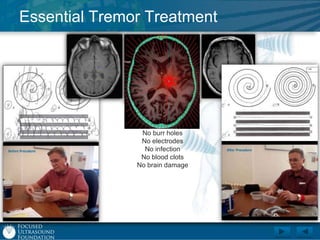
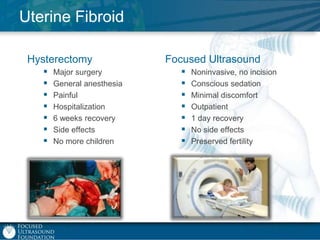
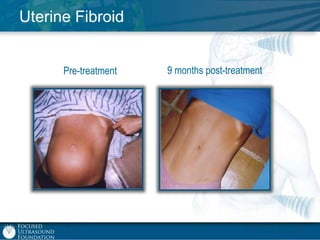
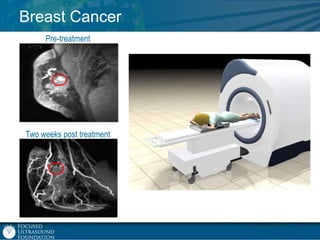
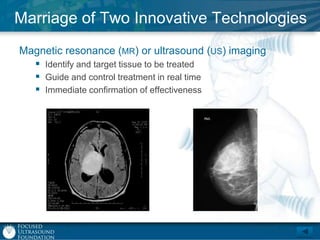
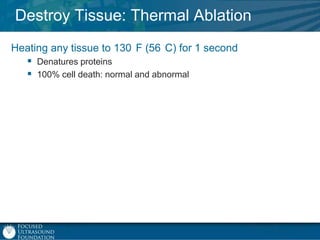
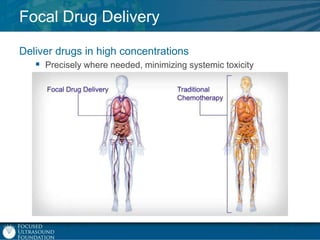
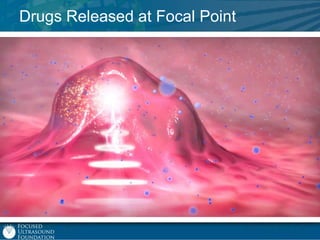
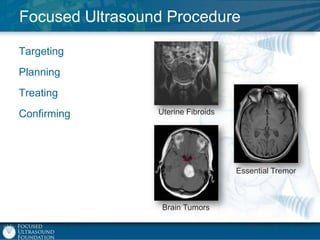
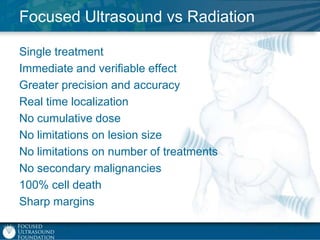
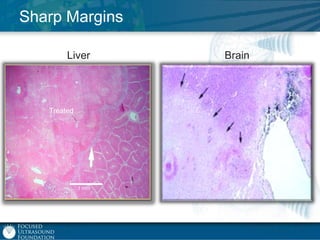
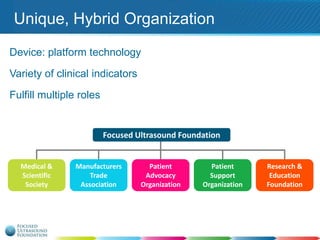
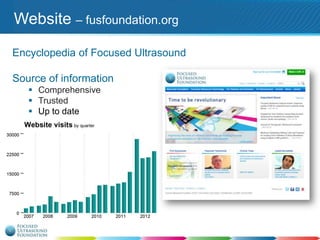
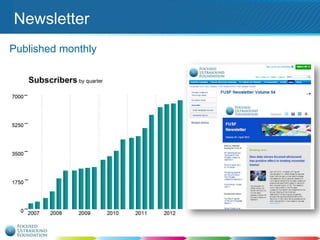
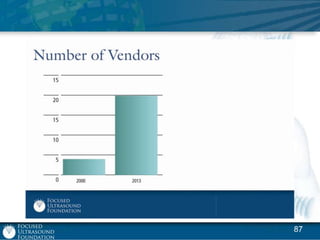
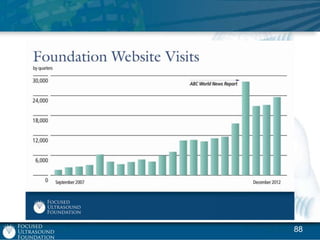
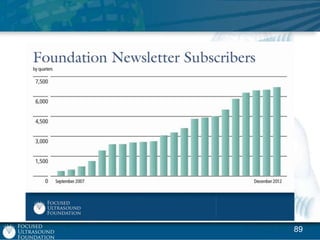
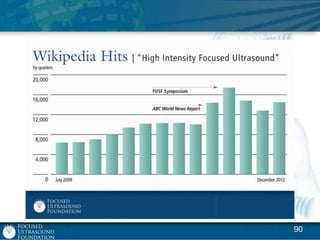
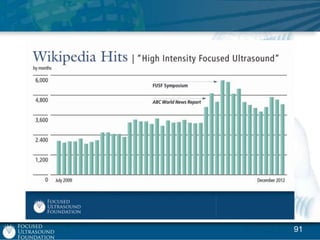
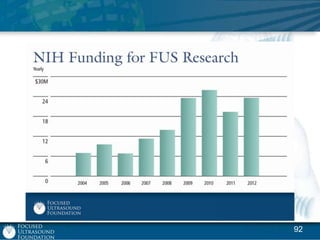
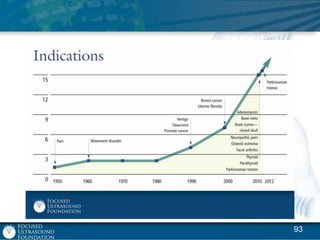
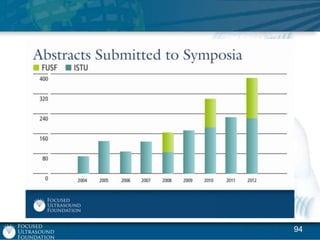
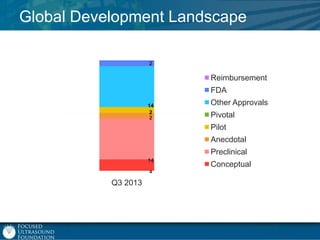
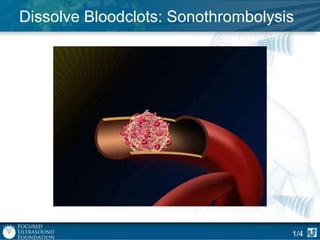
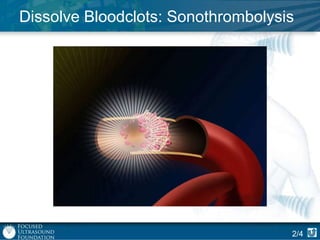
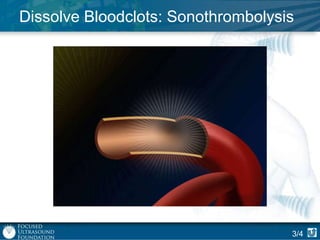
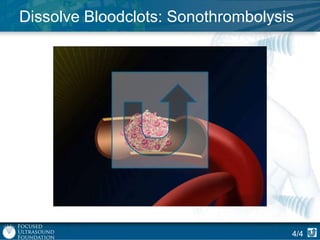
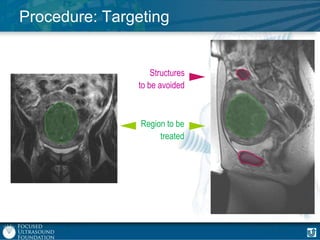
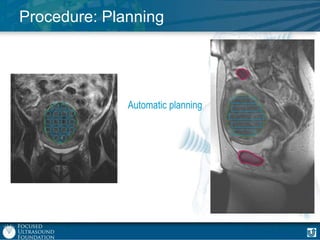
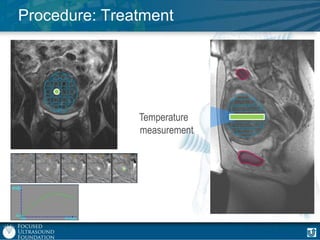
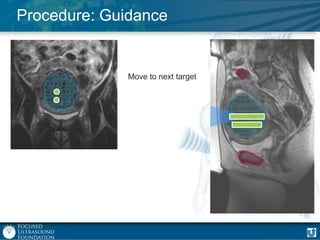
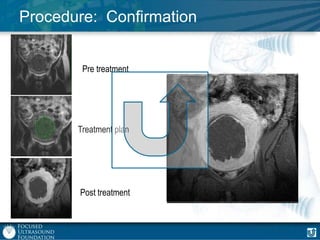
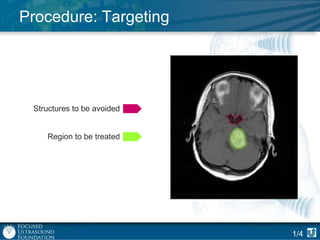
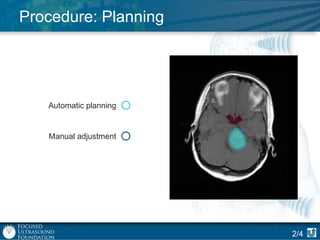
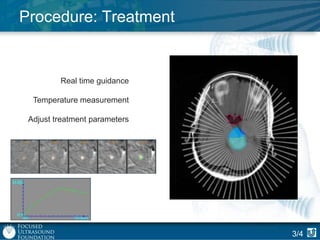
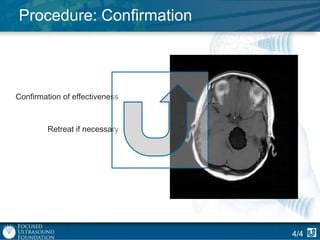
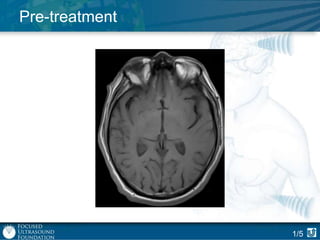
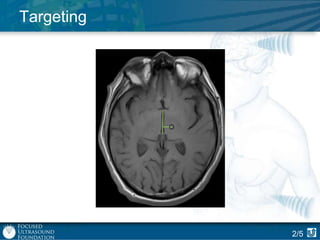
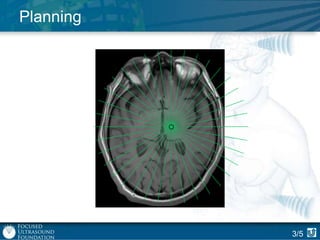
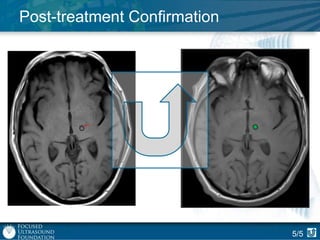
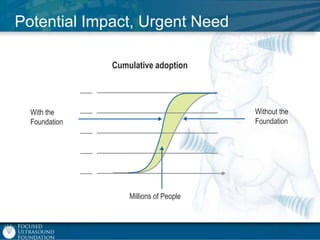
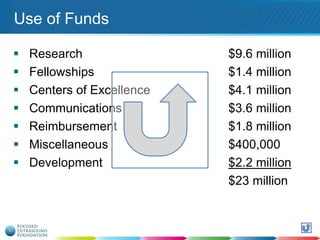
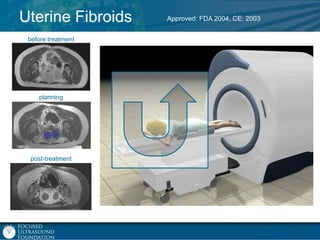
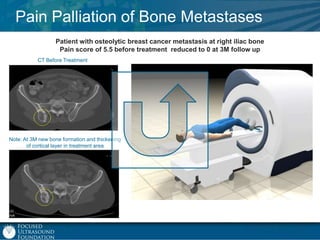
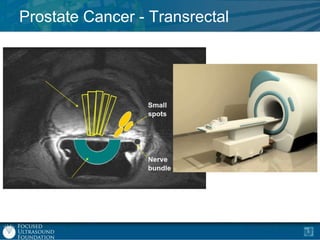
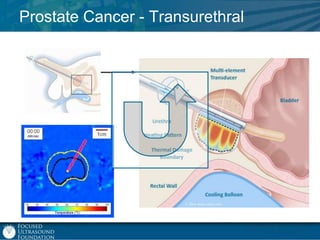
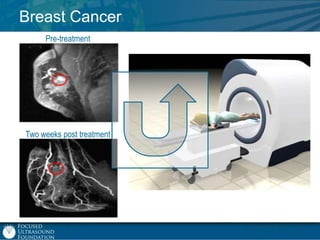
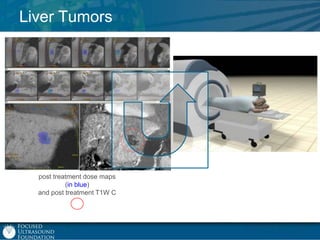
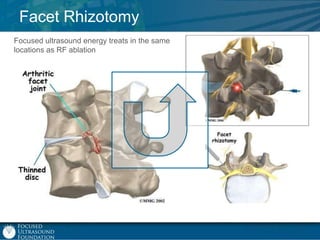
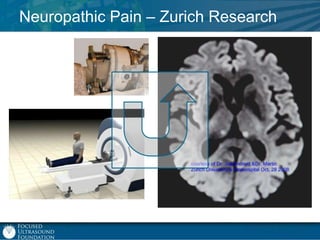
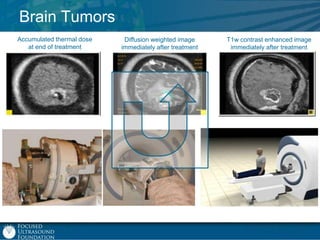
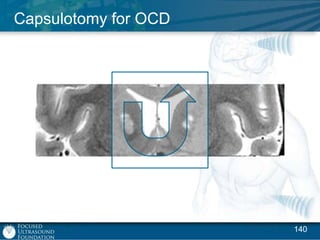
This document provides an overview of focused ultrasound technology. It discusses how focused ultrasound is a non-invasive therapeutic technology that can be used as an alternative or adjunct to surgery, radiation therapy, and drug delivery to treat conditions like tumors, metastatic cancer, Parkinson's disease, and back pain. It explains that focused ultrasound uses intersecting beams of ultrasound focused with precision to target and treat tissue while avoiding damage to surrounding areas. The document outlines the various effects it can have on tissue and discusses its applications and development. It positions focused ultrasound as a technology that could improve quality of life for millions of patients worldwide.