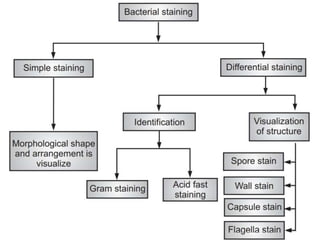
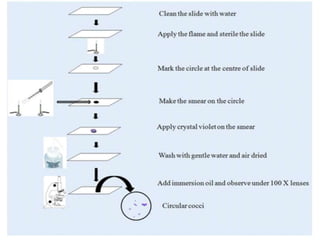
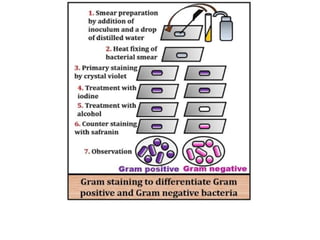
Staining techniques are used in microbiology to identify bacteria under a microscope. There are several types of staining including simple staining with one dye, Gram staining which differentiates bacteria as Gram-positive or Gram-negative based on cell wall structure, and acid-fast staining used to identify Mycobacterium species. Biochemical tests such as IMViC (Indole, Methyl Red, Voges-Proskauer, Citrate) are also used to identify bacteria based on their metabolic reactions and products.