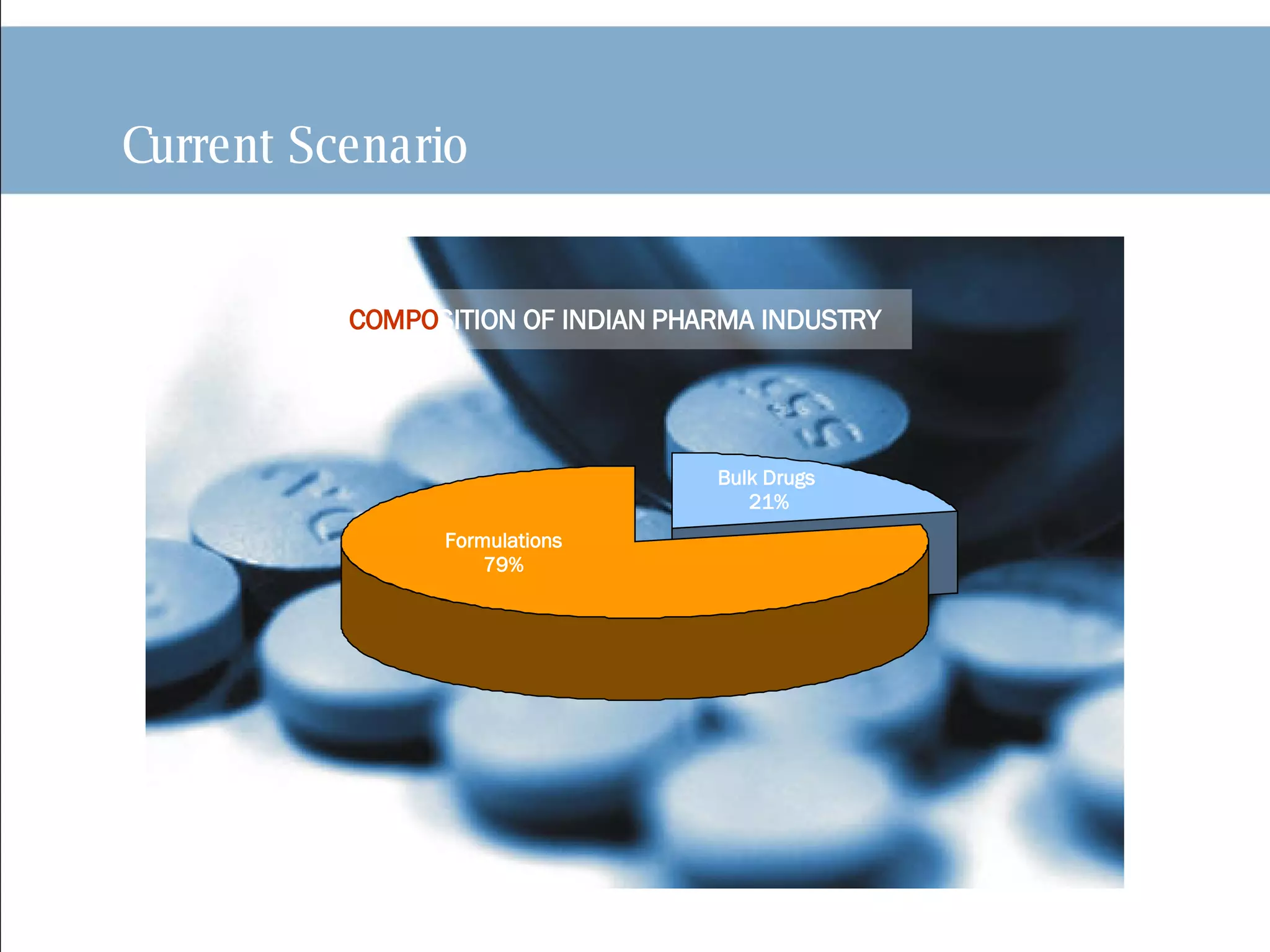
The document discusses India's pharmaceutical industry, including its current size and growth rate. It notes that formulations make up 79% of the industry while bulk drugs account for 21%. The background section outlines how India's Patents Act of 1970 only recognized process patents and led to increased domestic production. It also discusses how India transitioned to a product patent regime in 2005 to comply with TRIPS and how this has impacted multinational companies investing in India.