19 152-3-21-2011 colligative properties
•
1 like•638 views
3-21-2011 colligative properties
Report
Share
Report
Share
Download to read offline
Recommended
Recommended
More Related Content
What's hot
What's hot (20)
colligative property ,made by-chinmay jagadev pattanayak

colligative property ,made by-chinmay jagadev pattanayak
Diploma_I_Applied science(chemistry)_U-II(A) Preparation of solution 

Diploma_I_Applied science(chemistry)_U-II(A) Preparation of solution
Viewers also liked
Minunata si utila prezentareSmscluj2010cumiticonstruiestiunbrandpersonalinsocialmediaadihadean 1012210456...

Smscluj2010cumiticonstruiestiunbrandpersonalinsocialmediaadihadean 1012210456...SIMONA GABRIELA LUCA
2016Cu suflet echilibrat proiect colegiul tehnic dimitrie leonida zona libera de ...

Cu suflet echilibrat proiect colegiul tehnic dimitrie leonida zona libera de ...SIMONA GABRIELA LUCA
Viewers also liked (19)
Personal branding 20 may 2016 proiect ict_conferinta_final 2

Personal branding 20 may 2016 proiect ict_conferinta_final 2
Proiect vetpro ccd bucuresti romania@angus college scotia, uk_ 142_2012_en

Proiect vetpro ccd bucuresti romania@angus college scotia, uk_ 142_2012_en
Smscluj2010cumiticonstruiestiunbrandpersonalinsocialmediaadihadean 1012210456...

Smscluj2010cumiticonstruiestiunbrandpersonalinsocialmediaadihadean 1012210456...
Prezentare proiect comenius regio ccd buc anpcdefp simona luca

Prezentare proiect comenius regio ccd buc anpcdefp simona luca
Cu suflet echilibrat proiect colegiul tehnic dimitrie leonida zona libera de ...

Cu suflet echilibrat proiect colegiul tehnic dimitrie leonida zona libera de ...
Presentation initial survey teachers simona luca ccd bucuresti

Presentation initial survey teachers simona luca ccd bucuresti
Presentation initial survey teachers simona luca ccd bucuresti

Presentation initial survey teachers simona luca ccd bucuresti
Managerial skills for projects erasmus+ 2014 simona luca

Managerial skills for projects erasmus+ 2014 simona luca
Skills for projects Simona Luca 2014 Romana Romania

Skills for projects Simona Luca 2014 Romana Romania
Managerial skills for projects erasmus+ 2014 simona luca

Managerial skills for projects erasmus+ 2014 simona luca
Proiect vetpro ccd bucuresti romania@angus college scotia, uk_ 142_2012_en

Proiect vetpro ccd bucuresti romania@angus college scotia, uk_ 142_2012_en
Similar to 19 152-3-21-2011 colligative properties
Similar to 19 152-3-21-2011 colligative properties (20)
Chapter 12 6-8 Colligative Properties presntation.pptx

Chapter 12 6-8 Colligative Properties presntation.pptx
More from Joanne Cox
More from Joanne Cox (20)
Recently uploaded
Recently uploaded (20)
💞SEXY💞 UDAIPUR ESCORTS 09602870969 CaLL GiRLS in UdAiPuR EsCoRt SeRvIcE💞

💞SEXY💞 UDAIPUR ESCORTS 09602870969 CaLL GiRLS in UdAiPuR EsCoRt SeRvIcE💞
Call Girls Service In Udaipur 9602870969 Sajjangarh Udaipur EsCoRtS

Call Girls Service In Udaipur 9602870969 Sajjangarh Udaipur EsCoRtS
Pooja : 9892124323, Dharavi Call Girls. 7000 Cash Payment Free Home Delivery

Pooja : 9892124323, Dharavi Call Girls. 7000 Cash Payment Free Home Delivery
Nalasopara Call Girls Services 9892124323 Home and Hotel Delivery Free

Nalasopara Call Girls Services 9892124323 Home and Hotel Delivery Free
💞5✨ Hotel Karnal Call Girls 08168329307 Noor Mahal Karnal Escort Service

💞5✨ Hotel Karnal Call Girls 08168329307 Noor Mahal Karnal Escort Service
Bangalore Escorts 📞9955608600📞 Just📲 Call Rajveer Call Girls Service In Benga...

Bangalore Escorts 📞9955608600📞 Just📲 Call Rajveer Call Girls Service In Benga...
Call Girls Udaipur Just Call 9602870969 Top Class Call Girl Service Available

Call Girls Udaipur Just Call 9602870969 Top Class Call Girl Service Available
New Call Girls In Panipat 08168329307 Shamli Israna Escorts Service

New Call Girls In Panipat 08168329307 Shamli Israna Escorts Service
Rudraprayag call girls 📞 8617697112 At Low Cost Cash Payment Booking

Rudraprayag call girls 📞 8617697112 At Low Cost Cash Payment Booking
❤️Call Girls In Chandigarh 08168329307 Dera Bassi Zirakpur Panchkula Escort S...

❤️Call Girls In Chandigarh 08168329307 Dera Bassi Zirakpur Panchkula Escort S...
Zirakpur Call Girls👧 Book Now📱8146719683 📞👉Mohali Call Girl Service No Advanc...

Zirakpur Call Girls👧 Book Now📱8146719683 📞👉Mohali Call Girl Service No Advanc...
👉Amritsar Call Girl 👉📞 8725944379 👉📞 Just📲 Call Mack Call Girls Service In Am...

👉Amritsar Call Girl 👉📞 8725944379 👉📞 Just📲 Call Mack Call Girls Service In Am...
All Hotel Karnal Call Girls 08168329307 Noor Mahal Karnal Escort Service

All Hotel Karnal Call Girls 08168329307 Noor Mahal Karnal Escort Service
Call Girls Zirakpur👧 Book Now📱7837612180 📞👉Call Girl Service In Zirakpur No A...

Call Girls Zirakpur👧 Book Now📱7837612180 📞👉Call Girl Service In Zirakpur No A...
Call Girls in Bangalore Nisha 💋9136956627 Bangalore Call Girls

Call Girls in Bangalore Nisha 💋9136956627 Bangalore Call Girls
👉Chandigarh Call Girls 📞Book Now📞👉 9878799926 👉Zirakpur Call Girl Service No ...

👉Chandigarh Call Girls 📞Book Now📞👉 9878799926 👉Zirakpur Call Girl Service No ...
👉Chandigarh Call Girls 📞Book Now📞👉 9878799926 👉Zirakpur Call Girl Service No ...

👉Chandigarh Call Girls 📞Book Now📞👉 9878799926 👉Zirakpur Call Girl Service No ...
Call Girl In Zirakpur👧 Book Now📱7837612180 📞👉Zirakpur Call Girls Service No A...

Call Girl In Zirakpur👧 Book Now📱7837612180 📞👉Zirakpur Call Girls Service No A...
9892124323 Pooja Nehwal - Book Local Housewife call girls in Nalasopara at Ch...

9892124323 Pooja Nehwal - Book Local Housewife call girls in Nalasopara at Ch...
19 152-3-21-2011 colligative properties
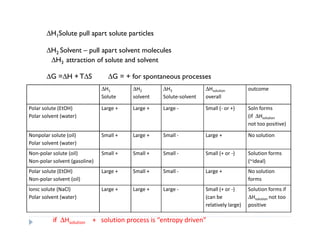
- 1. ∆H1Solute pull apart solute particles ∆H2 Solvent – pull apart solvent molecules ∆H3 attraction of solute and solvent ∆G =∆H + T∆S ∆G = + for spontaneous processes ∆H1 ∆H2 ∆H3 ∆Hsolution outcome Solute solvent Solute-solvent overall Polar solute (EtOH) Large + Large + Large - Small (- or +) Soln forms Polar solvent (water) (if ∆Hsolution not too positive) Nonpolar solute (oil) Small + Large + Small - Large + No solution Polar solvent (water) Non-polar solute (oil) Small + Small + Small - Small (+ or -) Solution forms Non-polar solvent (gasoline) (~ideal) Polar solute (EtOH) Large + Small + Small - Large + No solution Non-polar solvent (oil) forms Ionic solute (NaCl) Large + Large + Large - Small (+ or -) Solution forms if Polar solvent (water) (can be ∆Hsolution not too relatively large) positive if ∆Hsolution + solution process is “entropy driven”
- 2. Like dissolves like # 21 Which likely to be soluble in benzene CCl4 or NaCl Hexane or glycerol Acetic acid or hexanoic acid HCl or propyl chloride
- 3. Temp effect – gases Gas + liquid solution + energy Temp increase increases energy available Favors “dis” solution Thermal pollution – effect on fish
- 4. # 25 and 26 PbCl2 NaOH ∆H for dissociation to aqueous ions Using data from Ch 8 or appendix Will solubility increase with increasing temp?
- 5. Pressure effect on solubility Henry’s law Increased pressure increase gas solubility C g = k Pg Pg = partial pressure of the gas above liquid k – constant – characteristic of particular gas/liquid system Carbonated beverages The bends – Difference in solubility of gases (nitrogen or helium as carrier gas) with increasing P Decompressing after diving Hyperbaric chambers for CO poisoning, wound healing etc (direct solubility) Practice # 29
- 6. Typical Henry’s law problem He in water 3.8 X 10-4 M/atm @ 25 oC (k) ? M/mm Hg pHe =293 mm Hg @ 25 oC, what is [ ] in M C g = k Pg HW: 27, 29 Ch 10
- 7. Colligative properties Due to number of particles in solution Be familiar with Electrolytes (soluble salts, SA, SB) Completely dissociate into ions Non-electrolytes Molecular compounds (no ions) Polar molecular compounds are water soluble Weak electrolytes WA, WB – partially dissociate Note worksheet
- 8. Colligative properties Due to number of particles in solution 1 mole sugar per liter = ? mol/L particles? 1 mole NaCl per liter 1 mole aluminum sulfate per liter 1 mole acetic acid Electrolytes/nonelectrolytes
- 9. Vapor pressure Pressure of the vapor above a liquid or solution – depends on temp and identity Raoult’s law - mix with volatile solute VPsoln = XsoluteVP solute + XsolventVPsolvent evaporation
- 10. Ideal solution of 2 volatile solvents (liquids) 1. What is the VP of pure hexane ___ 2. What is the VP of pure pentane ___ 3. Which is more volatile? 4. If X = 0.5, what is VP due to pentane___ hexane ___total VP _____ Raoult’s Law VPT = XAVPA + XBVPB Related exercise: 1 mol benzene (C6H6), Po = 75 torr 2 mol toluene (C7H8) Po = 22 torr What is the mf of benzene in the mixture? What is the vp of the mixture? What is the mf of benzene in the vapor phase? Data is for a given temp since pentane is a liquid at RT, this must be for a higher T
- 11. VP lowering Non-volatile solutes decrease vp Independent of nature of solute Dependent on number of particles in solution Raoult’s law -- except solute does not exert vp (non- volatile) VPsolution = Xsolvent VPsolvent VPlowering = Xsolute Vpsolvent VPsolution + Vplowering = VPsolvent
- 12. VPsolution = Xsolvent VPsolvent VPlowering = Xsolute VPsolvent VPsolution + VPlowering = VPsolvent Normally used with non-electrolytes # 35 Note: mole fraction concentration unit used
- 13. Boiling point elevation If solutes depress vp, to boil (vp = 760) temp must be elevated Depends on concentration of particles in solution and on solvent Use molality mol solute particles/kg solvent BP elevation = k i m k= bp constant for solvent i= activity of solute (# particles/mol) m = molality of solute (mol solute/kg solvent) 31,32 (bp’s) Lab Wed benzoic acid in lauric acid
- 14. Freezing point depression Solutes interfere with crystal formation Salt added to ice – interferes with crystal structure, ice structures breaks down – endothermic process!!!! Ocean water – ice bergs are fresh water --salt is not included 1.86oC/mole of particles in 1 kg water Melting ice 31, 32 General Chemistry Online: FAQ: Solutions: Why does salt melt ice?
- 15. Review spec lab end of class Assignments due Make sugar solns Spec lab: Calc k Calc energies Calc unk conc Emitted and abs colors Resubmit HW Ch 10 28-30 (Henry’s) 39 40 42-44 (fp, bp)