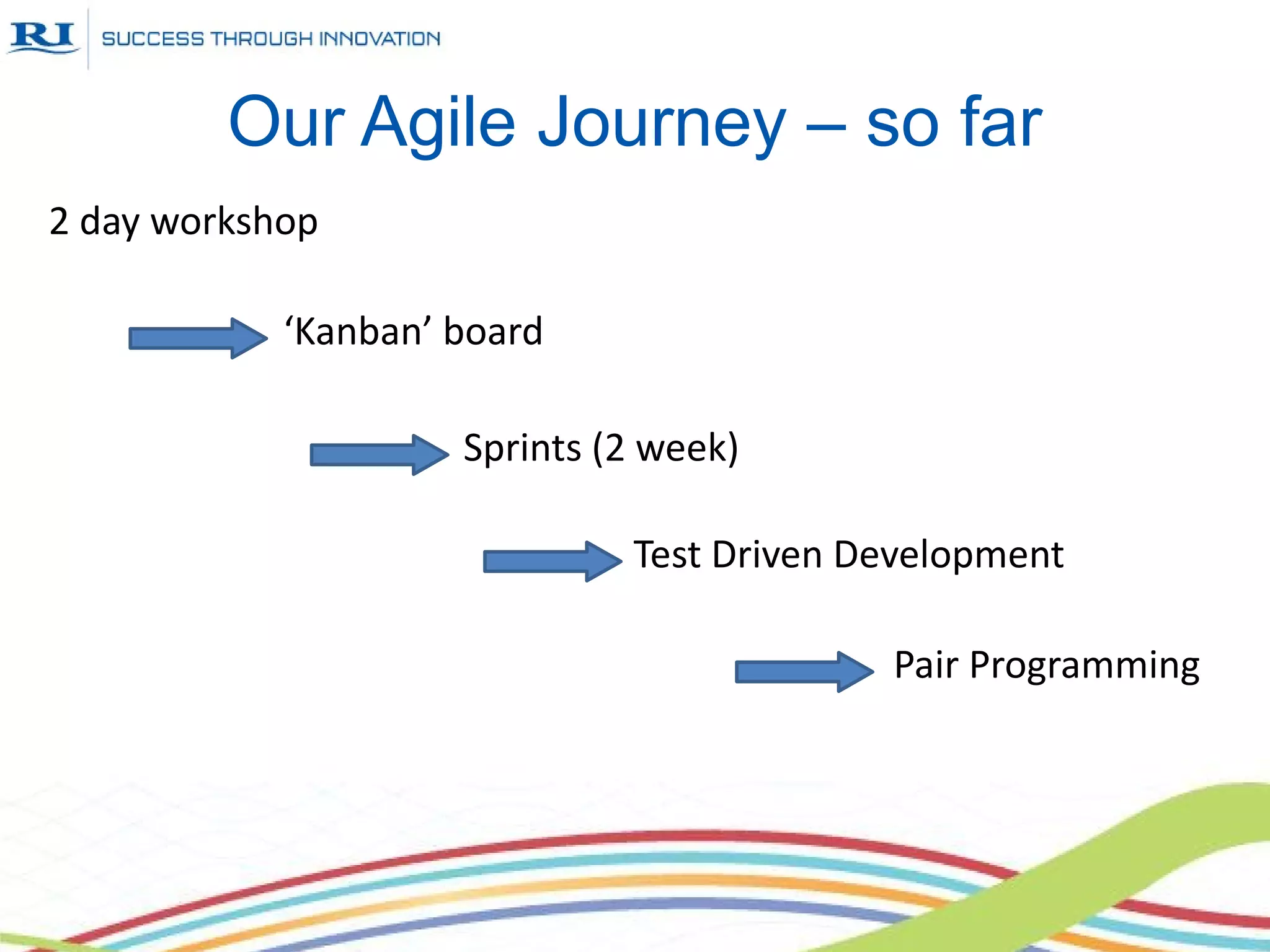
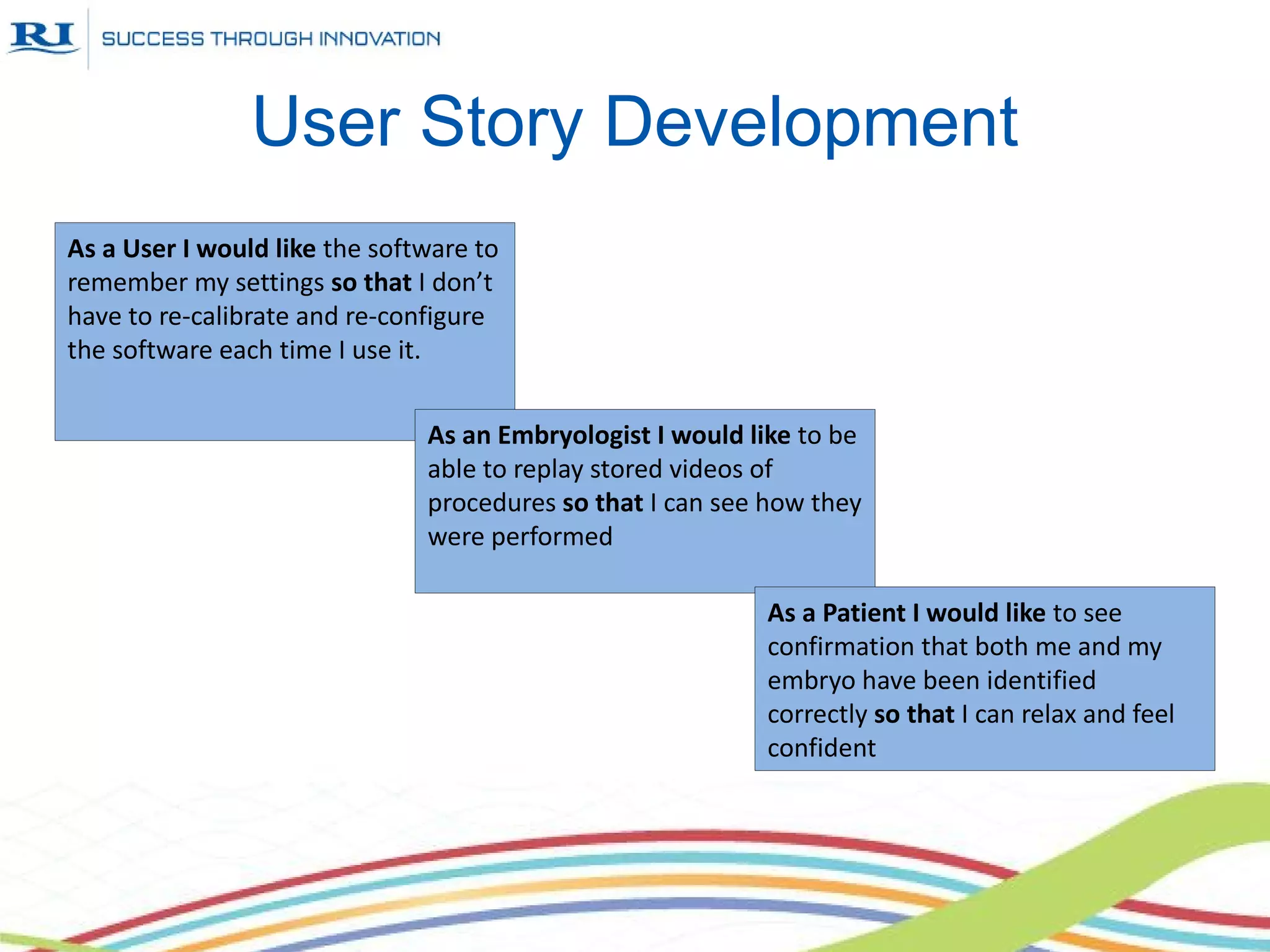
This document discusses Research Instruments' adoption of Agile practices for their software development process. It provides background on the company and describes their previous waterfall process. It then outlines the steps they took in adopting Agile, including workshops, Kanban boards, sprints, test-driven development, and pair programming. Benefits included improved team motivation, easier management, and higher quality. Adoption of Agile practices also led to changes in other departments and a better understanding with management and customers.