UTI AND HEMATURIA
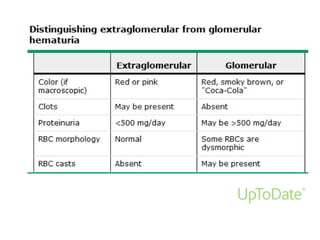
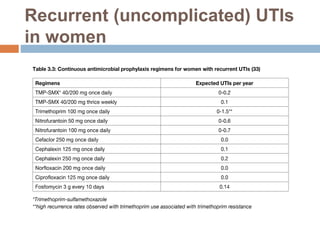
This document discusses urinary tract infections (UTIs) and hematuria. It covers topics such as the different levels of UTIs, grading severity, risk factors, definitions of uncomplicated and complicated UTIs, etiology, diagnosis and treatment of acute cystitis and pyelonephritis. It also discusses recurrent UTIs, UTIs during pregnancy, in postmenopausal women, and complicated cases involving diabetes, HIV, renal transplants and chronic kidney disease. The document provides detailed guidelines on diagnosis, treatment, antibiotic selection and follow up for the various types of UTIs and patient populations.






























































![ Viral pathogens, particularly the herpes group viruses
but also hepatitis B (HBV) and hepatitis C (HCV).
New viruses are recognized as opportunistic
pathogens with the use of more sensitive molecular
assays (eg, BK polyomavirus, human herpesvirus
[HHV]-6, -7, and -8
Tuberculosis and, increasingly, nontuberculous
mycobacteria
Gastrointestinal parasites (Cryptosporidium and
Microsporidium) and viruses (cytomegalovirus [CMV],
rotavirus) may be associated with diarrhea.
TIMING OF INFECTION
POSTTRANSPLANTATION](https://image.slidesharecdn.com/uti-160204125633/85/Uti-65-320.jpg)