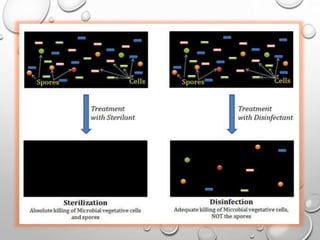
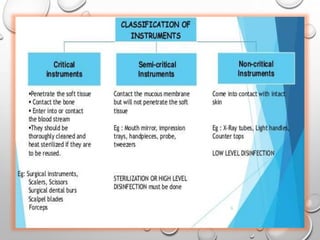
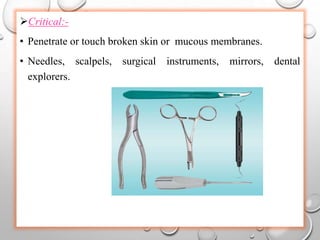
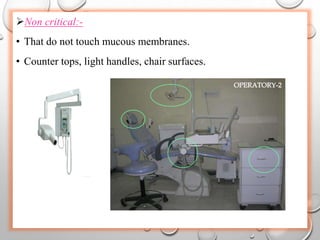
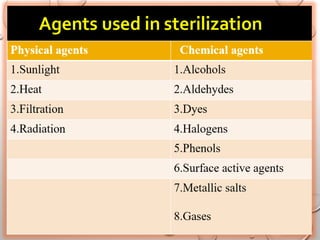
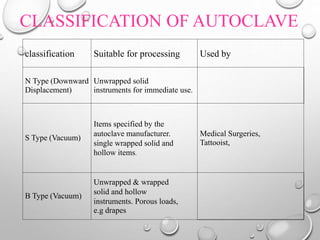
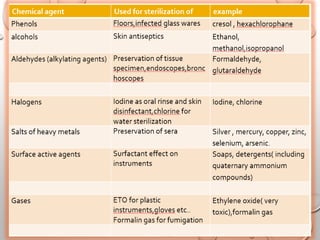
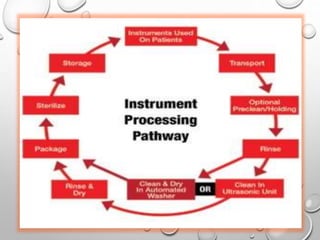
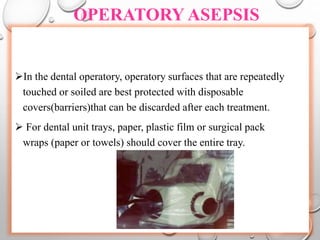
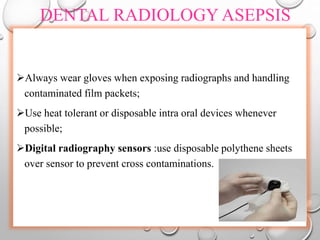
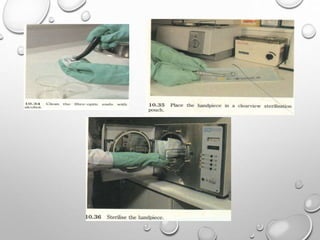
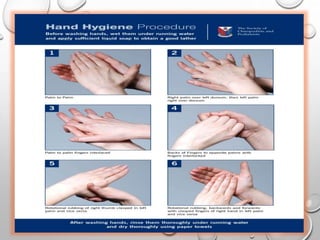
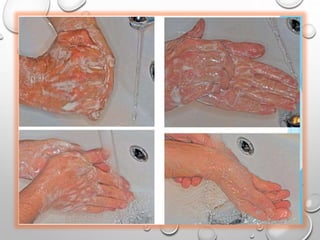
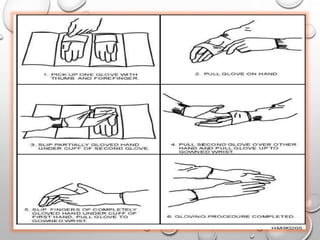
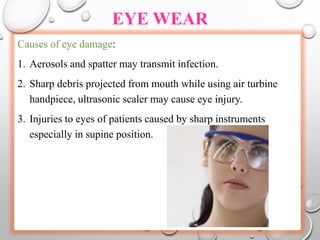
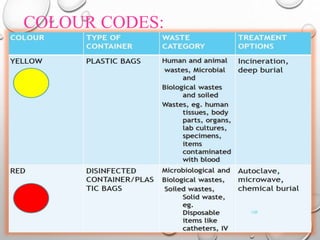
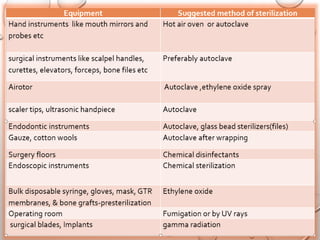
The document provides an extensive overview of sterilization and disinfection, detailing historical developments, definitions, and various methods utilized in infection control and sterilization processes. It discusses factors influencing sterilization efficacy, procedures for instrument cleaning, and the operation of different sterilization devices such as autoclaves, ultrasonic cleaners, and chemical vapors. Additionally, it describes the implications of various sterilization techniques, including advantages, disadvantages, and specific uses for different medical instruments.