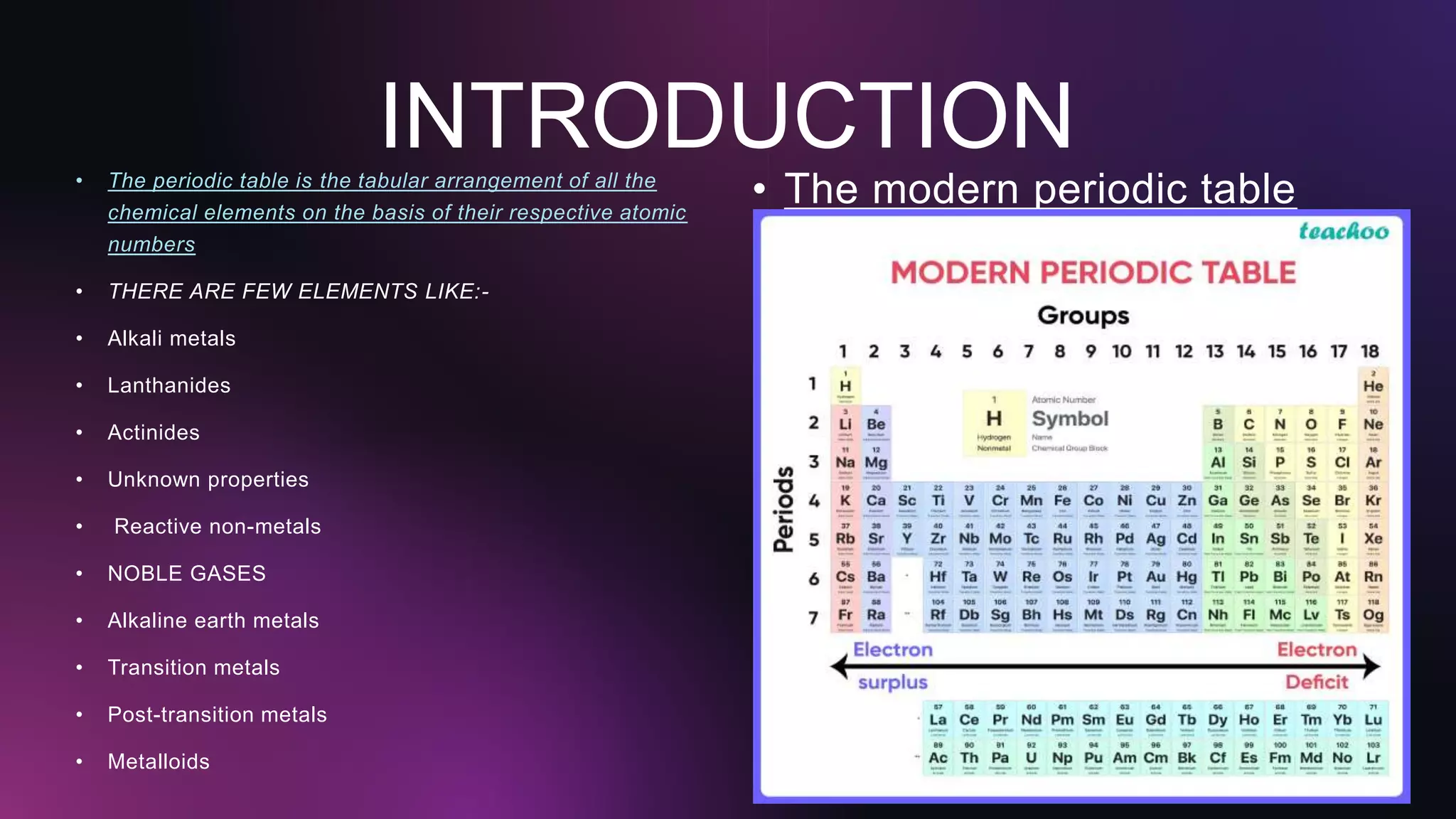
The document presents an overview of the modern periodic table, detailing its creation by key chemists such as Johann Wolfgang Döbereiner, John Newlands, and Dmitri Mendeleev. It highlights the advantages and limitations of the table, including its foundation on atomic numbers and issues such as the placement of hydrogen and the absence of isotopes. The presentation concludes with a call to appreciate the contributions of these scientists to chemistry.