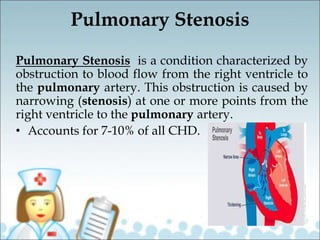
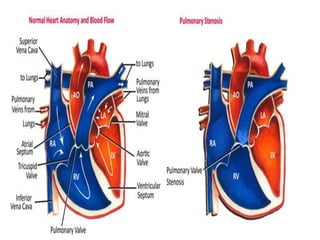
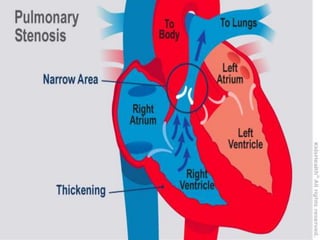
This document provides information on congenital heart disease (CHD), including:
- CHD occurs during the first 8 weeks of fetal development in approximately 5-8 per 1000 live births.
- Causes are usually multifactorial involving both genetic and environmental factors.
- Diagnosis involves physical examination, echocardiogram, chest x-ray, fetal echocardiography and cardiac catheterization.
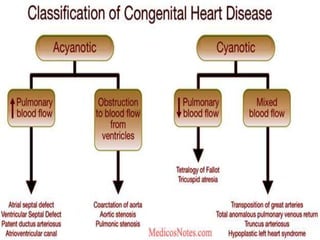
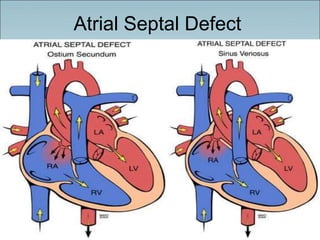
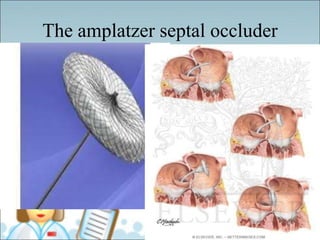
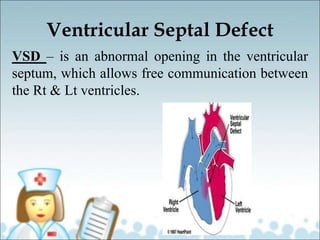
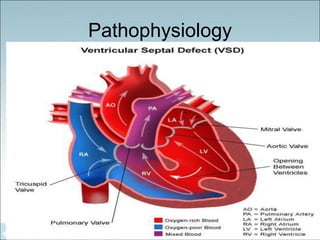
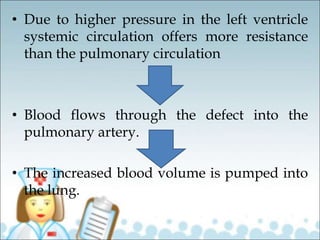
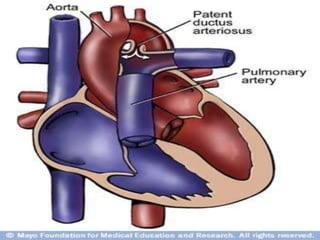
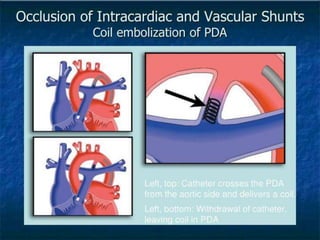
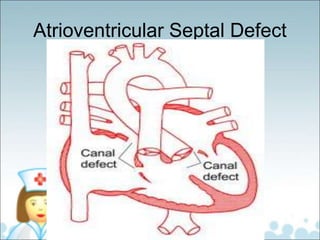
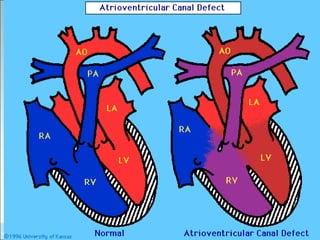
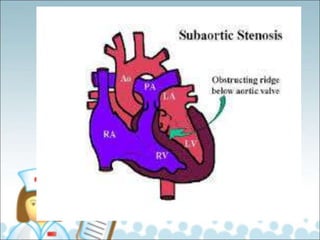
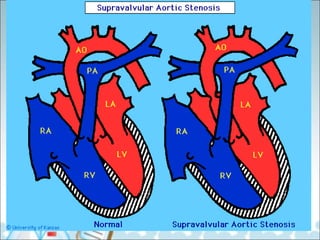
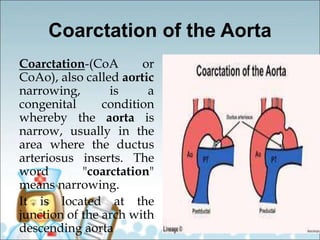
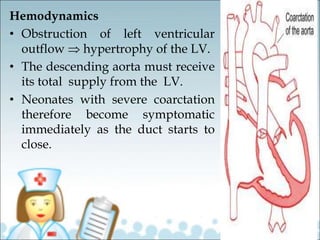
- Common types of CHD discussed include atrial septal defect (ASD), ventricular septal defect (VSD), patent ductus arteriosus (PDA), and atrioventricular septal defect (AVSD). Signs, symptoms, pathophysiology, and treatment are described for each