Pyrite oxidation leads to acidic conditions due toA. a slow chemi.pdf
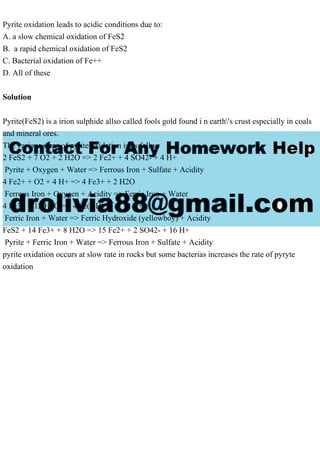
Pyrite oxidation leads to acidic conditions due to: A. a slow chemical oxidation of FeS2 B. a rapid chemical oxidation of FeS2 C. Bacterial oxidation of Fe++ D. All of these Solution Pyrite(FeS2) is a irion sulphide allso called fools gold found i n earth\'s crust especially in coals and mineral ores. The various steps of pyrite oxidation is as follws 2 FeS2 + 7 O2 + 2 H2O => 2 Fe2+ + 4 SO42- + 4 H+ Pyrite + Oxygen + Water => Ferrous Iron + Sulfate + Acidity 4 Fe2+ + O2 + 4 H+ => 4 Fe3+ + 2 H2O Ferrous Iron + Oxygen + Acidity => Ferric Iron + Water 4 Fe3+ + 12 H2O => 4 Fe(OH)3 ¯ + 12 H+ Ferric Iron + Water => Ferric Hydroxide (yellowboy) + Acidity FeS2 + 14 Fe3+ + 8 H2O => 15 Fe2+ + 2 SO42- + 16 H+ Pyrite + Ferric Iron + Water => Ferrous Iron + Sulfate + Acidity pyrite oxidation occurs at slow rate in rocks but some bacterias increases the rate of pyryte oxidation.
Recommended
Recommended
More Related Content
More from fazanmobiles
More from fazanmobiles (20)
Recently uploaded
Recently uploaded (20)
Pyrite oxidation leads to acidic conditions due toA. a slow chemi.pdf
- 1. Pyrite oxidation leads to acidic conditions due to: A. a slow chemical oxidation of FeS2 B. a rapid chemical oxidation of FeS2 C. Bacterial oxidation of Fe++ D. All of these Solution Pyrite(FeS2) is a irion sulphide allso called fools gold found i n earth's crust especially in coals and mineral ores. The various steps of pyrite oxidation is as follws 2 FeS2 + 7 O2 + 2 H2O => 2 Fe2+ + 4 SO42- + 4 H+ Pyrite + Oxygen + Water => Ferrous Iron + Sulfate + Acidity 4 Fe2+ + O2 + 4 H+ => 4 Fe3+ + 2 H2O Ferrous Iron + Oxygen + Acidity => Ferric Iron + Water 4 Fe3+ + 12 H2O => 4 Fe(OH)3 ¯ + 12 H+ Ferric Iron + Water => Ferric Hydroxide (yellowboy) + Acidity FeS2 + 14 Fe3+ + 8 H2O => 15 Fe2+ + 2 SO42- + 16 H+ Pyrite + Ferric Iron + Water => Ferrous Iron + Sulfate + Acidity pyrite oxidation occurs at slow rate in rocks but some bacterias increases the rate of pyryte oxidation