ann Neurol. 2010;68(1):18- 27. doi:10.1002/ana.22060 4. Fox SH, Katzenschlager R, Lim SY, et al. International Parkinson and movement disorder society evidence-based medicine review: Update on treatments for the motor symptoms of Parkinson’s disease: Treatment of Motor Symptoms in PD. Mov Disord. 2018;33(8):1248- 1266. doi:10.1002/mds.27372 5. Zafar S, Yaddanapudi SS. Parkinson Disease. In: StatPearls. StatPearls Publishing; 2022. Accessed August 23, 2022. http://www.ncbi.nlm.nih.gov/books/NBK470193/ UpToDate Links: https://www.uptodate.com/contents/management-of-nonmotor-symptoms-in-parkinson- disease?search=parkinsons%20disease%20non%20motor%20symptoms&source=search_resu lt&selectedTitle=1~150&usage_type=default&display_rank=1 https://www.uptodate.com/contents/initial-pharmacologic-treatment-of-parkinson- disease?search=parkinsons%20disease%20adult&source=search_result&selectedTitle=2~150 &usage_type=default&display_rank=2 https://www.uptodate.com/contents/medical-management-of-motor-fluctuations-and-dyskinesia- in-parkinson- disease?search=motor%20symptom%20fluctuations%20parkinsons&source=search_result&s electedTitle=2~150&usage_type=default&display_rank=2