This document discusses post-grant review proceedings under the America Invents Act, including:
1) Post-grant review (PGR) allows challenges to patent claims on any validity grounds within 9 months of a patent grant, while inter partes review (IPR) allows challenges based on printed prior art.
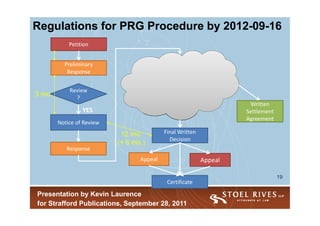
2) PGR regulations will be finalized by September 2016 and allow amendments, discovery, and oral hearings over 12-18 months. IPR has a lower threshold for initiation.
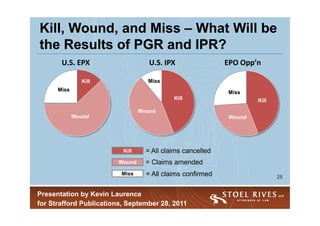
3) PGR may be less popular than IPR due to its shorter window, broader estoppel, and need to identify ripe patents for challenge like the EPO system. PGR outcomes will influence parallel litigation.