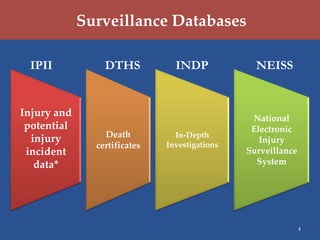
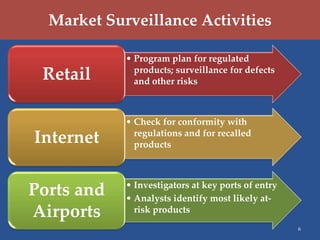
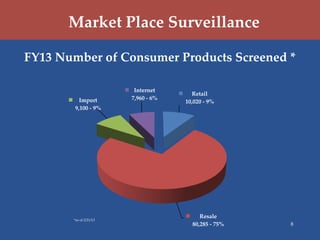
The U.S. Consumer Product Safety Commission (CPSC) focuses on preventing injuries and deaths through hazard identification, market surveillance, and compliance with voluntary safety standards. Key strategic goals include improving hazard data quality, reducing identification time for hazard trends, enhancing import surveillance, and increasing scrutiny of used consumer products. The CPSC utilizes various surveillance databases and monitors products through investigations, particularly at major U.S. ports.