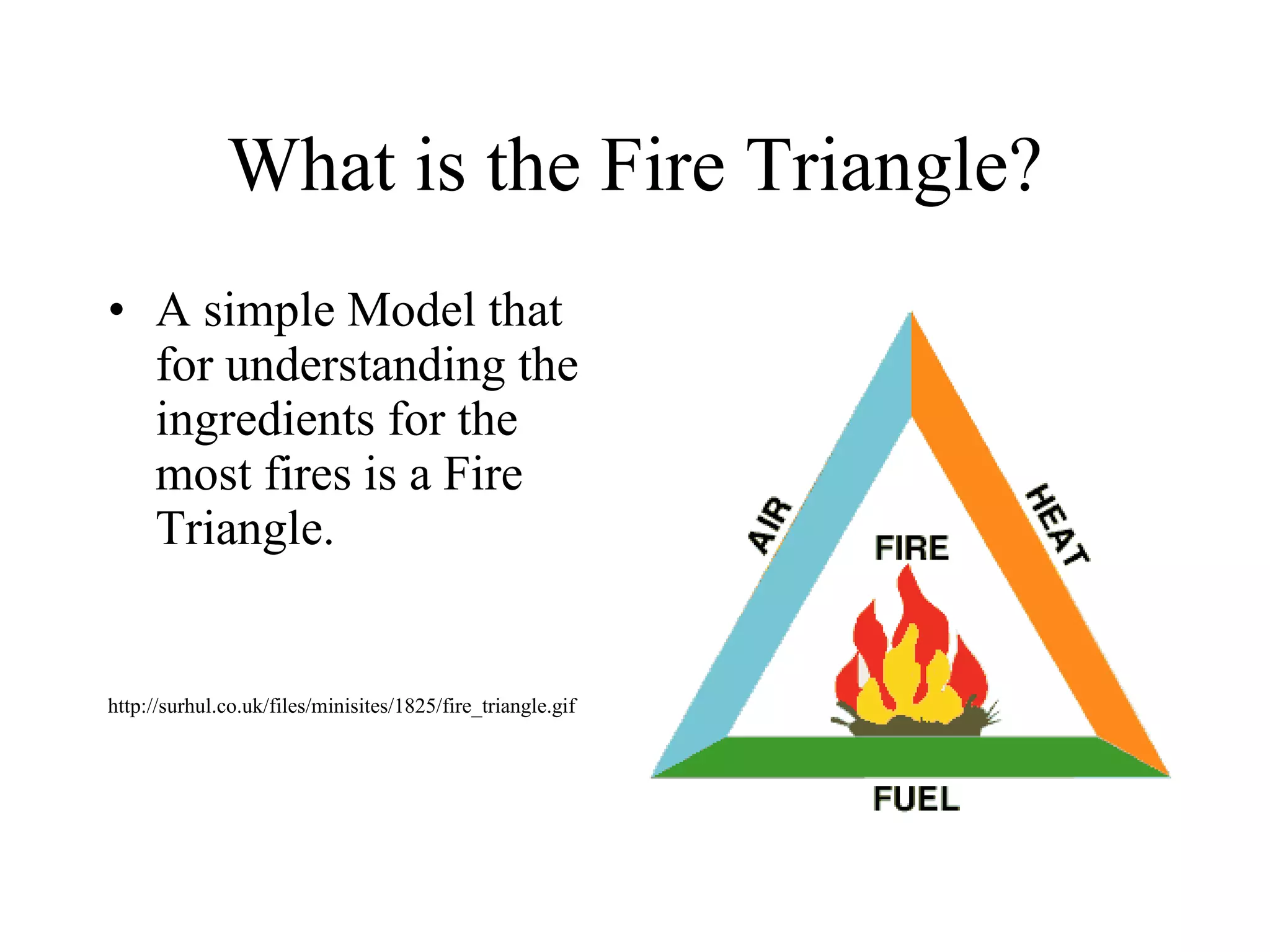
The document discusses combustion reactions and provides examples of combustion in engines and on stoves. It defines combustion as a reaction between oxygen and another compound that forms water and carbon oxide. The fire triangle is introduced as needing oxygen, fuel, and heat for fire to occur. Activation energy is the minimum energy needed for a chemical reaction. Heat is necessary to start and sustain fire according to the fire triangle model. A stove burner is given as a real-life example of the fire triangle, with the gas as fuel and a spark igniting it. A combustion engine is defined as using combustion in a chamber to directly power a movable component, and nitrous oxide helps engines by providing more oxygen for combustion.