BSI poster
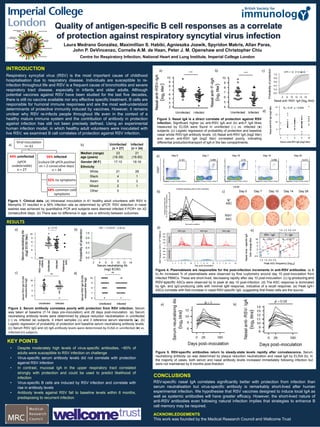
- 1. Quality of antigen-specific B cell responses as a correlate of protection against respiratory syncytial virus infection Laura Medrano González, Maximillian S. Habibi, Agnieszka Jozwik, Spyridon Makris, Allan Paras, John P. DeVincenzo, Cornelis A.M. de Haan, Peter J. M. Openshaw and Christopher Chiu Centre for Respiratory Infection, National Heart and Lung Institute, Imperial College London Respiratory syncytial virus (RSV) is the most important cause of childhood hospitalisation due to respiratory disease. Individuals are susceptible to re- infection throughout life and RSV is a frequent cause of bronchiolitis and severe respiratory tract disease, especially in infants and older adults. Although potential vaccines against RSV have been studied for the last five decades, there is still no vaccine available nor any effective specific treatment. B cells are responsible for humoral immune responses and are the most well-understood determinants of protective immunity induced by vaccines. However, it remains unclear why RSV re-infects people throughout life even in the context of a healthy mature immune system and the contribution of antibody in protection against infection has still not been precisely defined. Using an experimental human infection model, in which healthy adult volunteers were inoculated with live RSV, we examined B cell correlates of protection against RSV infection. INTRODUCTION RESULTS RSV-specific nasal IgA correlates significantly better with protection from infection than serum neutralisation but virus-specific antibody is remarkably short-lived after human experimental infection. We hypothesise that RSV vaccines designed to induce local IgA as well as systemic antibodies will have greater efficacy. However, the short-lived nature of anti-RSV antibodies even following natural infection implies that strategies to enhance B cell memory may be required. CONCLUSIONS Figure 5. RSV-specific antibodies return to steady-state levels rapidly after convalescence. Serum neutralizing antibody (a) was determined by plaque reduction neutralization and nasal IgA by ELISA (b). In the majority of cases, both serum and nasal antibody levels increased immediately following infection but were not maintained by 6 months post-finection Viral inocula+on n= 61 44% uninfected (qPCR undetectable) n = 27 56% infected (culture OR qPCR posi+ve on > 2 consecu+ve days) n = 34 32% no symptoms 68% common cold symptoms Figure 1. Clinical data. (a) Intranasal inoculation in 61 healthy adult volunteers with RSV A Memphis 37 resulted in a 56% infection rate as determined by qPCR. RSV detection in nasal washes was achieved by quantitative PCR and subjects were deemed infected if PCR+ on ≥2 consecutive days. (b) There was no difference in age, sex or ethnicity between outcomes. Antibody-secreting cells by flow cytometry 0 7 14 21 28 0 2 4 6 8 Day post-inoculation Antibodysecretingcells (%ofCD3-CD20-lymphocytes) Day post-inoculation ASCfrequency(SFU/106 PBMCs) 0 200 400 600 800 0 3 7 10 14 28 Uninfected 0 3 7 10 14 28 0 3 7 10 14 28 0 200 400 600 800 RSV-IgG Infected RSV-IgA RSV-IgM RSV+ ASC Ig+ ASC Day 0 Day 7 Day 10 Day 14 Day 28 Figure 4. Plasmablasts are responsible for the post-infection increments in anti-RSV antibodies. (a & b) An increased % of plasmablasts were observed by flow cuytometry around day 10 post-inoculation from infected PBMCs. These are short-lived, decreasing rapidly after day 10 post-inoculation. (c) Ig-producing and RSV-specific ASCs were observed by to peak at day 10 post-infection. (d) The ASC response is dominated by IgA- and IgG-producing cells with minimal IgM response, indicative of a recall response. (e) Peak IgA+ ASCs correlate with fold-increase in nasal RSV-specific IgA, suggesting that these cells are the source. Uninfected (n = 27) Infected (n = 34) Median (range) age (years) 23 (18-39) 21 (18-50) Gender (M:F) 17:10 18:16 Ethnicity White 21 28 Black 4 1 Asian 0 2 Mixed 2 1 Other 0 KEY POINTS • Despite moderately high levels of virus-specific antibodies, ~60% of adults were susceptible to RSV infection on challenge • Virus-specific serum antibody levels did not correlate with protection against RSV infection • In contrast, mucosal IgA in the upper respiratory tract correlated strongly with protection and could be used to predict likelihood of infection • Virus-specific B cells are induced by RSV infection and correlate with rise in antibody levels • Antibody levels against RSV fall to baseline levels within 6 months, predisposing to recurrent infection ACKNOWLEDGEMENTS This work was founded by the Medical Research Council and Wellcome Trust a) c) b) d) a) b) c) D d) e) a) b) b) a) Figure 2. Serum antibody correlates poorly with protection from RSV infection. Serum was taken at baseline (7-14 days pre-inoculation) and 28 days post-inoculation. (a) Serum neutralising antibody levels were determined by plaque reduction neutralisation in uninfected (○) vs. infected (●) subjects, 4 infant samples (∆) and 3 reference serum standards (■). (b) Logistic regression of probability of protection and baseline serum neutralising antibody levels. (c) Serum RSV IgG and (d) IgA an+body levels were determined by ELISA in uninfected (●) vs. infected (○) subjects. 6 8 10 12 14 16 0.0 0.2 0.4 0.6 0.8 1.0 Nasal anti- RSV- IgA (log2 titre) Probabilityofprotection OR = 1.9 (1.2 œ3.4) * protec%on'to'serum'neutralizing'an%body' Figure 1: RSV-specific nasal IgA is a superior correlate of protection to serum neutralization titer. (a) Healthy adult volunteers were inoculated with RSV A M37 via nasal drops and followed up with blood and nasal sampling for 28 days. (b) RSV detection in nasal washes was achieved by quantitative PCR and subjects were deemed infected if PCR+ on ≥2 consecutive days. Common-cold symptoms were determined by a cumulative modified Jackson score. (c) Serum and nasal lavage were taken at baseline (7-14 days pre-inoculation) and 28 days post-inoculation. Serum neutralizing antibody was determined by plaque reduction neutralization and nasal IgA by ELISA. Baseline serum neutralising (top), nasal IgA against whole RSV (middle), and nasal anti-RSV fusion (F) protein (bottom) antibody levels in uninfected (○) vs. infected (●) subjects. Serum neutralization includes 4 infant samples (∆) and 3 reference serum standards (■ Wyeth 06937, 06938, 06939, (d) Logistic regression of probability of protection and baseline serum neutralising (top), nasal whole RSV-IgA (middle), and nasal RSV fusion (F) protein IgA (bottom) antibody levels. Predicted probability (solid line) and pointwise 95% confidence band (shaded) are shown. OR = odds ratio (and 95% confidence interval), P-values for unpaired Mann- Whitney-Wilcoxon (U) test or Chi-squared test are shown (ns = P > 0.05, * = P < 0.05, ** = P < 0.01, *** = P < 0.001). Complete'data'for'61'subjects'(1'subject'is'excluded' from'nasal'lavage'as'sample'was'too'dilute'to' measure'anything'(subject'AW'from'2012))' Figure 1: RSV-specific nasal IgA is a superior correlate of protection to serum neutralization titer. (a) Healthy a volunteers were inoculated with RSV A M37 via nasal drops and followed up with blood and nasal sampling for 28 day RSV detection in nasal washes was achieved by quantitative PCR and subjects were deemed infected if PCR+ on ≥2 consecutive days. Common-cold symptoms were determined by a cumulative modified Jackson score. (c) Serum and nasal lavage were taken at baseline (7-14 days pre-inoculation) and 28 days post-inoculation. Serum neutralizing ant was determined by plaque reduction neutralization and nasal IgA by ELISA. Baseline serum neutralising (top), nasal I against whole RSV (middle), and nasal anti-RSV fusion (F) protein (bottom) antibody levels in uninfected (○) vs. infec (●) subjects. Serum neutralization includes 4 infant samples (∆) and 3 reference serum standards (■ Wyeth 06937, 06 06939, (d) Logistic regression of probability of protection and baseline serum neutralising (top), nasal whole RSV-IgA (middle), and nasal RSV fusion (F) protein IgA (bottom) antibody levels. Predicted probability (solid line) and pointwis 95% confidence band (shaded) are shown. OR = odds ratio (and 95% confidence interval), P-values for unpaired Man Whitney-Wilcoxon (U) test or Chi-squared test are shown (ns = P > 0.05, * = P < 0.05, ** = P < 0.01, *** = P < 0.001). Complete'data'for'61'subjects'(1'subject'is'excluded' from'nasal'lavage'as'sample'was'too'dilute'to' measure'anything'(subject'AW'from'2012))' b) a) c) d) Figure 3. Nasal IgA is a direct correlate of protection against RSV infection. Significant higher (a) anti-RSV IgA and (b) anti-F IgA titres measured by ELISA were found in uninfected (○) vs. infected (●) subjects. (c) Logistic regression of probability of protection and baseline nasal whole RSV-IgA antibody levels. (d) Nasal anti-RSV IgA (log2 titer) and serum anti-RSV IgA (log2 titer) correlated poorly, indicating differential production/transport of IgA in the two compartments.