Platelets play a key role in hemostasis by forming temporary platelet plugs that seal small breaks in blood vessels. When platelets come into contact with damaged vessel walls, they become activated, change shape, secrete chemicals that attract other platelets, and aggregate to form a platelet plug. This platelet plug is later stabilized by fibrin threads formed during the blood coagulation process to form a blood clot. Platelets are produced from megakaryocytes in the bone marrow and normally circulate in the bloodstream, where they have a lifespan of about 10 days.

![About 25% to 40% of the platelets are stored in the spleen and released as
needed. The remainder circulate freely in the blood and live for about 10 days.
NB:-Platelets release serotonin, a chemical vasoconstrictor.
The endothelium is normally very smooth and coated with prostacyclin, a
platelet repellent.
How does a blood clot differ from a platelet plug?
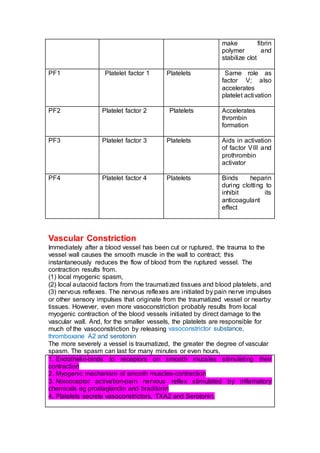
Clotting Factors (Procoagulants)
Number Name Origin Function
I Fibrinogen Liver Precursor of
fibrin
II Prothrombin Liver Precursor of
thrombin
III Tissue
thromboplastin
Perivascular
tissue
Activates factor
VII
V Proaccelerin:[Lieden
factor]
Liver Activates factor
VII; combines
with factor X to
form prothrombin
activator
VII Proconvertin Liver Activates factor X
in extrinsic
pathway
VIII Antihemophiliac
factor A
Liver Activates factor X
in intrinsic
pathway
IX Antihemophiliac
factor B
Liver Activates factor
VIII
X Thrombokinase Liver Combines with
factor V to form
prothrombin
activator
XI Antihemophiliac
factor C
Liver Activates factor
IX
XII Hageman factor Liver , platelets Activates factor
XI and plasmin;
converts
prekallikrein to
kallikrein
XIII Fibrin-stabilizing
factor
Platelets,
plasma
Cross-links fibrin
filaments to](https://image.slidesharecdn.com/plateletsandhemostasis-220514132853-b633896c/85/PLATELETS-AND-HEMOSTASIS-docx-2-320.jpg)