STUDY OF FERRITIN SELF-ASSEMBLY AND HETEROPOLYMER FORMATION USING FRET
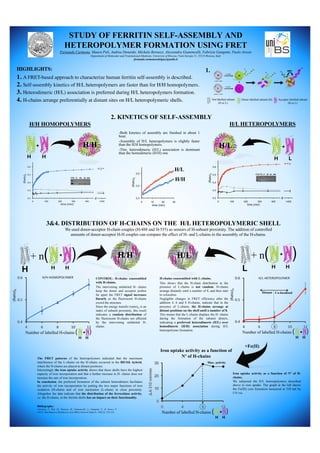
- 1. STUDY OF FERRITIN SELF-ASSEMBLY AND HETEROPOLYMER FORMATION USING FRET Fernando Carmona, Maura Poli, Andrea Denardo, Michela Bertuzzi, Alessandra Gianoncelli, Fabrizio Gangemi, Paolo Arosio Department of Molecular and Translational Medicine, University of Brescia, Viale Europa 11, 25123 Brescia, Italy fernando.carmonarodriguez@unibs.it HIGHLIGHTS: 1. A FRET-based approach to characterize human ferritin self-assembly is described. 2. Self-assembly kinetics of H/L heteropolymers are faster than for H/H homopolymers. 3. Heterodimeric (H/L) association is preferred during H/L heteropolymers formation. 4. H-chains arrange preferentially at distant sites on H/L heteropolymeric shells. + H H H/H + H L H/L : Not labelled subunit (H or L) : Donor labelled-subunit (H) : Acceptor labelled-subunit (H or L) H/H HOMOPOLYMERS H/L HETEROPOLYMERS 2. KINETICS OF SELF-ASSEMBLY 1. 3&4. DISTRIBUTION OF H-CHAINS ON THE H/L HETEROPOLYMERIC SHELL H/L L + H H + n( )H/H H + H H + n( ) + H H Number of labelled H-chains ( ) -Both kinetics of assembly are finished in about 1 hour. -Assembly of H/L heteropolymers is slightly faster than the H/H homopolymers. -This, heterodimeric (H/L) association is dominant than the homodimeric (H/H) one. We used donor-acceptor H-chain couples (H-488 and H-555) as sensors of H-subunit proximity. The addition of controlled amounts of donor-acceptor H-H couples can compare the effect of H- and L-chains in the assembly of the H-chains. CONTROL: H-chains coassembled with H-chains. The intervening unlabeled H- chains keep the donor and acceptor probes far apart the FRET signal increases linearly as the fluorescent H-chains crowd the structure. Since the energy transfer (ratio)A is an index of subunit proximity, this result indicates a random distribution of the fluorescent H-chains not affected by the intervening unlabeled H- chains. H-chains coassembled with L-chains. This shows that the H-chain distribution in the presence of L-chains is not random. H-chains arrange distantly until a number of 8, and then start to colocalize. Negligible changes in FRET efficiency after the addition 4, 6 and 8 H-chains, indicate that in the presence of L-chains, the H-chains arrange at distant positions on the shell until a number of 8. This means that the L-chains displace the H- chains during the formation of the subunit dimers, indicating a preferred heterodimeric (H/L) over homodimeric (H/H) association during H/L heteropolymer formation. +Fe(II) + H H Number of labelled H-chains ( ) Iron uptake activity as a function of Nº of H-chains + H H Number of labelled H-chains ( ) Distant Co-localized Max. activity The FRET patterns of the heteropolymers indicated that the maximum interference of the L-chains on the H-chains occurred in the 8H/16L hybrid, where the H-chains are placed at distant positions. Interestingly the iron uptake activity shows that these shells have the highest capacity of iron incorporation and that a further increase in H- chains does not increase the rate of iron incorporation. In conclusion, the preferred formation of the subunit heterodimers facilitates the activity of iron incorporation by putting the two major functions of iron oxidation (H-chain) and of iron nucleation (L-chain) in close proximity. Altogether the data indicate that the distribution of the ferroxidase activity, i.e. the H-chains, in the ferritin shells has an impact on their functionality. Iron uptake activity as a function of Nº of H- chains. We subjected the H/L heteropolymers described above to iron uptake. The graph at the left shows the Fe(III) core formation measured at 310 nm by UV/vis. Bibliography: Carmona, F., Poli, M., Bertuzzi, M., Gianoncelli, A., Gangemi, F., & Arosio, P. (2017), Biochimica et Biophysica Acta (BBA)-General Subjects, 1861(3), 522-532.