This study analyzed data from over 30,000 patients with heart failure (HF) to identify predictors of adverse outcomes based on left ventricular ejection fraction (LVEF). Patients were categorized as having preserved LVEF (≥50%), borderline LVEF (41-49%), or reduced LVEF (≤40%). Over a median follow up of 1.8 years, 26.8% of patients died, 26.9% were hospitalized for HF, and 67.4% were hospitalized for any reason. Multivariable models found that nearly all tested predictors, such as demographics, medical history, and comorbidities, performed similarly for predicting death and hospitalization across the different LVEF



![Allen et al Prognostic Factors by LVEF Group 637
preserved nor frankly reduced LVEF (ie, borderline or mildly
reduced LVEF [HF-BREF]). As such, it is unclear under
what circumstances and to what extent we can and should be
grouping HF patients together with varying level of LVEF for
risk assessment, versus considering risk uniquely in different
HF patient populations.
Understanding the relative importance of common predic-
tor variables for important clinical outcomes among different
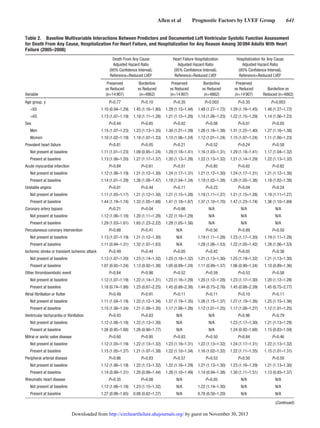
Table 1. Continued
Characteristics
Overall Cohort
(N=30 094)
Preserved LVEF
(n=14 907)
Borderline LVEF
(n=4862)
Reduced LVEF
(n=10 325)
D Values Reduced
vs Preserved
D Values Reduced
vs Borderline
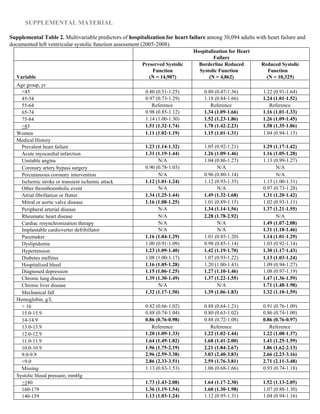
Missing 1861 (6.2) 622 (4.2) 289 (5.9) 950 (9.2)
Systolic blood pressure, mm Hg; mean (SD) 131.1 (19.4) 133.4 (19.9) 131.2 (19.1) 127.7 (18.2)
Systolic blood pressure category, mm Hg; n (%) 0.28 0.19
≥180 689 (2.3) 433 (2.9) 109 (2.2) 147 (1.4)
160–179 1832 (6.1) 1124 (7.5) 291 (6.0) 417 (4.0)
140–159 5217 (17.3) 2906 (19.5) 866 (17.8) 1445 (14.0)
130–139 5646 (18.8) 2943 (19.7) 914 (18.8) 1789 (17.3)
121–129 4496 (14.9) 2238 (15.0) 729 (15.0) 1529 (14.8)
110–120 8275 (27.5) 3564 (23.9) 1363 (28.0) 3348 (32.4)
100–109 1571 (5.2) 679 (4.6) 235 (4.8) 657 (6.4)
<100 904 (3.0) 345 (2.3) 142 (2.9) 417 (4.0)
Missing 1464 (4.9) 675 (4.5) 213 (4.4) 576 (5.6)
HDL cholesterol, g/dL; mean (SD) 47.4 (14.7)
HDL cholesterol category, g/dL; n (%) 0.16 0.05
≥60 4736 (15.7) 2669 (17.9) 691 (14.2) 1376 (13.3)
50–50.9 5153 (17.1) 2676 (18.0) 789 (16.2) 1688 (16.4)
40–49.9 8188 (27.2) 4056 (27.2) 1348 (27.7) 2784 (27.0)
35–39.9 4134 (13.7) 1969 (13.2) 702 (14.4) 1463 (14.2)
<35 4728 (15.7) 2105 (14.1) 873 (18.0) 1750 (17.0)
Missing 3155 (10.5) 1432 (9.6) 459 (9.4) 1264 (12.2)
LDL cholesterol, g/dL; mean (SD) 96.5 (34.2)
LDL cholesterol category, g/dL; n (%) 0.02 0.03
≥200 262 (0.9) 108 (0.7) 50 (1.0) 104 (1.0)
160–199.9 1011 (3.4) 477 (3.2) 165 (3.4) 369 (3.6)
130–159.9 2820 (9.4) 1357 (9.1) 462 (9.5) 1001 (9.7)
100–129.9 6442 (21.4) 3304 (22.2) 1011 (20.8) 2127 (20.6)
70–99.9 10 617 (35.3) 5355 (35.9) 1725 (35.5) 3537 (34.3)
<70 5539 (18.4) 2763 (18.5) 956 (19.7) 1820 (17.6)
Missing 3403 (11.3) 1543 (10.4) 493 (10.1) 1367 (13.2)
Total cholesterol, g/dL; mean (SD) 173.4 (44.2) 173.8 (43.2) 172.5 (45.8) 173.2 (44.9)
Total cholesterol category, g/dL; n (%) 0.04 0.05
>240 1817 (6.0) 876 (5.9) 313 (6.4) 628 (6.1)
200–240 4667 (15.5) 2367 (15.9) 730 (15.0) 1570 (15.2)
<200 20 764 (69.0) 10 378 (69.6) 3418 (70.3) 6968 (67.5)
Missing 2846 (9.5) 1286 (8.6) 401 (8.2) 1159 (11.2)
Serum sodium, mmol/L; mean (SD) 139.7 (3.5) 139.6 (3.6) 139.8 (3.7) 139.7 (3.3)
Serum sodium category, mmol/L; n (%) 0.15 0.07
≥150 16 (0.1) 9 (0.1) 3 (0.1) 4 (0.0)
140–149 15503 (51.5) 7854 (52.7) 2506 (51.5) 5143 (49.8)
130–139 11299 (37.5) 5751 (38.6) 1796 (36.9) 3752 (36.3)
<130 310 (1.0) 195 (1.3) 41 (0.8) 74 (0.7)
Missing 2966 (9.9) 1098 (7.4) 516 (10.6) 1352 (13.1)
HDL indicates high-density lipoprotein; LDL, low-density lipoprotein; and LVEF, left ventricular ejection fraction.
by guest on November 30, 2013http://circheartfailure.ahajournals.org/Downloaded from](https://image.slidesharecdn.com/risk-factors-for-adverse-coutcomes-160210064536/85/Risk-factors-for-adverse-coutcomes-4-320.jpg)



![644 Circ Heart Fail July 2013
75.3%; P<0.001), trended toward more often women (47.3%
versus 46.0%; P=0.06), had more prevalent HF (73.0% versus
60.1%; P<0.001), had less myocardial infarction (12.3 versus
13.9%), and generally had more comorbidity, including cere-
brovascular disease (23.0% versus 21.1%; P<0.001), diabetes
mellitus (26.2% versus 24.0%; P<0.001), dementia (10.0%
versus 7.3%; P<0.001), and chronic lung disease (43.3%
versus 41.1%); during follow-up there was no significant dif-
ference between death (27.8% for those without a measure
of LVEF; P=0.08) but had lower rates of HF hospitaliza-
tion (22.6%; P<0.001) and all-cause hospitalization (63.4%;
P<0.001).
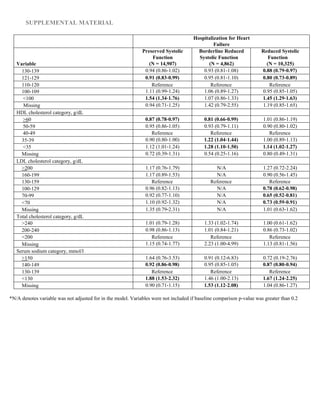
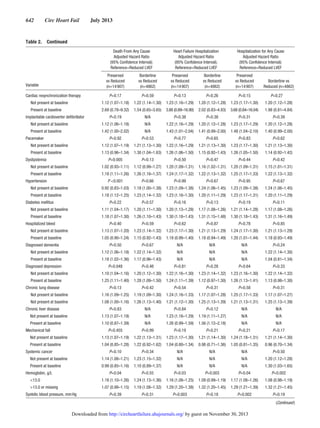
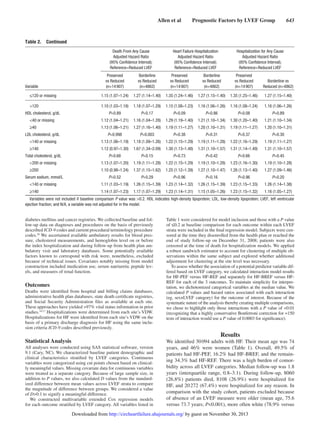
In multivariable Cox models for all-cause death (Figure
[A]), advanced age and severe anemia showed the strongest
association with the outcome across all strata of LVEF (rec-
ognizing that models did not include a measure of renal func-
tion). Only systolic blood pressures <100 mm Hg showed
significant association with increased mortality. Past medical
history factors were either neutral or weakly associated with
death, except for prior coronary revascularization, which was
associated with survival.
In multivariable models for hospitalization from HF (Figure
[B]), advanced age and anemia continued to be strong predic-
tors of the outcome across all strata of LVEF. Only patients
aged >85 years seemed to have a significantly increased risk
of HF hospitalization. Hypertension, more than hypotension,
was predictive of HF hospitalization. A variety of medical his-
tory carried small increased risk of HF hospitalization; nota-
bly prior coronary revascularization was not associated with
hospitalization from HF.
Multivariable models for all-cause hospitalization (Figure
[C]) were quite similar to those for HF hospitalization, despite
the majority of all-cause admissions having a non-HF primary
discharge diagnosis code. Anemia continued to show the
largest adjusted hazards ratios for the outcome. Progressive
hypertension was increasingly predictive of all-cause hospi-
talization; systolic blood pressure <100 mm Hg was also asso-
ciated with an increase in all-cause hospitalization, creating
a U-shaped association for systolic blood pressure overall.
Advanced age was not predictive of all-cause hospitalization,
except for mild associations in the HF-BREF group and at age
≥85 years in the HF-REF group.
We found that multivariable models for each of the 3 out-
comes were highly consistent across HF-PEF, HF-BREF, and
HF-REF patients (Figure 1; complete data included as tables
in the online-only Data Supplement). In simplified interaction
models (Table 2), very few of the risk factors had a signifi-
cant interaction with LVEF for any of the 3 outcomes (using
P value <0.01). For the outcome of all-cause mortality, only a
baseline history of dyslipidemia and hypertension performed
differently by HF-PEF versus HF-REF and only low-density
lipoprotein differed by HF-BREF versus HF-REF. For the
outcome of heart failure hospitalization, only systolic blood
pressure differed by HF-PEF versus HF-REF and only age
differed by HF-BREF versus HF-REF. For the outcome of all-
cause hospitalization, only systolic blood pressure differed by
HF-PEF versus HF-REF and only age and hemoglobin dif-
fered by HF-BREF versus HF-REF.
Discussion
Within a large, contemporary, multicenter cohort of patients
with HF, we found that commonly available risk factors car-
ried surprisingly similar prognostic information for a variety
of outcomes across all LVEF categories. Despite the existence
of a variety of published HF risk models, this systematic
assessment of relative risk factor performance across 3 LVEF
strata for death, HF-related hospitalization, and all-cause hos-
pitalization within a diverse, representative HF population
provides novel HF risk information. For example, the popu-
larized Seattle Heart Failure Model4
was derived and validated
in randomized trial populations of patients with HF-REF. Its
performance has subsequently been tested in a variety of other
cohorts, including patients with a range of LVEF,21,22
but this
piece-meal approach makes comparisons of individual risk
factor performance across different LVEF categories more dif-
ficult. Others have begun to look at the comparative prognostic
performance of single risk factors by LVEF categories, such
as a recent analysis showing that for a given serum B-type
natriuretic peptide level the prognosis is essentially the same
for patients with HF-PEF as those with HF-REF.23
Here, the
analytic approach was specifically designed to provide infor-
mation on comparative risk factor performance across a wide
range of covariates by LVEF categories. We essentially found
that none of the risk factors consistently interacted with LVEF.
Although LVEF dictates responsiveness to certain HF ther-
apies,24
our data demonstrate that common risk factors have
quite similar prognostic performance across major strata of
LVEF. The number of statistically significant differences in
risk factor performance across LVEF strata in our models
was not much different than would have been predicted by
chance alone. We conclude that a parsimonious approach to
HF risk modeling is appropriate in most circumstances, at
least for discrimination among singular end points. This may
have important practical implications for HF risk stratifica-
tion efforts, particularly because LVEF has been difficult to
automatically extract from most electronic medical records
without manual chart review.
Our findings extend those of previous HF risk studies,
particularly into the population of patients with HF-PEF and
HF-BREF. Our results are consistent with several smaller
studies of hospitalized patients with HF-PEF, which have
observed higher risks of hospitalization in patients with diabe-
tes mellitus, depressive symptoms, and anemia.25–27
Potential Limitations
Because of the large sample size, some associations within
individual risk models may be statistically significant but not
clinically meaningful. More important, even with the relative
power of this sample size, we found strikingly few statistically
and even fewer clinically significant differences between risk
models, strengthening the primary conclusion that risk factor
and overall model performance was quite similar within LVEF
strata. Second, insured populations in our participating health
plans may not be fully representative of the general popula-
tion. Nevertheless, the breadth of geographic and demographic
diversity represented across 4 geographically diverse health
plans, as well as the community-based nature of healthcare
by guest on November 30, 2013http://circheartfailure.ahajournals.org/Downloaded from](https://image.slidesharecdn.com/risk-factors-for-adverse-coutcomes-160210064536/85/Risk-factors-for-adverse-coutcomes-11-320.jpg)