Embed presentation

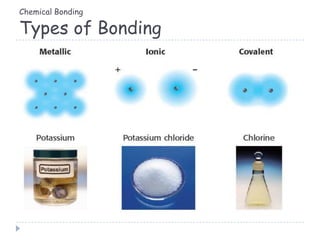

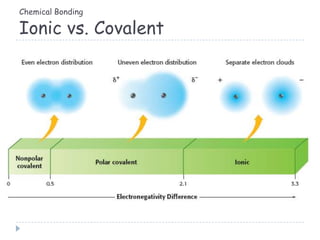
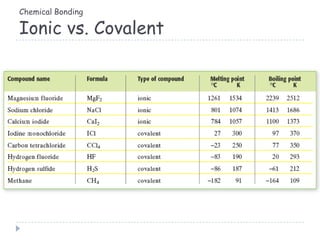
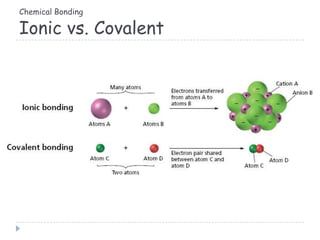
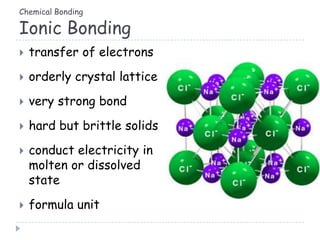
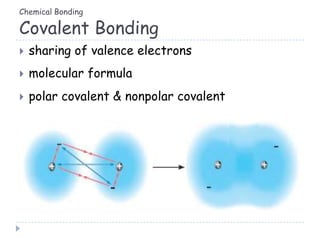

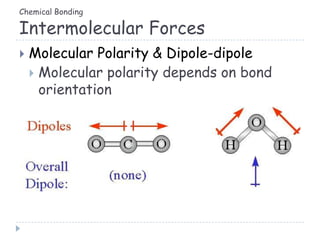
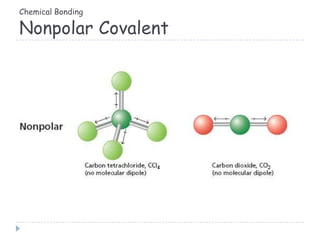
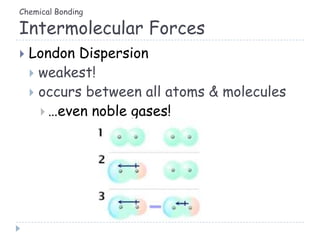
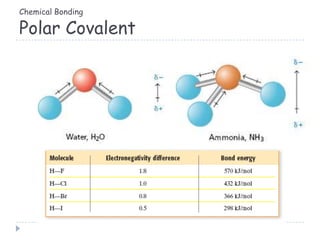
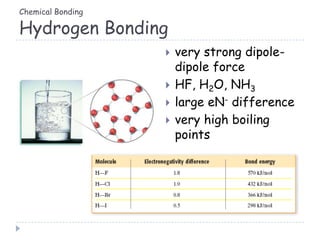

The document discusses different types of chemical bonding including metallic bonding, ionic bonding, and covalent bonding. Metallic bonding is described as involving delocalized electrons in a "sea of electrons" that provides properties like luster, malleability, and ductility. Ionic bonding involves the transfer of electrons and results in orderly crystal lattices and very strong bonds, forming hard but brittle solids. Covalent bonding involves the sharing of valence electrons and can be polar or nonpolar. Intermolecular forces like dipole-dipole interactions and London dispersion are also discussed.













