lecture37.pdf
•
0 likes•17 views
This document discusses electrochemical cells and half-reactions. It defines electrochemical cells as consisting of two electrodes separated by an electrolyte. In a galvanic cell, spontaneous reactions occur that produce electricity. In an electrolytic cell, an external current is used to drive nonspontaneous reactions. Redox reactions can be written as the sum of oxidation and reduction half-reactions. The document also describes different types of electrodes, including gas electrodes using hydrogen and redox electrodes like the Daniell cell. It explains how the cell potential is the difference between electrode potentials and relates this to the maximum work done by the cell.
Report
Share
Report
Share
Download to read offline
Recommended
Electrochemistry 12

it contain class 12 starting material. student use this for maharashtra board, cbse board etc.
animation only work when you download.
electrochemistry12.pdf

This document discusses redox reactions, electrochemistry, and electrochemical cells. It begins by defining key concepts like oxidation, reduction, oxidizing agents, and reducing agents. It then provides examples of redox reactions and discusses how electrochemical cells work. The rest of the document covers topics like cell notation, standard electrode potentials, how to determine if a redox reaction is spontaneous, the relationship between cell potential and Gibbs free energy, the effect of concentration on cell potential, corrosion, batteries, and different types of electrochemical cells like voltaic cells, electrolytic cells, and fuel cells.
electrochemistry12.pdf

This document discusses redox reactions, electrochemistry, and electrochemical cells. It begins by defining key concepts like oxidation, reduction, oxidizing agents, and reducing agents. It then provides examples of redox reactions and discusses how electrochemical cells work. The rest of the document covers topics like cell notation, standard electrode potentials, how to determine if a redox reaction is spontaneous, the relationship between cell potential and Gibbs free energy, the effect of concentration on cell potential, corrosion, batteries, and different types of electrochemical cells like voltaic cells, electrolytic cells, and fuel cells.
Electrochemistry

This document provides an overview of electrochemistry and galvanic cells. Some key points:
- Electron transfer reactions are oxidation-reduction (redox) reactions that can generate electric current or be driven by an applied current, making it the field of electrochemistry.
- Galvanic cells use spontaneous redox reactions to generate electricity, with oxidation occurring at the anode and reduction at the cathode. The potential difference between electrodes is called the cell voltage.
- Standard electrode potentials (E°) describe the tendency of half-reactions to occur and can be used to predict spontaneity of redox reactions in cells. Nernst equation relates cell potential to concentrations.
23sec3

An electrolytic cell uses electrical energy to drive non-spontaneous redox reactions by pushing electrons from the anode to the cathode. In electrolytic cells, oxidation occurs at the anode and reduction occurs at the cathode, unlike voltaic cells where the reactions are spontaneous. A common example is the electrolysis of water, where applying a current splits water into hydrogen gas at the cathode and oxygen gas at the anode. Electrolysis can also be used to produce chlorine from molten sodium chloride or to electroplate metals onto objects by reducing metal ions at the cathode.
New chm-152-unit-8-power-points-sp13-140227172047-phpapp01

The document provides information about electrochemistry including:
1) It discusses voltaic (galvanic) cells and electrolytic cells, how they are constructed using two electrodes in an electrolyte solution, and the definitions of anode and cathode.
2) It describes the zinc-copper cell as an example, showing the oxidation and reduction half-reactions, overall reaction, and cell notation. The initial voltage is given as 1.10 volts.
3) It explains how standard electrode potentials are measured relative to the standard hydrogen electrode, which has a defined potential of 0.00 V. Standard potentials allow comparison of an electrode's ability to be reduced or act as an oxidizing agent.
Chapter 12 -electrochemical cells

This document provides information about oxidation-reduction (redox) reactions and electrochemical cells. It discusses how redox reactions can be separated into half-reactions that occur at different electrodes. As an example, it examines the redox reaction between copper and silver ions and how this reaction spontaneously occurs when the metals are placed in their respective ion solutions, but not in the reverse direction. It also explains how electrochemical cells like galvanic cells and electrolytic cells use separated half-reactions to generate a voltage or cause non-spontaneous reactions, respectively. Key concepts covered include the standard hydrogen electrode, conventions for assigning electrode potentials, and how voltages relate to the thermodynamics of redox processes.
Recommended
Electrochemistry 12

it contain class 12 starting material. student use this for maharashtra board, cbse board etc.
animation only work when you download.
electrochemistry12.pdf

This document discusses redox reactions, electrochemistry, and electrochemical cells. It begins by defining key concepts like oxidation, reduction, oxidizing agents, and reducing agents. It then provides examples of redox reactions and discusses how electrochemical cells work. The rest of the document covers topics like cell notation, standard electrode potentials, how to determine if a redox reaction is spontaneous, the relationship between cell potential and Gibbs free energy, the effect of concentration on cell potential, corrosion, batteries, and different types of electrochemical cells like voltaic cells, electrolytic cells, and fuel cells.
electrochemistry12.pdf

This document discusses redox reactions, electrochemistry, and electrochemical cells. It begins by defining key concepts like oxidation, reduction, oxidizing agents, and reducing agents. It then provides examples of redox reactions and discusses how electrochemical cells work. The rest of the document covers topics like cell notation, standard electrode potentials, how to determine if a redox reaction is spontaneous, the relationship between cell potential and Gibbs free energy, the effect of concentration on cell potential, corrosion, batteries, and different types of electrochemical cells like voltaic cells, electrolytic cells, and fuel cells.
Electrochemistry

This document provides an overview of electrochemistry and galvanic cells. Some key points:
- Electron transfer reactions are oxidation-reduction (redox) reactions that can generate electric current or be driven by an applied current, making it the field of electrochemistry.
- Galvanic cells use spontaneous redox reactions to generate electricity, with oxidation occurring at the anode and reduction at the cathode. The potential difference between electrodes is called the cell voltage.
- Standard electrode potentials (E°) describe the tendency of half-reactions to occur and can be used to predict spontaneity of redox reactions in cells. Nernst equation relates cell potential to concentrations.
23sec3

An electrolytic cell uses electrical energy to drive non-spontaneous redox reactions by pushing electrons from the anode to the cathode. In electrolytic cells, oxidation occurs at the anode and reduction occurs at the cathode, unlike voltaic cells where the reactions are spontaneous. A common example is the electrolysis of water, where applying a current splits water into hydrogen gas at the cathode and oxygen gas at the anode. Electrolysis can also be used to produce chlorine from molten sodium chloride or to electroplate metals onto objects by reducing metal ions at the cathode.
New chm-152-unit-8-power-points-sp13-140227172047-phpapp01

The document provides information about electrochemistry including:
1) It discusses voltaic (galvanic) cells and electrolytic cells, how they are constructed using two electrodes in an electrolyte solution, and the definitions of anode and cathode.
2) It describes the zinc-copper cell as an example, showing the oxidation and reduction half-reactions, overall reaction, and cell notation. The initial voltage is given as 1.10 volts.
3) It explains how standard electrode potentials are measured relative to the standard hydrogen electrode, which has a defined potential of 0.00 V. Standard potentials allow comparison of an electrode's ability to be reduced or act as an oxidizing agent.
Chapter 12 -electrochemical cells

This document provides information about oxidation-reduction (redox) reactions and electrochemical cells. It discusses how redox reactions can be separated into half-reactions that occur at different electrodes. As an example, it examines the redox reaction between copper and silver ions and how this reaction spontaneously occurs when the metals are placed in their respective ion solutions, but not in the reverse direction. It also explains how electrochemical cells like galvanic cells and electrolytic cells use separated half-reactions to generate a voltage or cause non-spontaneous reactions, respectively. Key concepts covered include the standard hydrogen electrode, conventions for assigning electrode potentials, and how voltages relate to the thermodynamics of redox processes.
Electrochemistry

Types of electrodes, Applications of EMF, Determination of pH, Faradays law of electrolysis, Electrolysis of sodium chloride, Ionic strength
Electrochemistry Lec 2021_.ppt

This document provides an overview of electrochemistry concepts including:
- Types of electrochemical processes including reversible and irreversible processes.
- Oxidation-reduction reactions and how they involve oxidation and reduction half-reactions.
- Galvanic/voltaic cells and how they generate electricity from spontaneous redox reactions.
- Components of electrochemical cells including electrodes, salt bridges, and how they allow indirect redox reactions.
- Standard electrode potentials and how they are used to determine if a reaction is spontaneous.
- The Nernst equation and how it describes the dependence of electrode potential on ion concentration.
Electrochemistry_Grade_12.pptx

This document discusses various topics in electrochemistry including redox reactions, balancing redox equations, galvanic cells, standard reduction potentials, and applications such as corrosion and electrolysis. It defines key terms like oxidation, reduction, and half-reactions. Methods for balancing redox equations under acidic and basic conditions are explained. Components of galvanic cells like anodes, cathodes, and salt bridges are defined. Standard reduction potentials are used to determine cell potentials. Examples of galvanic cells and their notations are provided. Corrosion prevention methods and commercial electrolysis processes are briefly described.
Electrochemistry apps of redox

This document provides an overview of redox reactions and electrochemistry applications. It discusses oxidation-reduction concepts like oxidation states and the OIL RIG mnemonic. Examples of redox reactions and electrochemistry applications are given, including galvanic cells, corrosion, electrolysis, and batteries. Key concepts covered include cell potential, the Nernst equation, and how concentration affects cell potential. Diagrams illustrate galvanic cells and how they function.
Lecture 21- Electrochemical cells

This document discusses electrochemical cells and how they produce electric current from redox reactions. It explains that a voltaic cell separates the oxidation and reduction half-reactions into different compartments connected by a salt bridge and wire. The document provides examples of how to construct half-cells for zinc-copper and aluminum-nickel voltaic cells and calculates cell voltage using reduction potentials from a table.
apchapt17.ppt

This document provides information on oxidation-reduction (redox) reactions and electrochemistry:
[1] Redox reactions involve the transfer of electrons between oxidizing and reducing agents. Common examples are corrosion reactions.
[2] Galvanic (voltaic) cells generate electricity through spontaneous redox reactions. The anode is where oxidation occurs and electrons are released. The cathode is where reduction occurs and electrons are gained.
[3] Cell potential depends on the relative tendencies of substances to be oxidized or reduced, as measured by standard reduction potentials. More negative potentials indicate greater reducing ability; more positive potentials indicate greater oxidizing ability.
Electrochemistry by rawat

This document discusses redox reactions and electrochemistry. It covers topics such as oxidation numbers, galvanic cells, cell notation, standard electrode potentials, and how cell potential relates to Gibbs free energy and equilibrium constants. It also discusses corrosion, batteries, fuel cells, and the differences between voltaic, electrolytic, and fuel cells. Redox reactions allow the interconversion of electrical and chemical energy.
كيمياء كهربائية.pdf

This document discusses various topics in electrochemistry including different types of electrodes, applications of electromotive force measurements, Faraday's law of electrolysis, and determination of pH. It describes metal-metal ion electrodes, metal-amalgam electrodes, metal insoluble metal salt electrodes, and gas electrodes. It also discusses applications for determining ion valency, solubility products, and thermodynamic properties from electromotive force measurements. Determination of pH using glass electrodes and the electrolysis of sodium chloride and copper sulfate solutions are summarized. Ionic strength and electrode potential are also briefly covered.
Electrochemistry

This document provides an overview of electrochemistry. It begins by defining electrochemistry as the study of chemical reactions at the interface of an electrode and electrolyte involving the interaction of electrical and chemical changes. The document then discusses the history and founders of electrochemistry, including Faraday's two laws of electrolysis. It explains key concepts such as oxidation-reduction reactions, balancing redox equations, and the Nernst equation. The document also covers applications including batteries, corrosion, electrolysis, and branches of electrochemistry like bioelectrochemistry and nanoelectrochemistry.
IB Chemistry on Electrolysis and Faraday's Law

This document provides a tutorial on electrolysis and Faraday's law. It discusses the differences between voltaic cells and electrolytic cells. In a voltaic cell, chemical energy is converted to electrical energy through spontaneous redox reactions. In an electrolytic cell, electrical energy is converted to chemical energy by using an external voltage to drive non-spontaneous redox reactions, such as decomposing ionic compounds through electrolysis of molten salts or aqueous solutions. Several examples of voltaic and electrolytic cells are presented, including calculations of cell potentials using standard reduction potentials. Factors that influence which ions are discharged during electrolysis are also described.
Class 11 Chapter 8 Redox Reactions.pptx

1) The document discusses classical ideas of oxidation and reduction reactions by defining them as addition or removal of oxygen, hydrogen, or electronegative/electropositive elements.
2) It then moves to discussing redox reactions in terms of electron transfer, defining oxidation as loss of electrons and reduction as gain of electrons.
3) Rules for calculating oxidation numbers are provided, including that the sum of oxidation numbers in a compound or ion must equal the overall charge. Stock notation is also introduced for representing oxidation states.
4) Examples are given of identifying oxidizing and reducing agents, balancing redox reactions using the oxidation number method, and classifying reactions as redox based on changes in oxidation numbers.
3rd Lecture on Electrochemistry | Chemistry Part I | 12th Std

This document summarizes key aspects of electrochemistry including:
1) An electrochemical cell consists of two electrodes immersed in an electrolyte that allows ionic conduction. Oxidation occurs at the anode and reduction at the cathode.
2) There are two types of cells - electrolytic cells use an external power source to drive nonspontaneous reactions, while galvanic/voltaic cells generate power from spontaneous reactions.
3) The electrolysis of molten NaCl produces chlorine gas at the anode and deposits metallic sodium at the cathode, while electrolysis of aqueous NaCl produces hydrogen and chlorine gas due to the lower standard potentials of sodium and chlorine ions compared to water.
electrochemistry-141128223112-conversion-gate02.pptx

Conductors and Non-Conductors
Substances can be classified as conductors and non-conductors based on their ability to conduct electricity.
Conductors: Substances that allow electric current to flow through them are called conductors. For example, Plastic, Wood, etc.
Non-Conductors: Non-conductors are insulators that do not allow electricity to pass through them. For example, Copper, Iron, etc.
Types of Conductors
Conductors are divided into two groups: Metallic conductors and Electrolytes.
Metallic Conductors: These conductors conduct electricity by the movement of electrons without any chemical change during the process. This type of conduction happens in solids and in the molten state.
Electrolytes: They conduct electricity by the movement of the ions in the solutions. It is present in the aqueous solution.
Distinguish between Metallic and Electrolytic Conduction
Metallic Conduction Electrolytic Conduction
The movement of electrons causes the electric current The movement of ions causes the electric current
There is no chemical reaction Ions get ionised or reduced at the electrodes
There is no transfer of matter It involves the transfer of matter in the form of ions
Follows Ohm’s law Follows Ohm’s law
Resistance increases with an increase in temperature Resistance decreases with an increase in temperature
Faraday’s law is not followed Follows Faraday’s law
Electrolytes
(a) Substances whose aqueous solutions allow the conductance of electric current and are chemically decomposed are called electrolytes.
(b) The positively charged ions furnished by the electrolyte are called cations, while the negatively charged ions furnished by the electrolyte are called anions.
Types of Electrolytes
(a) Weak electrolytes: Electrolytes that are decomposable to a very small extent in their dilute solutions are called weak electrolytes. For example, organic acids, inorganic acids and bases etc.
(b) Strong electrolytes: Electrolytes that are highly decomposable in aqueous solution and conduct electricity frequently are called electrolytes. For example, mineral acid and salts of strong acid.
Electrode
For the electric current to pass through an electrolytic conductor, the two rods or plates called electrodes are always needed. These plates are connected to the terminals of the battery to form a cell. The electrode through which the electric current flows into the electrolytic solution is called the anode, also called the positive electrode, and anions are oxidised here.
An electrode through which the electric current flows out of the electrolytic solution is called the cathode, also called the negative electrode, and cations are reduced there.
Electrolysis
Electrolysis is the process of chemical deposition of the electrolyte by passing an electric current. Electrolysis takes place in an electrolytic cell. This cell will convert the electrical energy to chemical energy.
Basic of Corrosion

Corrosion is the deterioration of materials through chemical reactions with their environment. It occurs via electrochemical processes where the metal surface acts as an anode and electrons flow to a cathode. Factors like material properties, environment, temperature affect corrosion. Corrosion causes economic losses and safety issues. Understanding corrosion mechanisms and factors is important for material selection, design, and prevention of failures in engineering structures.
New chm-152-unit-9-power-points-sp13-140227172048-phpapp02

This document discusses electrolysis and Faraday's law of electrolysis. It begins by introducing electrolytic cells and predicting the products of electrolysis. It then explains that according to Faraday's law, the amount of substance produced at each electrode is directly proportional to the quantity of electricity passing through the cell. The document provides an example calculation applying Faraday's law to determine the current needed in an electrolysis process. It also discusses some key aspects of Faraday's law, such as defining the faraday as one mole of electrons.
Module 2_S7 and S8_Electrochemical Cells.pptx

An electrochemical cell consists of two electrodes separated by an electrolyte. There are two types: galvanic cells and electrolytic cells. A galvanic cell converts the chemical energy of a spontaneous redox reaction directly into electrical energy. The Nernst equation relates the cell potential (E) of a galvanic cell to the standard potential (E0), temperature, and reaction quotient through the concentrations of reactants and products. It allows calculation of cell potential under non-standard conditions.
B.tech. ii engineering chemistry unit 5 A electrochemistry

Arrhenius proposed the theory of electrolytic dissociation to explain the properties of electrolytic solutions. The theory states that when an electrolyte dissolves in water, it breaks up into ions - positively charged cations and negatively charged anions. This process is called ionization. Ions are constantly recombining and dissociating, reaching a state of dynamic equilibrium. The extent of ionization depends on an equilibrium constant. Strong electrolytes have a high equilibrium constant and ionize completely, while weak electrolytes have a low constant and only partially ionize.
Chapter - 6 (Electrochemistry).ppt

Electroanalytical methods provide several advantages for quantitative analytical chemistry. They involve measuring the electrical properties of analyte solutions in electrochemical cells. Some key points:
- Electroanalytical methods allow easy automation through electrical signal measurements. They can also determine low analyte concentrations without difficulty.
- Electrochemical processes involve the transfer of electrons between substances during redox reactions. This occurs at the interface between electrodes and solutions in electrochemical cells.
- Advantages include low cost compared to spectroscopy and the ability to easily automate measurements and detect low analyte concentrations through electrical signals.
Large Language Model (LLM) and it’s Geospatial Applications

Large Language Model (LLM) and it’s Geospatial Applications.
Artificial Intelligence for XMLDevelopment

In the rapidly evolving landscape of technologies, XML continues to play a vital role in structuring, storing, and transporting data across diverse systems. The recent advancements in artificial intelligence (AI) present new methodologies for enhancing XML development workflows, introducing efficiency, automation, and intelligent capabilities. This presentation will outline the scope and perspective of utilizing AI in XML development. The potential benefits and the possible pitfalls will be highlighted, providing a balanced view of the subject.
We will explore the capabilities of AI in understanding XML markup languages and autonomously creating structured XML content. Additionally, we will examine the capacity of AI to enrich plain text with appropriate XML markup. Practical examples and methodological guidelines will be provided to elucidate how AI can be effectively prompted to interpret and generate accurate XML markup.
Further emphasis will be placed on the role of AI in developing XSLT, or schemas such as XSD and Schematron. We will address the techniques and strategies adopted to create prompts for generating code, explaining code, or refactoring the code, and the results achieved.
The discussion will extend to how AI can be used to transform XML content. In particular, the focus will be on the use of AI XPath extension functions in XSLT, Schematron, Schematron Quick Fixes, or for XML content refactoring.
The presentation aims to deliver a comprehensive overview of AI usage in XML development, providing attendees with the necessary knowledge to make informed decisions. Whether you’re at the early stages of adopting AI or considering integrating it in advanced XML development, this presentation will cover all levels of expertise.
By highlighting the potential advantages and challenges of integrating AI with XML development tools and languages, the presentation seeks to inspire thoughtful conversation around the future of XML development. We’ll not only delve into the technical aspects of AI-powered XML development but also discuss practical implications and possible future directions.
How to Get CNIC Information System with Paksim Ga.pptx

Pakdata Cf is a groundbreaking system designed to streamline and facilitate access to CNIC information. This innovative platform leverages advanced technology to provide users with efficient and secure access to their CNIC details.
Building RAG with self-deployed Milvus vector database and Snowpark Container...

This talk will give hands-on advice on building RAG applications with an open-source Milvus database deployed as a docker container. We will also introduce the integration of Milvus with Snowpark Container Services.
More Related Content
Similar to lecture37.pdf
Electrochemistry

Types of electrodes, Applications of EMF, Determination of pH, Faradays law of electrolysis, Electrolysis of sodium chloride, Ionic strength
Electrochemistry Lec 2021_.ppt

This document provides an overview of electrochemistry concepts including:
- Types of electrochemical processes including reversible and irreversible processes.
- Oxidation-reduction reactions and how they involve oxidation and reduction half-reactions.
- Galvanic/voltaic cells and how they generate electricity from spontaneous redox reactions.
- Components of electrochemical cells including electrodes, salt bridges, and how they allow indirect redox reactions.
- Standard electrode potentials and how they are used to determine if a reaction is spontaneous.
- The Nernst equation and how it describes the dependence of electrode potential on ion concentration.
Electrochemistry_Grade_12.pptx

This document discusses various topics in electrochemistry including redox reactions, balancing redox equations, galvanic cells, standard reduction potentials, and applications such as corrosion and electrolysis. It defines key terms like oxidation, reduction, and half-reactions. Methods for balancing redox equations under acidic and basic conditions are explained. Components of galvanic cells like anodes, cathodes, and salt bridges are defined. Standard reduction potentials are used to determine cell potentials. Examples of galvanic cells and their notations are provided. Corrosion prevention methods and commercial electrolysis processes are briefly described.
Electrochemistry apps of redox

This document provides an overview of redox reactions and electrochemistry applications. It discusses oxidation-reduction concepts like oxidation states and the OIL RIG mnemonic. Examples of redox reactions and electrochemistry applications are given, including galvanic cells, corrosion, electrolysis, and batteries. Key concepts covered include cell potential, the Nernst equation, and how concentration affects cell potential. Diagrams illustrate galvanic cells and how they function.
Lecture 21- Electrochemical cells

This document discusses electrochemical cells and how they produce electric current from redox reactions. It explains that a voltaic cell separates the oxidation and reduction half-reactions into different compartments connected by a salt bridge and wire. The document provides examples of how to construct half-cells for zinc-copper and aluminum-nickel voltaic cells and calculates cell voltage using reduction potentials from a table.
apchapt17.ppt

This document provides information on oxidation-reduction (redox) reactions and electrochemistry:
[1] Redox reactions involve the transfer of electrons between oxidizing and reducing agents. Common examples are corrosion reactions.
[2] Galvanic (voltaic) cells generate electricity through spontaneous redox reactions. The anode is where oxidation occurs and electrons are released. The cathode is where reduction occurs and electrons are gained.
[3] Cell potential depends on the relative tendencies of substances to be oxidized or reduced, as measured by standard reduction potentials. More negative potentials indicate greater reducing ability; more positive potentials indicate greater oxidizing ability.
Electrochemistry by rawat

This document discusses redox reactions and electrochemistry. It covers topics such as oxidation numbers, galvanic cells, cell notation, standard electrode potentials, and how cell potential relates to Gibbs free energy and equilibrium constants. It also discusses corrosion, batteries, fuel cells, and the differences between voltaic, electrolytic, and fuel cells. Redox reactions allow the interconversion of electrical and chemical energy.
كيمياء كهربائية.pdf

This document discusses various topics in electrochemistry including different types of electrodes, applications of electromotive force measurements, Faraday's law of electrolysis, and determination of pH. It describes metal-metal ion electrodes, metal-amalgam electrodes, metal insoluble metal salt electrodes, and gas electrodes. It also discusses applications for determining ion valency, solubility products, and thermodynamic properties from electromotive force measurements. Determination of pH using glass electrodes and the electrolysis of sodium chloride and copper sulfate solutions are summarized. Ionic strength and electrode potential are also briefly covered.
Electrochemistry

This document provides an overview of electrochemistry. It begins by defining electrochemistry as the study of chemical reactions at the interface of an electrode and electrolyte involving the interaction of electrical and chemical changes. The document then discusses the history and founders of electrochemistry, including Faraday's two laws of electrolysis. It explains key concepts such as oxidation-reduction reactions, balancing redox equations, and the Nernst equation. The document also covers applications including batteries, corrosion, electrolysis, and branches of electrochemistry like bioelectrochemistry and nanoelectrochemistry.
IB Chemistry on Electrolysis and Faraday's Law

This document provides a tutorial on electrolysis and Faraday's law. It discusses the differences between voltaic cells and electrolytic cells. In a voltaic cell, chemical energy is converted to electrical energy through spontaneous redox reactions. In an electrolytic cell, electrical energy is converted to chemical energy by using an external voltage to drive non-spontaneous redox reactions, such as decomposing ionic compounds through electrolysis of molten salts or aqueous solutions. Several examples of voltaic and electrolytic cells are presented, including calculations of cell potentials using standard reduction potentials. Factors that influence which ions are discharged during electrolysis are also described.
Class 11 Chapter 8 Redox Reactions.pptx

1) The document discusses classical ideas of oxidation and reduction reactions by defining them as addition or removal of oxygen, hydrogen, or electronegative/electropositive elements.
2) It then moves to discussing redox reactions in terms of electron transfer, defining oxidation as loss of electrons and reduction as gain of electrons.
3) Rules for calculating oxidation numbers are provided, including that the sum of oxidation numbers in a compound or ion must equal the overall charge. Stock notation is also introduced for representing oxidation states.
4) Examples are given of identifying oxidizing and reducing agents, balancing redox reactions using the oxidation number method, and classifying reactions as redox based on changes in oxidation numbers.
3rd Lecture on Electrochemistry | Chemistry Part I | 12th Std

This document summarizes key aspects of electrochemistry including:
1) An electrochemical cell consists of two electrodes immersed in an electrolyte that allows ionic conduction. Oxidation occurs at the anode and reduction at the cathode.
2) There are two types of cells - electrolytic cells use an external power source to drive nonspontaneous reactions, while galvanic/voltaic cells generate power from spontaneous reactions.
3) The electrolysis of molten NaCl produces chlorine gas at the anode and deposits metallic sodium at the cathode, while electrolysis of aqueous NaCl produces hydrogen and chlorine gas due to the lower standard potentials of sodium and chlorine ions compared to water.
electrochemistry-141128223112-conversion-gate02.pptx

Conductors and Non-Conductors
Substances can be classified as conductors and non-conductors based on their ability to conduct electricity.
Conductors: Substances that allow electric current to flow through them are called conductors. For example, Plastic, Wood, etc.
Non-Conductors: Non-conductors are insulators that do not allow electricity to pass through them. For example, Copper, Iron, etc.
Types of Conductors
Conductors are divided into two groups: Metallic conductors and Electrolytes.
Metallic Conductors: These conductors conduct electricity by the movement of electrons without any chemical change during the process. This type of conduction happens in solids and in the molten state.
Electrolytes: They conduct electricity by the movement of the ions in the solutions. It is present in the aqueous solution.
Distinguish between Metallic and Electrolytic Conduction
Metallic Conduction Electrolytic Conduction
The movement of electrons causes the electric current The movement of ions causes the electric current
There is no chemical reaction Ions get ionised or reduced at the electrodes
There is no transfer of matter It involves the transfer of matter in the form of ions
Follows Ohm’s law Follows Ohm’s law
Resistance increases with an increase in temperature Resistance decreases with an increase in temperature
Faraday’s law is not followed Follows Faraday’s law
Electrolytes
(a) Substances whose aqueous solutions allow the conductance of electric current and are chemically decomposed are called electrolytes.
(b) The positively charged ions furnished by the electrolyte are called cations, while the negatively charged ions furnished by the electrolyte are called anions.
Types of Electrolytes
(a) Weak electrolytes: Electrolytes that are decomposable to a very small extent in their dilute solutions are called weak electrolytes. For example, organic acids, inorganic acids and bases etc.
(b) Strong electrolytes: Electrolytes that are highly decomposable in aqueous solution and conduct electricity frequently are called electrolytes. For example, mineral acid and salts of strong acid.
Electrode
For the electric current to pass through an electrolytic conductor, the two rods or plates called electrodes are always needed. These plates are connected to the terminals of the battery to form a cell. The electrode through which the electric current flows into the electrolytic solution is called the anode, also called the positive electrode, and anions are oxidised here.
An electrode through which the electric current flows out of the electrolytic solution is called the cathode, also called the negative electrode, and cations are reduced there.
Electrolysis
Electrolysis is the process of chemical deposition of the electrolyte by passing an electric current. Electrolysis takes place in an electrolytic cell. This cell will convert the electrical energy to chemical energy.
Basic of Corrosion

Corrosion is the deterioration of materials through chemical reactions with their environment. It occurs via electrochemical processes where the metal surface acts as an anode and electrons flow to a cathode. Factors like material properties, environment, temperature affect corrosion. Corrosion causes economic losses and safety issues. Understanding corrosion mechanisms and factors is important for material selection, design, and prevention of failures in engineering structures.
New chm-152-unit-9-power-points-sp13-140227172048-phpapp02

This document discusses electrolysis and Faraday's law of electrolysis. It begins by introducing electrolytic cells and predicting the products of electrolysis. It then explains that according to Faraday's law, the amount of substance produced at each electrode is directly proportional to the quantity of electricity passing through the cell. The document provides an example calculation applying Faraday's law to determine the current needed in an electrolysis process. It also discusses some key aspects of Faraday's law, such as defining the faraday as one mole of electrons.
Module 2_S7 and S8_Electrochemical Cells.pptx

An electrochemical cell consists of two electrodes separated by an electrolyte. There are two types: galvanic cells and electrolytic cells. A galvanic cell converts the chemical energy of a spontaneous redox reaction directly into electrical energy. The Nernst equation relates the cell potential (E) of a galvanic cell to the standard potential (E0), temperature, and reaction quotient through the concentrations of reactants and products. It allows calculation of cell potential under non-standard conditions.
B.tech. ii engineering chemistry unit 5 A electrochemistry

Arrhenius proposed the theory of electrolytic dissociation to explain the properties of electrolytic solutions. The theory states that when an electrolyte dissolves in water, it breaks up into ions - positively charged cations and negatively charged anions. This process is called ionization. Ions are constantly recombining and dissociating, reaching a state of dynamic equilibrium. The extent of ionization depends on an equilibrium constant. Strong electrolytes have a high equilibrium constant and ionize completely, while weak electrolytes have a low constant and only partially ionize.
Chapter - 6 (Electrochemistry).ppt

Electroanalytical methods provide several advantages for quantitative analytical chemistry. They involve measuring the electrical properties of analyte solutions in electrochemical cells. Some key points:
- Electroanalytical methods allow easy automation through electrical signal measurements. They can also determine low analyte concentrations without difficulty.
- Electrochemical processes involve the transfer of electrons between substances during redox reactions. This occurs at the interface between electrodes and solutions in electrochemical cells.
- Advantages include low cost compared to spectroscopy and the ability to easily automate measurements and detect low analyte concentrations through electrical signals.
Similar to lecture37.pdf (18)
3rd Lecture on Electrochemistry | Chemistry Part I | 12th Std

3rd Lecture on Electrochemistry | Chemistry Part I | 12th Std
electrochemistry-141128223112-conversion-gate02.pptx

electrochemistry-141128223112-conversion-gate02.pptx
New chm-152-unit-9-power-points-sp13-140227172048-phpapp02

New chm-152-unit-9-power-points-sp13-140227172048-phpapp02
B.tech. ii engineering chemistry unit 5 A electrochemistry

B.tech. ii engineering chemistry unit 5 A electrochemistry
Recently uploaded
Large Language Model (LLM) and it’s Geospatial Applications

Large Language Model (LLM) and it’s Geospatial Applications.
Artificial Intelligence for XMLDevelopment

In the rapidly evolving landscape of technologies, XML continues to play a vital role in structuring, storing, and transporting data across diverse systems. The recent advancements in artificial intelligence (AI) present new methodologies for enhancing XML development workflows, introducing efficiency, automation, and intelligent capabilities. This presentation will outline the scope and perspective of utilizing AI in XML development. The potential benefits and the possible pitfalls will be highlighted, providing a balanced view of the subject.
We will explore the capabilities of AI in understanding XML markup languages and autonomously creating structured XML content. Additionally, we will examine the capacity of AI to enrich plain text with appropriate XML markup. Practical examples and methodological guidelines will be provided to elucidate how AI can be effectively prompted to interpret and generate accurate XML markup.
Further emphasis will be placed on the role of AI in developing XSLT, or schemas such as XSD and Schematron. We will address the techniques and strategies adopted to create prompts for generating code, explaining code, or refactoring the code, and the results achieved.
The discussion will extend to how AI can be used to transform XML content. In particular, the focus will be on the use of AI XPath extension functions in XSLT, Schematron, Schematron Quick Fixes, or for XML content refactoring.
The presentation aims to deliver a comprehensive overview of AI usage in XML development, providing attendees with the necessary knowledge to make informed decisions. Whether you’re at the early stages of adopting AI or considering integrating it in advanced XML development, this presentation will cover all levels of expertise.
By highlighting the potential advantages and challenges of integrating AI with XML development tools and languages, the presentation seeks to inspire thoughtful conversation around the future of XML development. We’ll not only delve into the technical aspects of AI-powered XML development but also discuss practical implications and possible future directions.
How to Get CNIC Information System with Paksim Ga.pptx

Pakdata Cf is a groundbreaking system designed to streamline and facilitate access to CNIC information. This innovative platform leverages advanced technology to provide users with efficient and secure access to their CNIC details.
Building RAG with self-deployed Milvus vector database and Snowpark Container...

This talk will give hands-on advice on building RAG applications with an open-source Milvus database deployed as a docker container. We will also introduce the integration of Milvus with Snowpark Container Services.
UiPath Test Automation using UiPath Test Suite series, part 6

Welcome to UiPath Test Automation using UiPath Test Suite series part 6. In this session, we will cover Test Automation with generative AI and Open AI.
UiPath Test Automation with generative AI and Open AI webinar offers an in-depth exploration of leveraging cutting-edge technologies for test automation within the UiPath platform. Attendees will delve into the integration of generative AI, a test automation solution, with Open AI advanced natural language processing capabilities.
Throughout the session, participants will discover how this synergy empowers testers to automate repetitive tasks, enhance testing accuracy, and expedite the software testing life cycle. Topics covered include the seamless integration process, practical use cases, and the benefits of harnessing AI-driven automation for UiPath testing initiatives. By attending this webinar, testers, and automation professionals can gain valuable insights into harnessing the power of AI to optimize their test automation workflows within the UiPath ecosystem, ultimately driving efficiency and quality in software development processes.
What will you get from this session?
1. Insights into integrating generative AI.
2. Understanding how this integration enhances test automation within the UiPath platform
3. Practical demonstrations
4. Exploration of real-world use cases illustrating the benefits of AI-driven test automation for UiPath
Topics covered:
What is generative AI
Test Automation with generative AI and Open AI.
UiPath integration with generative AI
Speaker:
Deepak Rai, Automation Practice Lead, Boundaryless Group and UiPath MVP
20240607 QFM018 Elixir Reading List May 2024

Everything I found interesting about the Elixir programming ecosystem in May 2024
みなさんこんにちはこれ何文字まで入るの?40文字以下不可とか本当に意味わからないけどこれ限界文字数書いてないからマジでやばい文字数いけるんじゃないの?えこ...

ここ3000字までしか入らないけどタイトルの方がたくさん文字入ると思います。
20 Comprehensive Checklist of Designing and Developing a Website

Dive into the world of Website Designing and Developing with Pixlogix! Looking to create a stunning online presence? Look no further! Our comprehensive checklist covers everything you need to know to craft a website that stands out. From user-friendly design to seamless functionality, we've got you covered. Don't miss out on this invaluable resource! Check out our checklist now at Pixlogix and start your journey towards a captivating online presence today.
Enchancing adoption of Open Source Libraries. A case study on Albumentations.AI

Enchancing adoption of Open Source Libraries. A case study on Albumentations.AIVladimir Iglovikov, Ph.D.
Presented by Vladimir Iglovikov:
- https://www.linkedin.com/in/iglovikov/
- https://x.com/viglovikov
- https://www.instagram.com/ternaus/
This presentation delves into the journey of Albumentations.ai, a highly successful open-source library for data augmentation.
Created out of a necessity for superior performance in Kaggle competitions, Albumentations has grown to become a widely used tool among data scientists and machine learning practitioners.
This case study covers various aspects, including:
People: The contributors and community that have supported Albumentations.
Metrics: The success indicators such as downloads, daily active users, GitHub stars, and financial contributions.
Challenges: The hurdles in monetizing open-source projects and measuring user engagement.
Development Practices: Best practices for creating, maintaining, and scaling open-source libraries, including code hygiene, CI/CD, and fast iteration.
Community Building: Strategies for making adoption easy, iterating quickly, and fostering a vibrant, engaged community.
Marketing: Both online and offline marketing tactics, focusing on real, impactful interactions and collaborations.
Mental Health: Maintaining balance and not feeling pressured by user demands.
Key insights include the importance of automation, making the adoption process seamless, and leveraging offline interactions for marketing. The presentation also emphasizes the need for continuous small improvements and building a friendly, inclusive community that contributes to the project's growth.
Vladimir Iglovikov brings his extensive experience as a Kaggle Grandmaster, ex-Staff ML Engineer at Lyft, sharing valuable lessons and practical advice for anyone looking to enhance the adoption of their open-source projects.
Explore more about Albumentations and join the community at:
GitHub: https://github.com/albumentations-team/albumentations
Website: https://albumentations.ai/
LinkedIn: https://www.linkedin.com/company/100504475
Twitter: https://x.com/albumentationsIntroducing Milvus Lite: Easy-to-Install, Easy-to-Use vector database for you...

Join us to introduce Milvus Lite, a vector database that can run on notebooks and laptops, share the same API with Milvus, and integrate with every popular GenAI framework. This webinar is perfect for developers seeking easy-to-use, well-integrated vector databases for their GenAI apps.
GraphSummit Singapore | The Future of Agility: Supercharging Digital Transfor...

Leonard Jayamohan, Partner & Generative AI Lead, Deloitte
This keynote will reveal how Deloitte leverages Neo4j’s graph power for groundbreaking digital twin solutions, achieving a staggering 100x performance boost. Discover the essential role knowledge graphs play in successful generative AI implementations. Plus, get an exclusive look at an innovative Neo4j + Generative AI solution Deloitte is developing in-house.
TrustArc Webinar - 2024 Global Privacy Survey

How does your privacy program stack up against your peers? What challenges are privacy teams tackling and prioritizing in 2024?
In the fifth annual Global Privacy Benchmarks Survey, we asked over 1,800 global privacy professionals and business executives to share their perspectives on the current state of privacy inside and outside of their organizations. This year’s report focused on emerging areas of importance for privacy and compliance professionals, including considerations and implications of Artificial Intelligence (AI) technologies, building brand trust, and different approaches for achieving higher privacy competence scores.
See how organizational priorities and strategic approaches to data security and privacy are evolving around the globe.
This webinar will review:
- The top 10 privacy insights from the fifth annual Global Privacy Benchmarks Survey
- The top challenges for privacy leaders, practitioners, and organizations in 2024
- Key themes to consider in developing and maintaining your privacy program
Essentials of Automations: The Art of Triggers and Actions in FME

In this second installment of our Essentials of Automations webinar series, we’ll explore the landscape of triggers and actions, guiding you through the nuances of authoring and adapting workspaces for seamless automations. Gain an understanding of the full spectrum of triggers and actions available in FME, empowering you to enhance your workspaces for efficient automation.
We’ll kick things off by showcasing the most commonly used event-based triggers, introducing you to various automation workflows like manual triggers, schedules, directory watchers, and more. Plus, see how these elements play out in real scenarios.
Whether you’re tweaking your current setup or building from the ground up, this session will arm you with the tools and insights needed to transform your FME usage into a powerhouse of productivity. Join us to discover effective strategies that simplify complex processes, enhancing your productivity and transforming your data management practices with FME. Let’s turn complexity into clarity and make your workspaces work wonders!
Let's Integrate MuleSoft RPA, COMPOSER, APM with AWS IDP along with Slack

Discover the seamless integration of RPA (Robotic Process Automation), COMPOSER, and APM with AWS IDP enhanced with Slack notifications. Explore how these technologies converge to streamline workflows, optimize performance, and ensure secure access, all while leveraging the power of AWS IDP and real-time communication via Slack notifications.
Cosa hanno in comune un mattoncino Lego e la backdoor XZ?

ABSTRACT: A prima vista, un mattoncino Lego e la backdoor XZ potrebbero avere in comune il fatto di essere entrambi blocchi di costruzione, o dipendenze di progetti creativi e software. La realtà è che un mattoncino Lego e il caso della backdoor XZ hanno molto di più di tutto ciò in comune.
Partecipate alla presentazione per immergervi in una storia di interoperabilità, standard e formati aperti, per poi discutere del ruolo importante che i contributori hanno in una comunità open source sostenibile.
BIO: Sostenitrice del software libero e dei formati standard e aperti. È stata un membro attivo dei progetti Fedora e openSUSE e ha co-fondato l'Associazione LibreItalia dove è stata coinvolta in diversi eventi, migrazioni e formazione relativi a LibreOffice. In precedenza ha lavorato a migrazioni e corsi di formazione su LibreOffice per diverse amministrazioni pubbliche e privati. Da gennaio 2020 lavora in SUSE come Software Release Engineer per Uyuni e SUSE Manager e quando non segue la sua passione per i computer e per Geeko coltiva la sua curiosità per l'astronomia (da cui deriva il suo nickname deneb_alpha).
Uni Systems Copilot event_05062024_C.Vlachos.pdf

Unlocking Productivity: Leveraging the Potential of Copilot in Microsoft 365, a presentation by Christoforos Vlachos, Senior Solutions Manager – Modern Workplace, Uni Systems
Observability Concepts EVERY Developer Should Know -- DeveloperWeek Europe.pdf

Monitoring and observability aren’t traditionally found in software curriculums and many of us cobble this knowledge together from whatever vendor or ecosystem we were first introduced to and whatever is a part of your current company’s observability stack.
While the dev and ops silo continues to crumble….many organizations still relegate monitoring & observability as the purview of ops, infra and SRE teams. This is a mistake - achieving a highly observable system requires collaboration up and down the stack.
I, a former op, would like to extend an invitation to all application developers to join the observability party will share these foundational concepts to build on:
Recently uploaded (20)
Large Language Model (LLM) and it’s Geospatial Applications

Large Language Model (LLM) and it’s Geospatial Applications
How to Get CNIC Information System with Paksim Ga.pptx

How to Get CNIC Information System with Paksim Ga.pptx
Building RAG with self-deployed Milvus vector database and Snowpark Container...

Building RAG with self-deployed Milvus vector database and Snowpark Container...
UiPath Test Automation using UiPath Test Suite series, part 6

UiPath Test Automation using UiPath Test Suite series, part 6
みなさんこんにちはこれ何文字まで入るの?40文字以下不可とか本当に意味わからないけどこれ限界文字数書いてないからマジでやばい文字数いけるんじゃないの?えこ...

みなさんこんにちはこれ何文字まで入るの?40文字以下不可とか本当に意味わからないけどこれ限界文字数書いてないからマジでやばい文字数いけるんじゃないの?えこ...
20 Comprehensive Checklist of Designing and Developing a Website

20 Comprehensive Checklist of Designing and Developing a Website
Enchancing adoption of Open Source Libraries. A case study on Albumentations.AI

Enchancing adoption of Open Source Libraries. A case study on Albumentations.AI
Introducing Milvus Lite: Easy-to-Install, Easy-to-Use vector database for you...

Introducing Milvus Lite: Easy-to-Install, Easy-to-Use vector database for you...
GraphSummit Singapore | The Future of Agility: Supercharging Digital Transfor...

GraphSummit Singapore | The Future of Agility: Supercharging Digital Transfor...
Essentials of Automations: The Art of Triggers and Actions in FME

Essentials of Automations: The Art of Triggers and Actions in FME
Let's Integrate MuleSoft RPA, COMPOSER, APM with AWS IDP along with Slack

Let's Integrate MuleSoft RPA, COMPOSER, APM with AWS IDP along with Slack
Cosa hanno in comune un mattoncino Lego e la backdoor XZ?

Cosa hanno in comune un mattoncino Lego e la backdoor XZ?
Monitoring Java Application Security with JDK Tools and JFR Events

Monitoring Java Application Security with JDK Tools and JFR Events
Observability Concepts EVERY Developer Should Know -- DeveloperWeek Europe.pdf

Observability Concepts EVERY Developer Should Know -- DeveloperWeek Europe.pdf
lecture37.pdf
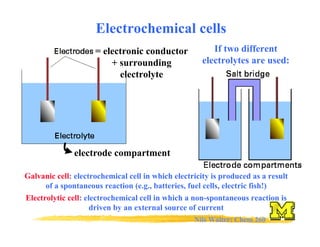
- 1. Nils Walter: Chem 260 Electrochemical cells = electronic conductor + surrounding electrolyte electrode compartment If two different electrolytes are used: Electrolytic cell: electrochemical cell in which a non-spontaneous reaction is driven by an external source of current Galvanic cell: electrochemical cell in which electricity is produced as a result of a spontaneous reaction (e.g., batteries, fuel cells, electric fish!)
- 2. Nils Walter: Chem 260 Reactions at electrodes: Half-reactions Redox reactions: Reactions in which electrons are transferred from one species to another +II -II 0 -II 0 +IV reduced oxidized Any redox reactions can be expressed as the difference between two reduction half-reactions in which e- are taken up CuS(s) + O2(g) → → → → Cu(s) + SO2(g) E.g., Reduction of Cu2+: Cu2+(aq) + 2e- → → → → Cu(s) Reduction of Zn2+: Zn2+(aq) + 2e- → → → → Zn(s) Cu2+(aq) + Zn(s) → → → → Cu(s) + Zn2+(aq) More complex: MnO4 -(aq) + 8H+ + 5e- → → → → Mn2+(aq) + 4H2O(l) Half-reactions are only a formal way of writing a redox reaction Difference:
- 3. Nils Walter: Chem 260 Carrying the concept further Reduction of Cu2+: Cu2+(aq) + 2e- → → → → Cu(s) In general: redox couple Ox/Red, half-reaction Ox + ν ν ν νe- → → → → Red Any reaction can be expressed in redox half-reactions: Expansion of gas: H2(g, pi) → → → → H2(g, pf) 2 H+(aq) + 2e- → → → → H2(g, pi) 2 H+(aq) + 2e- → → → → H2(g, pf) Dissolution of a sparingly soluble salt: AgCl(s) → → → → Ag+(aq) + Cl-(aq) Ag+(aq) + e- → → → → Ag(s) AgCl(s) + e- → → → → Ag(s) + Cl-(aq) Reaction quotients: ] [ − ≈ = − Cl a Q Cl ] [ 1 1 + ≈ = + Ag a Q Ag
- 4. Nils Walter: Chem 260 Reactions at electrodes Galvanic cell: Electrolytic cell: Red1 → → → → Ox1 + ν ν ν νe- Ox2 + ν ν ν νe- → → → → Red2 Half-reactions Red1 → → → → Ox1 + ν ν ν νe- Ox2 + ν ν ν νe- → → → → Red2
- 5. Nils Walter: Chem 260 Insoluble-salt electrode: metal (e.g., Pt) solution (e.g., H+) 2H+ + 2e- H2(g) 2 2 ) ( + = H a H p Q Types of electrodes I Pt(s)| || | H2(g)| || | H+(aq) Gas electrode: Gas (e.g., H2) metal (e.g., Ag) solution (e.g., Cl-) AgCl(s) + e- Ag(s) + Cl-(aq) − = Cl a Q Ag(s)| || | AgCl(s)| || | Cl-(aq) Porous, insoluble salt (e.g., AgCl)
- 6. Nils Walter: Chem 260 Fe3+ + e- Fe2+ + + = = 3 2 Re Fe Fe Ox d a a a a Q Types of electrodes and how to put them together in a galvanic cell Pt(s)| || | Fe2+(aq),Fe3+(aq) + + = 2 2 Cu Zn a a Q Zn(s)| || | ZnSO4(aq) | || | | || | CuSO4(aq)| || | Cu(s) Redox electrode: Daniell cell: Right: Cu2+(aq) + 2e- → → → → Cu(s) Left: Zn2+(aq) + 2e- → → → → Zn(s) Cu2+(aq) + Zn(s) Cu(s) + Zn2+(aq)
- 7. Nils Walter: Chem 260 Cell reaction and potential Cell reaction: Difference of electrode half-reactions (Reduction at Cathode - Oxidation at Anode) Cathode (Right): Cu2+(aq) + 2e- → → → → Cu(s) Anode (Left): Zn2+(aq) + 2e- → → → → Zn(s) Overall (R-L): Cu2+(aq) + Zn(s) Cu(s) + Zn2+(aq) e- disappear this way the reaction becomes spontaneous } Cell potential E: Potential difference between the electrodes Maximum electrical work done in a galvanic cell: w’ = - ν ν ν νF × × × × E = ∆ ∆ ∆ ∆rG Ox + ν ν ν νe- → → → → Red Þ Þ Þ Þ ν ν ν νNA e- transferred per mole of reaction Þ Þ Þ Þ ν ν ν νNA × × × × (-e) = -ν ν ν νF charge transferred Faraday constant = eNA = 96,485 Cmol-1