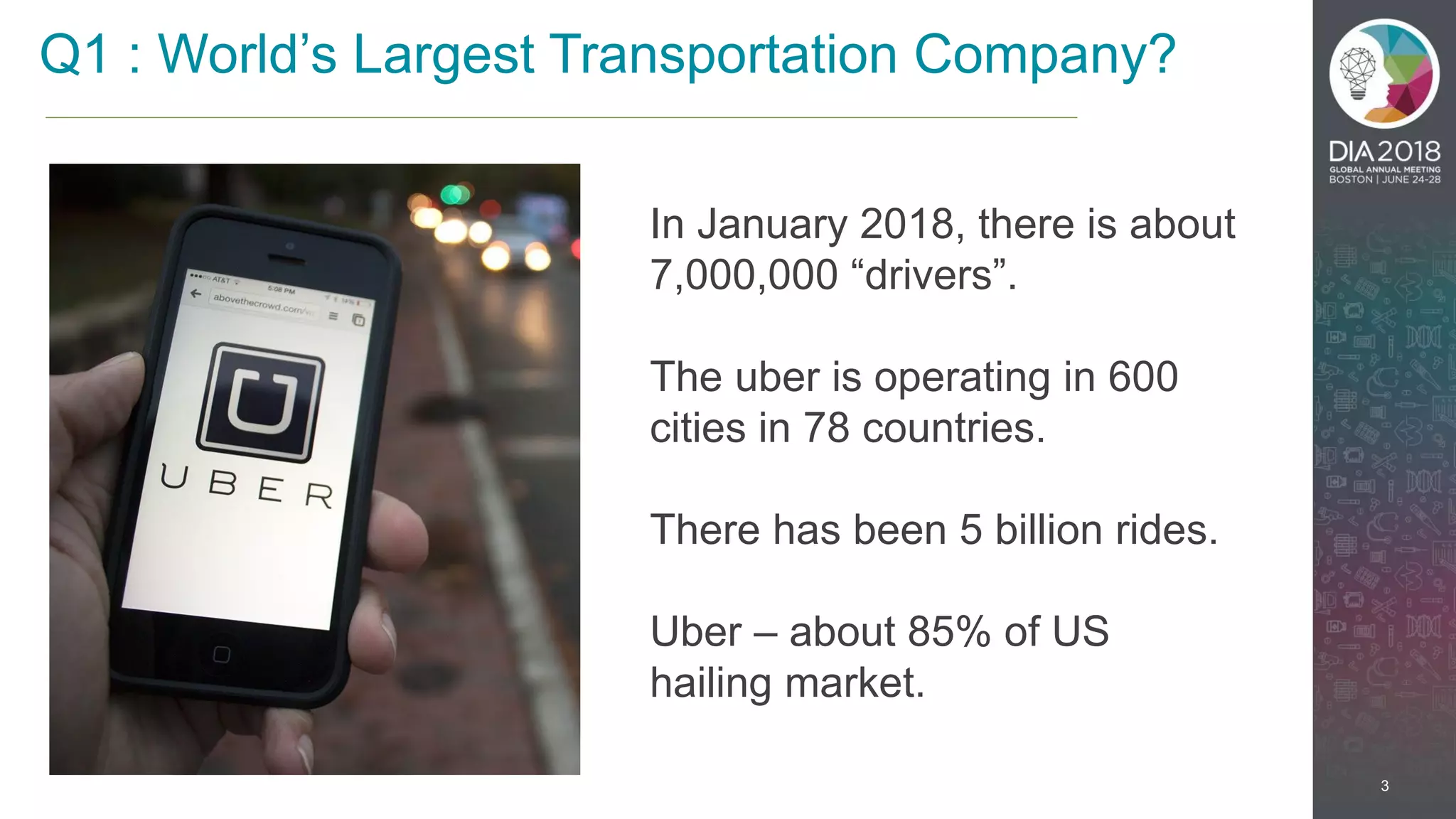
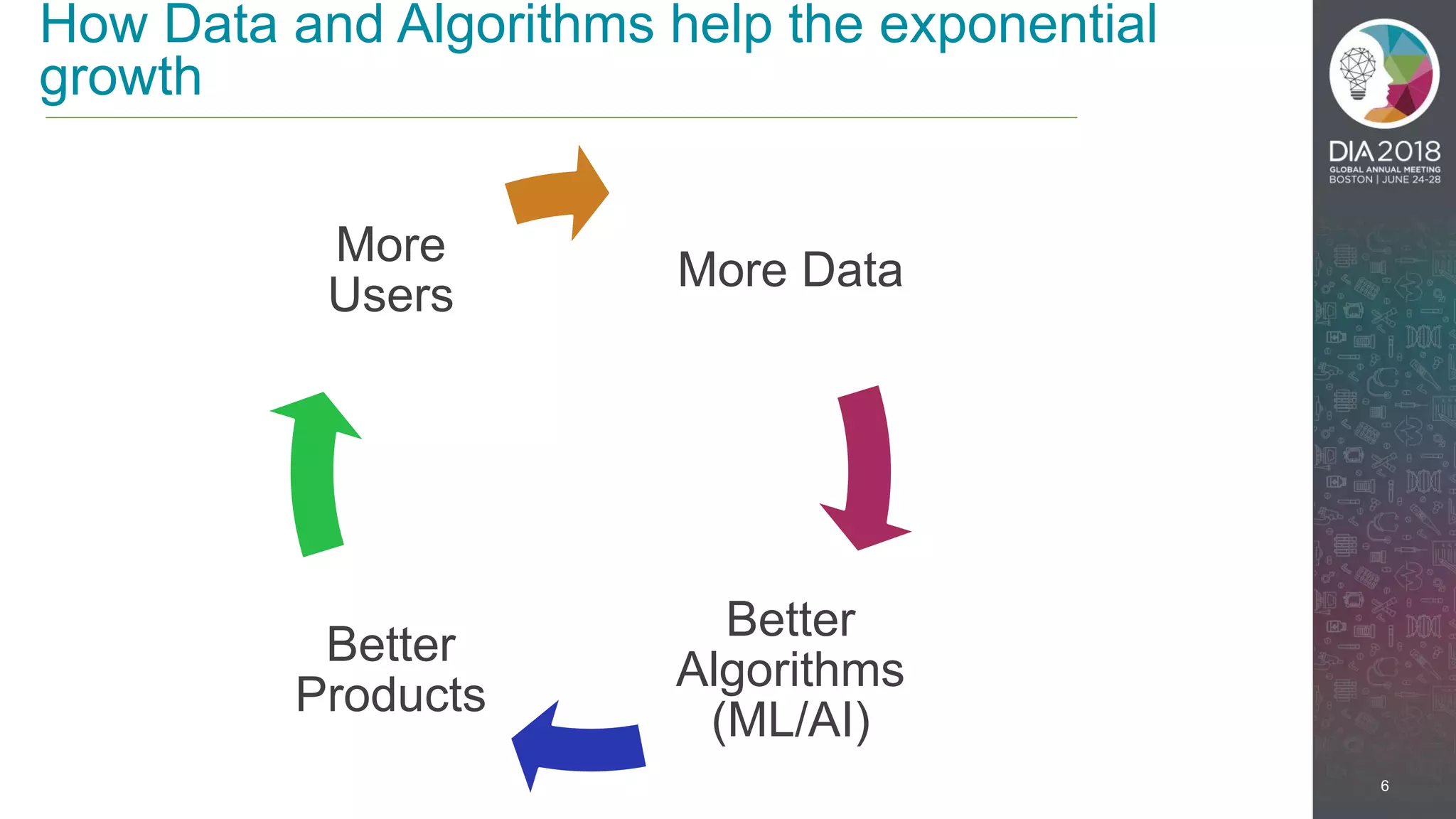
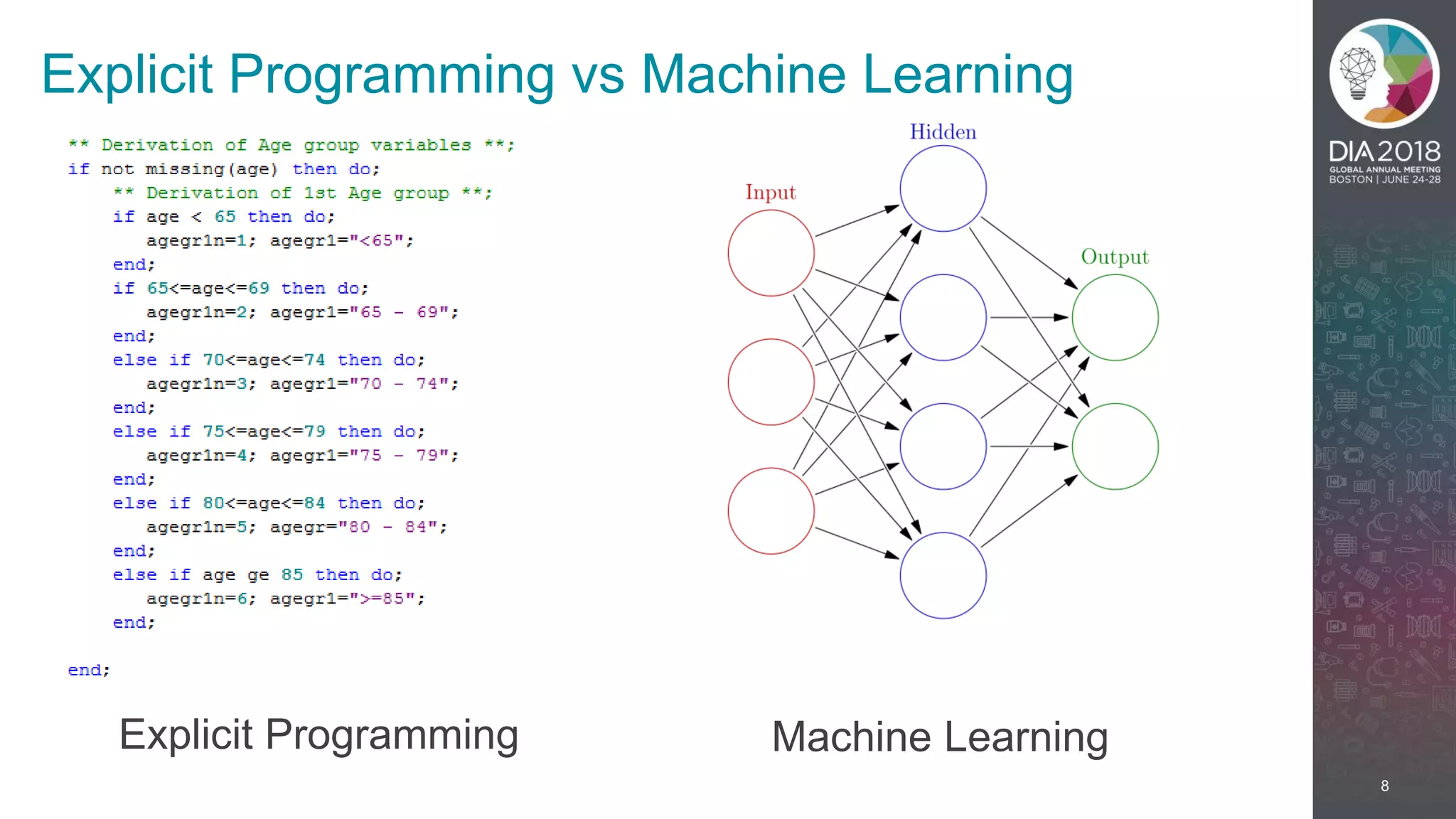
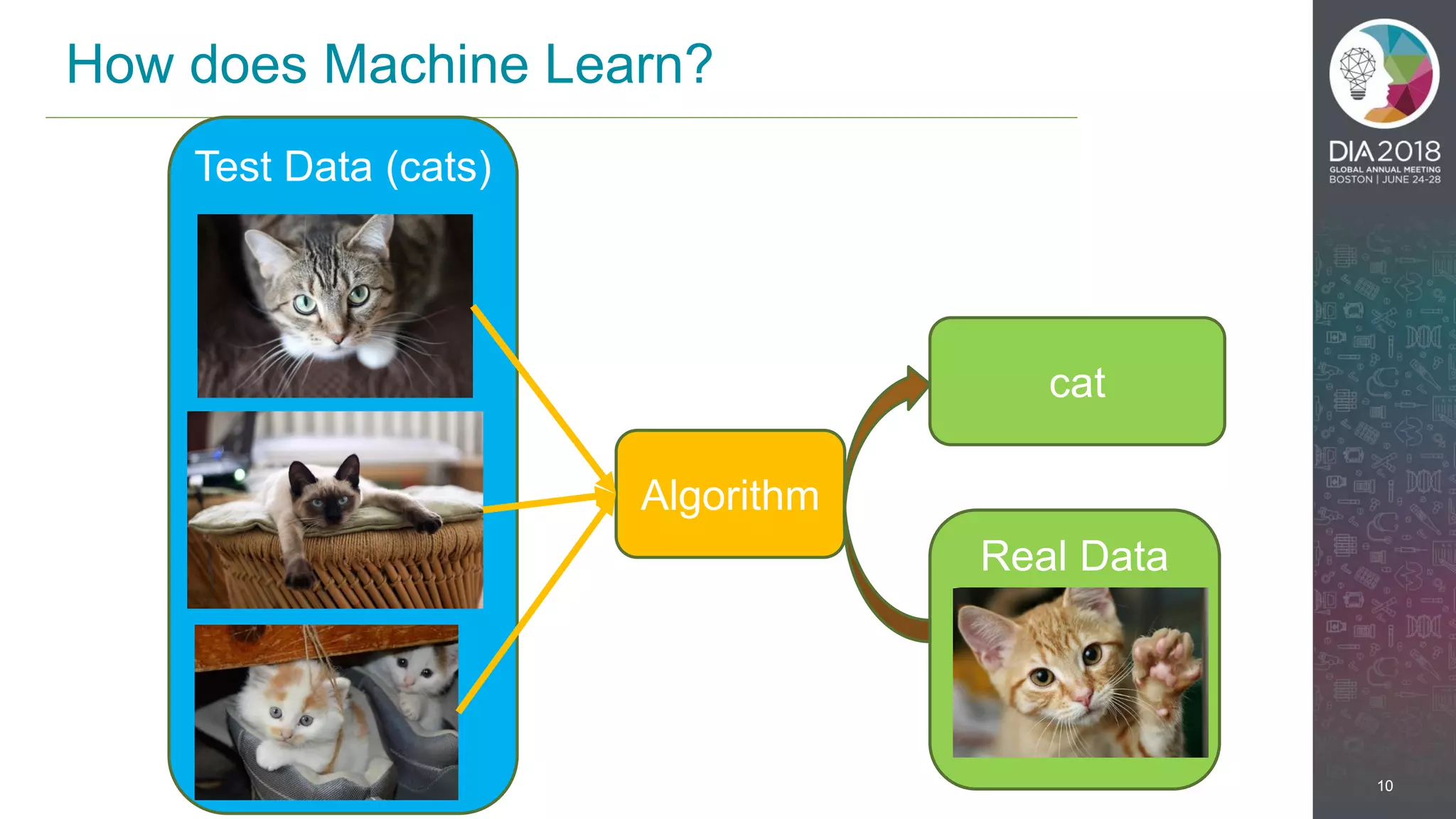
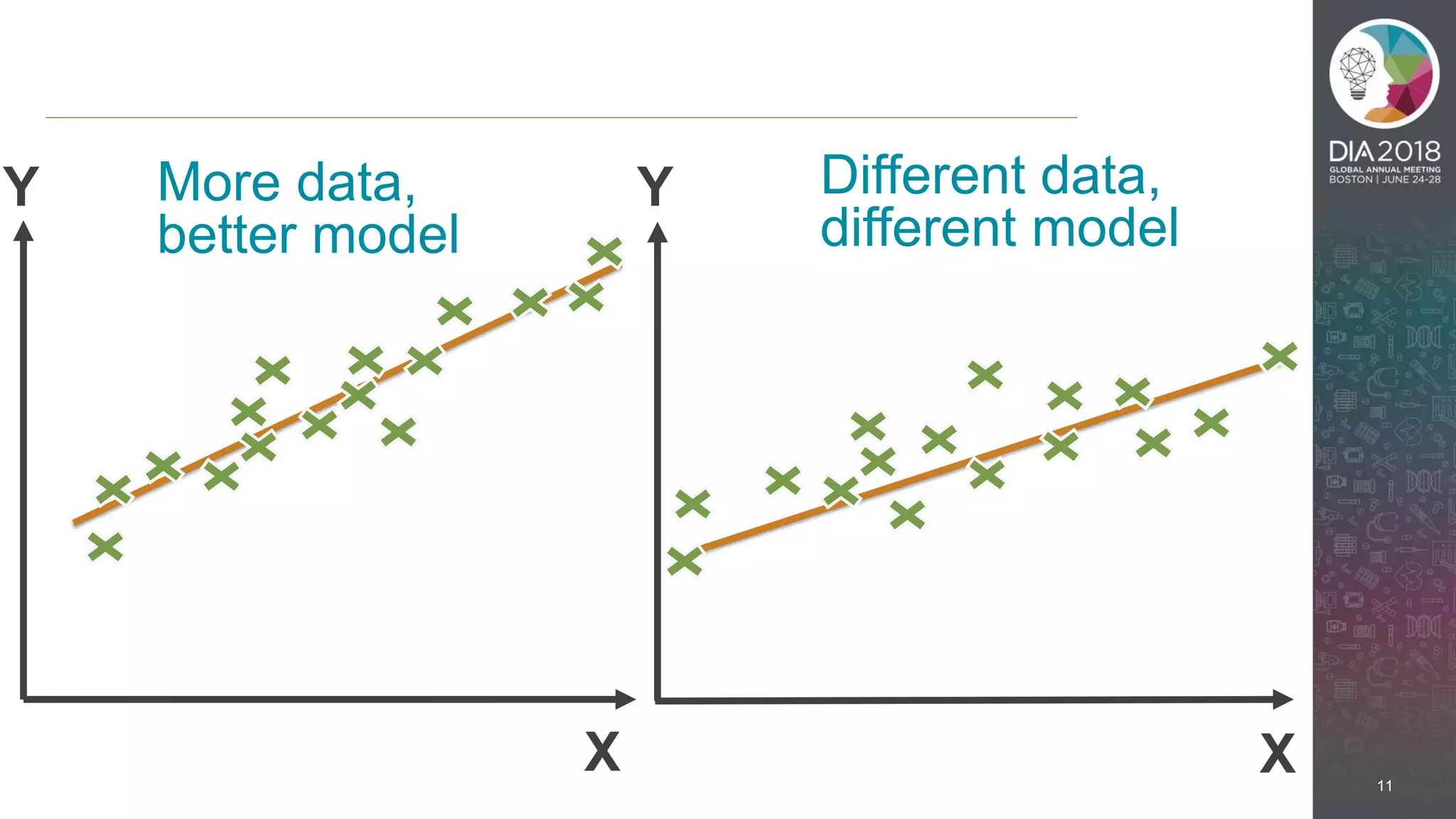
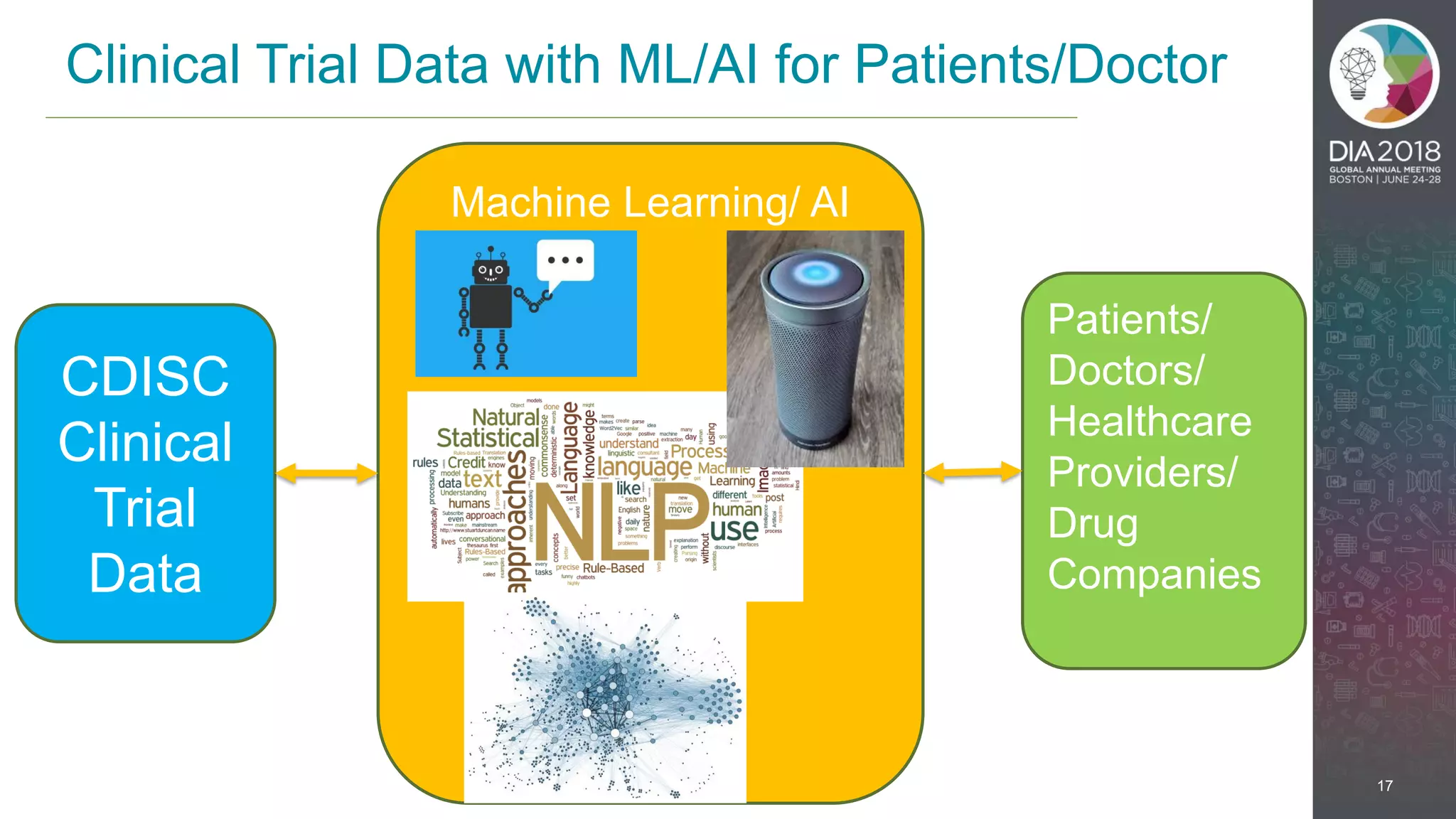
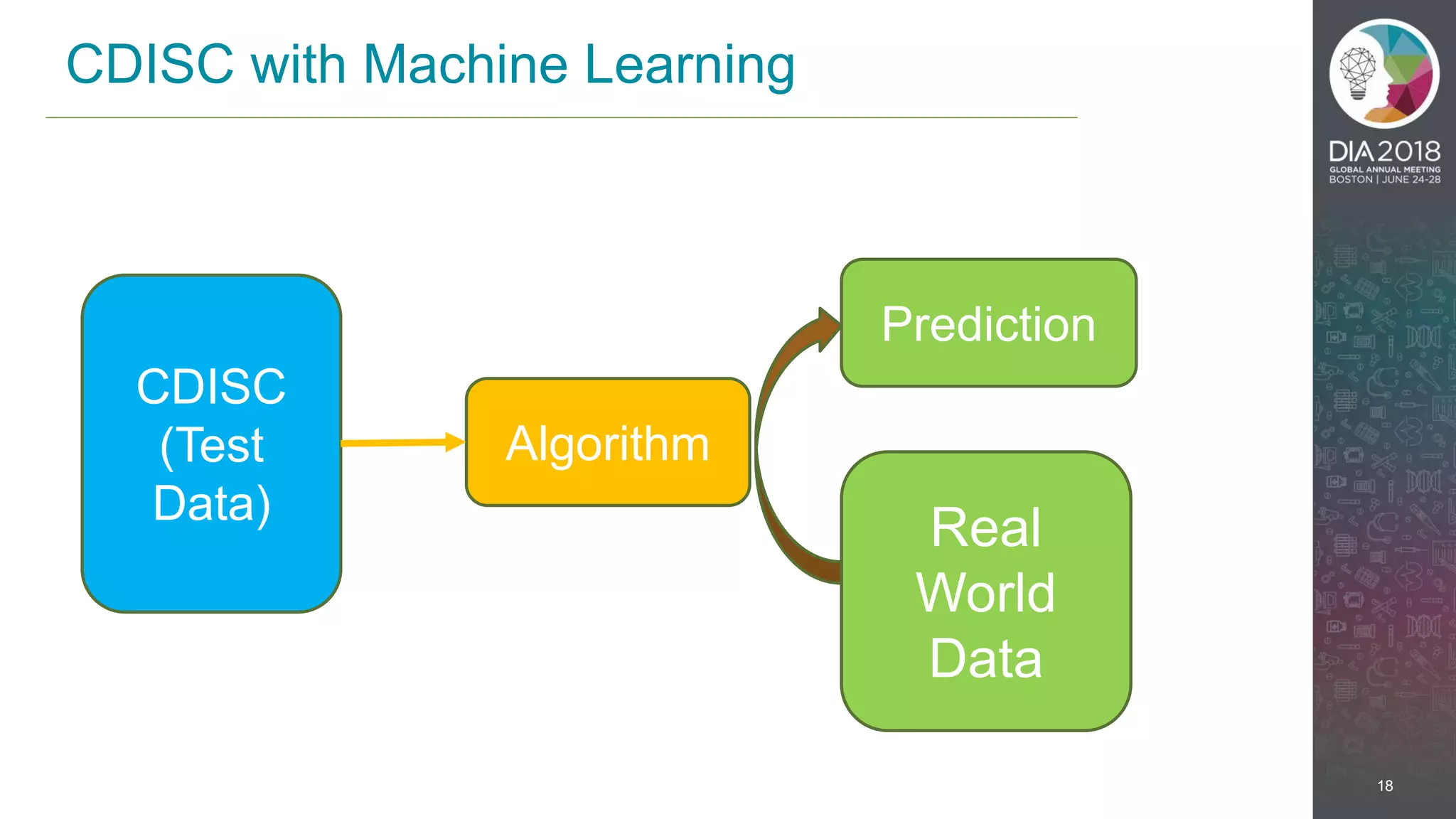
The presentation discusses the intersection of machine learning and Clinical Data Interchange Standards Consortium (CDISC), emphasizing how these technologies can transform pharmaceutical data management and clinical trials. It highlights the exponential growth driven by better data and algorithms, illustrating the practical applications in healthcare. The speaker, Kevin Lee, posits that machine learning can facilitate enhanced patient outcomes and more effective drug development.