Autacoids.pptx
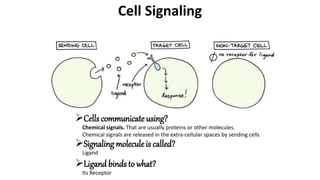
- 1. Cells communicate using? Chemical signals. That are usually proteins or other molecules. Chemical signals are released in the extra-cellular spaces by sending cells Cell Signaling Signaling molecule is called? Ligand Ligand binds to what? Its Receptor
- 2. When a ligandbinds to its receptor the receptor pass on a chain of chemical messengers in the cell. It leads to changes in the cell Gene expression. Or any other activity like cell division. Cell Signaling Thus, the INTERCELLULAR (between-cells) signal is convertedinto INTRACELLULAR (within- cell) signals.
- 3. Cell Signaling Forms of Signaling Not all sending and receiving cells are net-doorneighbors. Therefore, cell signaling is classified on the basis of distance that the signals travel to reachthe target. 1. Paracrine signaling 2. Synaptic signaling 3. Autocrine signaling 4. Endocrine signaling 5. Signaling through cell to cell contact
- 4. Cell Signaling 1. Paracrine Signaling In this type of signaling cells communicate over relatively short distance. Cells locally coordinate activities with their neighbors.
- 5. Cell Signaling 2. Synaptic Signaling Signal transmission fromaxon of one neuron to the dendrites of other neuron at the synapse is calledsynaptic signaling. When impulse reaches synapse it triggers the release of NEUROTRANSMITTERSthat binds to the receptors of receiving neurons. The neurotransmitters that are release at the synapse are quickly degraded or takin back up by the sending cell in order to prepare For the next signal.
- 6. Cell Signaling 3. Autocrine Signaling In this type cell signals its self. Cells release a ligand that binds to receptor on its own surface. It is important duringdevelopment, helping cells take on and reinforce their correct identities. This type of signalingalso takes place during cancer. Figure: Difference between Paracrine and Autocrine signaling.
- 7. Cell Signaling 4. Endocrine Signaling When cells need to transmit signals over long distance they useendocrine signaling. In this type circulatory systemis used as a distribution network for the message to send. What is the example? Hormones (growth hormone release by pituitary gland and promote growth in bones and cartilage)
- 8. Cells bind to one anotherwiththe help of surface receptors and transmit the signals. This type of signalingis important in immune system, where immune cells use cell-surface markers to recognize “self” cells (bodies own cells) and cells infectedby pathogens. Cell Signaling 5. Signaling through Cell-Cell contact Figure: Cell-Cell contact signaling Figure: NK immune cell recognizes a healthy cell of the body to a “self” marker on the cell’s surface. Naturel killer (NK) cell Healthy cell
- 9. Name derivedfromthe words, “AUTOS” means “self” and “ÄKOS” means healingsubstance. Produced by a wide variety of cells in the body, having intense activity, and act locally at the site of synthesis and release. They are alsocalled“local hormones”. Question: They are different fromhormones in what way? Autacoids
- 10. Autacoids Classification of Autacoids They are classified into four major classes: 1. Amine Autacoids • Histamine • Serotonin 2. Lipid-derivedAutacoids • Prostaglandins • Leukotrienes • Platelet-activating factor 3. Peptide Autacoids • Plasma Kinins (Bradykinin, Kallidin) • Angiotensin 4. Other Autacoids • Gastric Autacoids (Gastrin) • Cytokines • Somatostatin • Vasoactive Intestinal Peptides • Endothelins • ets
- 11. Autacoids 1. Amine Autacoids Histamine • Histamine meaning “Tissue Amine”, (Histos- tissue). • It is abundantly present in animal Tissues and also in plants like “stingingnettle”. •Histamine mostly is present in mast cells bound to acidic protein and heparin (negativelycharged) because histamine possesses positive charge. • Tissues rich in histamine are: skin, gastric and intestinal mucosa, lungs, liver and placenta. • Non-mast cells histamine is present in: CNS (neurotransmitter), epidermis, gastric mucosa and growing regions. • Turnover of non-mast cells histamine is fast than the mast cells histamine.
- 12. Autacoids 1. Amine Autacoids Histamine Histamine Lipid mediators: Leukotrienes and Prostaglandins Cytokines IL-4, IL-13 TNF-α Mast-cell activation • increase vascular permeability • smooth muscle contraction • promotes Th2 differentiation • promotes IgE production • promotes tissue inflammation • increase vascular permeability • cause smooth muscle contraction • stimulates mucus secretion • Chemoattractants for T cells, eosinophils, mast cells & basophils
- 13. Autacoids 1. Amine Autacoids Histamine Synthesis & Metabolism • Histamine is formed by theDecarboxylationof the amino acid(AA) histidine by the enzymeL-Histidinedecarboxylase. • There are two pathways of its metabolism 1. Methylation: Histamine is convertedintoN-Methyl histamine by N- methyltransferase and further intoN-MethylImidazole Acetic Acidby monoamine oxidase-B. 2. Oxidative deamination: Histamineis convertedinto Imidazoleacetic Acidby Di-amine oxidase andfurther intoImidazole Acetic AcidRiboside by Phosphoribosyl Transferase. • Non-mastcell sites of histamine production contribute significantly to the daily excretionof histamine metabolitesin the urine.
- 14. Autacoids 1. Amine Autacoids Histamine Storage 1. Histamine foundin individual organs and tissues is synthesized locally and stored. 2. Mastcells (withintissues) and basophils (withinblood) synthesize histamine andstore it in secretory granules. 3. It is also foundin non-mastcells, including the hypothalamus regionof the brainwhere it functions as a neurotransmitter. 4. In secretory granules the pHis of 5.5, positively charged histamine is foundin complexwithnegatively chargedacidic groups on other granular constituents, primarily proteases and heparinor chondroitinsulphate proteoglycans. *Basophils: Are a type of white blood cell. Basophils are the least common type of granulocyte, representing about 0.5% to 1% of circulating white blood cells. However, they are the largest type of granulocyte. They are responsible for inflammatory reactions during immune response, as well as in the formation of acute and chronic allergic diseases, including anaphylaxis, asthma, atopic dermatitis and hay fever. They also produce compounds that coordinate immune responses, including histamine and serotonin that induce inflammation, heparin that prevents blood clotting.
- 15. Autacoids 1. Amine Autacoids Histamine Storage 1. Non-mastcell sites of histamine formation include the epidermis, enterochromaffin-like cells of gastricmucosa*, neurons in the CNS andcells in regenerating or rapidly growing tissues. *Enterochromaffin-like cells of gastric mucosa: Enterochromaffin-like (ECL) cells are neuroendocrine cells in the gastric mucosa that control acid secretion by releasing histamine as a paracrine stimulant.
- 16. Autacoids 1. Amine Autacoids Release of Histamine fromStorage sites 1. Histamine is releasedfromthe mast cells by exocytotic or non-exocytotic mechanisms. • Exocytotic includes the release of histamine fromhistaminic granules/vesicles out of the cells. • Non-exocytotic includes the release of histamine by mastcell lysis, modificationof mastcell membranes, and physical displacement of histamine e.g., Mechanical injury. • Non-exocytotic pathway is followedwhenantigen directly interacts withthe mast cells or basophils. • In the exocytoticpathway the stimuli include components of complement systemC3a and C5a, which interacts withspecific surface receptors andthe combinationof antigenwith IgE antibodies. Mast cell Nucleus Granules Histamine Receptor site IgE sensitized Mast cell IgE sensitized mast cell with allergen bound IgE Degranulation. Release of histamine. Antihistamines Pharmacological effects Allergen
- 17. Autacoids 1. Amine Autacoids • In commonwithmany secretory processes, histamine release is initiatedby a rise in cytosolic Ca+2. Mast cell Nucleus Granules Histamine Receptor site IgE sensitized Mast cell IgE sensitized mast cell with allergen bound IgE Degranulation. Release of histamine. Antihistamines Pharmacological effects Allergen 2. Modulationof Histamine release • Certainendogenous and exogenous compounds modulate the antigen-mediatedrelease of histamine fromsensitizedtissues. • Histamine inhibits its ownrelease in skinmastcells and blood basophils by binding to H2 histamine receptors, which whenactivated, inhibit degranulation. • This feedback inhibition does not appear to occur in lung mast cells. • Various basic drugs, such as morphine and tubocurarine, release histamine. • Agents that increase cAMP formation(e.g. β-adrenoceptor agonists) inhibit histamine secretion. • Muscarinic and α-adrenergicagonists enhance mastcell
- 18. Autacoids 1. Amine Autacoids Histamine release by Drugs, Peptides, Venoms and other agents: • Mechanical injury and many compoundsstimulate the release of histamine frommast cells. • Responses most likely occur following intravenous injections of several substances. • Tubocurarine, succinylcholine, morphine, some antibiotics, radiocontrast media (iodine contrast media for the circulatory system, angiography; Bariumfor the digestive system), andcertaincarbohydrate plasma expanders (dextrose, hydroxyethyl starch) also may elicit the response. • Basicpolypeptides oftenare effective histamine releasers, and over a limitedrange, their potency generally increases withthe number of basic groups. For example, bradykininis a poor histamine releaser, whereas kallidin (Lys-bradykinin) and substance P, withmore positively chargedamino acids (lysine, arginine, histideine), are more active. • Some Venoms, such as that of the wasp, containpotent histamine-releasing peptides.
- 20. Autacoids 1. Amine Autacoids Histamine Receptors H1 H2 H3 a H4 G protein Coupling (Second Messenger) Gq/11 ( Ca+2, NO, and cGMP) GS ( cAMP) Gi/o ( cAMP, MAP Kinase) Gi/o ( cAMP) Distribution Smooth muscles, endothelial cells, CNS Gastric parietal cells, Cardiac muscles, mast cells, CNS CNS: Pre- and Post- Synaptic. Cells of hematopoietic origin Representative Agonist Chlorpheniramine Ranitidine Tiprolisant JNJ7777120
- 21. Autacoids 1. Amine Autacoids Activationof H1 receptors on vascular endotheliumstimulates eNOS to produce NO, which diffuses to nearby smooth muscle cells to increase cyclic GMP andcause relaxation. Stimulation of H1 receptors on smooth muscle mobilize Ca2+ and cause contraction. Activationof H2 receptors on the same smooth muscle cell will link via Gs to enhancedcyclicAMP accumulation, activationof PKA, and thento relaxation.
- 24. Autacoids 1. Amine Autacoids Physiological and Pharmacological effects of Histamine 1. H1 and H2 receptors: • H1 and H2 receptors are distributedwidely in the periphery andin the CNS and their activationby histamine can exert local or widespreadeffects • Histamine causes itching and stimulatessecretionfromnasal mucosa. • It contracts many smooth muscles, such as those of the bronchi and gut, but markedly relaxes others, including those in small bloodvessels. • Is a potent stimulus of gastric acid secretion. • Less-prominenteffects include the formationof edemaand stimulationof sensory nerve endings. • Bronchoconstrictionand contractionof the gut are mediatedby H1 receptors. • In the CNS, H1 activationinhibits appetite and increases wakefulness. • Gastricsecretion results fromthe activationof H2 receptors. • Vascular dilation, are mediatedby bothH1 and H2 receptor stimulation.
- 25. Autacoids 1. Amine Autacoids Physiological and Pharmacological effects of Histamine 1. H3 and H4 receptors: • The H3 receptors are expressedmainly in the CNS, especially in the basal ganglia, hippocampus, and cortex. • PresynapticH3 receptors functionas autoreceptors on histaminergic neurons, inhibiting histamine release and modulating the release of other neurotransmitters. • H3 receptors are also foundpostsynaptically, especially in the basal ganglia, but their function is still being unraveled. • H3 agonists promote sleep, and H3 antagonists promote wakefulness. • The H4 receptors primarily are foundin eosinophils, dendritic cells, mast cells, monocytes, basophils, and T cells but have also been detectedin the GI tract, dermal fibroblasts, CNS, andprimary sensory afferent neurons. • Activationof H4 receptors has been associatedwithinductionof cellular shape change, chemotaxis, secretion of cytokines, and upregulationof adhesionmolecules, suggesting that H4 antagonists may be useful inhibitors of allergicand inflammatory responses
- 26. Autacoids 1. Amine Autacoids Physiological and Pharmacological effects of Histamine 1. Cardiovascular System(CVS): • Histamine dilates resistance vessels, increase capillary permeability and lower systemic bloodpressure. • Vasodilationis due to H1 or H2 receptors. H1 receptors have a higher affinity for histamine and cause Ca2+- dependentactivationof eNOS in endothelial cells; NO diffuses to vascular smooth muscle, increasing cyclic GMP andcausing rapidand short-livedvasodilation. By contrast, activation of H2 receptors on vascular smooth muscle stimulates the cyclicAMP–PKApathway, causing dilationthat develops more slowly and is more sustained. • Histamine’seffect on small vessels resultsin efflux of plasma proteinand fluidintothe extracellular spaces and an increase in lymph flow, causing edema(H1 receptors). • On heart, It increases the force of contractionof both atrial andventricular muscle by promoting the influx of Ca2+ thus increases heart rate. • It reduces bloodpressure. • Histamine directly contracts or, more rarely, relaxes various extravascular smooth muscles.
- 27. Autacoids 1. Amine Autacoids Physiological and Pharmacological effects of Histamine 1. Cardiovascular System(CVS): • Histamine stimulates various nerve endings, causing sensory effects. In the epidermis, it causes itch; in the dermis, it evokes pain, sometimes accompaniedby itching. • Histamine givenin large doses or releasedduring systemic anaphylaxis causes a profoundand progressive fall in bloodpressure. Also, due to Dilationof small bloodvessels plasma escapes fromcirculation. • Decrease bloodvolume, reduce venous return, andgreatly lower cardiac output. • Histamine is the toxinin foodpoisoning fromspoiledscombroidfish such as tuna. Symptoms include severe nausea, vomiting, headache, flushing, and sweating. • Histamine toxicity also canfollowred wine consumptionin persons with a diminishedability to degrade histamine. The symptoms of histamine poisoning can be suppressedby H1 antagonists.
- 28. Autacoids 1. Amine Autacoids Physiological and Pharmacological effects of Histamine 2. Extra Vascular smooth muscles: • Histamine directly contracts or, more rarely, relaxes various extravascular smooth muscles. • Contractionis due to activationof H1 receptors on smooth muscle to increase intracellular Ca2+, and relaxationis mainly due to activationof H2 receptors. • Although the spasmogenicinfluence of H1 receptors is dominantin humanbronchial muscle, H2 receptors withdilator functionalso are present. • Patientswithbronchial asthma and certainother pulmonary diseases are much more sensitive to the bronchoconstrictor effects of histamine. • Pregnant womensuffering anaphylactic reactions may abort as a result of histamine-induced uterine contractions.
- 29. Autacoids 1. Amine Autacoids Physiological and Pharmacological effects of Histamine 3. Bronchiolar smooth muscles: • Histamine causes bronchoconstriction mediatedby H1 receptors. Bronchoconstrictionto histamine can be blockedby autonomic-blocking drugs such as ganglion-blocking agents (Hexamethoniumand nicotine) as well as by H1-receptor antagonists. • Curiously, a fewspecies (eg, rabbit) respondto histamine with bronchodilation, reflecting the dominance of the H2 receptor in their airways. • Histamine causes contractionof intestinal smooth muscles. This action of histamine is mediatedby H1 receptors
- 30. Autacoids 1. Amine Autacoids Physiological and Pharmacological effects of Histamine 4. Peripheral nerve endings: • Histamine stimulates various nerve endings, causing sensory effects. • In the epidermis, it causes itch; in the dermis, it evokes pain, sometimes accompaniedby itching. • Stimulantactions on nerve endings contribute to the “flare” component of the triple response andto indirect effects of histamine on the bronchi and other organs. Triple response: • If histamine is injectedintradermally, it elicits a characteristic phenomenonknownas the triple response. This consists of the following: • A localized“reddening” aroundthe injectionsite, appearing withina few seconds, and maximal at about 1 min. • A “flare” or red flush extending about 1 cm beyondthe original red spot and developing more slowly. • A “wheal” or swelling that is discernible in 1–2 minat the injectionsite.
- 31. Autacoids 1. Amine Autacoids Physiological and Pharmacological effects of Histamine Triple response: • The initial redspot (a few millimeters) results fromthe direct vasodilating effect of histamine (H1 receptor–mediated NOproduction). • The flare is due to histamine-inducedstimulationof axon reflexes that cause vasodilation, and • the wheal reflects histamine’scapacity to increase capillary permeability (edemaformation)
- 32. Autacoids 1. Amine Autacoids Physiological and Pharmacological effects of Histamine 5. Secretory tissues: • Histamine has long been recognized as a powerful stimulant of gastric acidsecretionand, to a lesser extent, of gastric pepsinand intrinsic factor production. The effect is causedby activationof H2 receptors on gastric parietal cells and is associatedwithincreasedadenylyl cyclase activity, cAMP concentration, andintracellular Ca2+ concentration. • Histamine also stimulates secretionin the small and large intestine. In contrast, H3-selective histamine agonists inhibit acidsecretionstimulatedby foodor pentagastrinin several species. • Histamine has much smaller effects on the activity of other glandular tissue at ordinary concentrations. Very high concentrations cancause catecholamine release fromthe adrenal medulla.
- 33. Autacoids 1. Amine Autacoids Physiological and Pharmacological effects of Histamine 6. Histamine Shock: • Histamine givenin large doses or releasedduring systemic anaphylaxis causes a profoundand progressive fall in bloodpressure. • As the small bloodvessels dilate, they traplarge amounts of blood, their permeability increases, and plasma escapes fromthe circulation. • These effects, resembling surgical or traumatic shock, diminish effective bloodvolume, reduce venous return, and greatly lower cardiac output.
- 34. Autacoids 1. Amine Autacoids Physiological and Pharmacological effects of Histamine
- 35. Autacoids 1. Amine Autacoids Physiological and Pharmacological effects of Histamine 7. Histamine toxicity: • Adverse effects of histamine release, like those following administrationof histamine, are dose related. Flushing, hypotension, tachycardia, headache, urticaria, bronchoconstriction, and gastrointestinal upset are noted. • These effects are also observedafter the ingestion of spoiledfish (scombroidfish poisoning), and histamine producedby bacterial actionin the flesh of improperly stored fish is the major causative agent.
- 36. Autacoids 1. Amine Autacoids Histamine receptor antagonists 1. FirstGenerationAntihistamines: • DoxepinHCl. • Carbinoxamine Maleate. • Clemastine fumarate. • Diphenhydramine HCl. • Dimenhydrinate. • Chlorpheniramine maleate. • Brompheniramine maleate. • Hydroxyzine HCl. • Hydroxyzine pamoate. • Cyclizine HCl. • Meclizine HCl. • Promethazine HCl. • Cyproheptadine HCl.
- 37. Autacoids 1. Amine Autacoids Histamine receptor antagonists 2. SecondGenerationAntihistamines: • Olopatadine HCl. • Acrivastine. • Cetrizine HCl. • Levocetrizine HCl. • Alcaftadine. • Bepotastine besilate. • Desloratadine. • Fexofenadine HCl. • Ketotifenfumarate. • Loratadine. • Azelastine HCl. • Emedastine. • Epinastine. • Ebastine.
- 38. Autacoids 1. Amine Autacoids Histamine receptor antagonists 1. Effects of antihistamines on physiological system: • Smooth Muscles: Inhibitvasodilator effect on vascular smooth muscles. Inhibitcontractionof respiratory smooth muscles. Also inhibits venous constriction. Strongly block the increasedcapillary permeability and formationof edemaand wheal causedby histamine. H1 antagonists suppress the action of histamine on nerve endings, including the flare component of the triple response and the itching causedby intradermal injection. H1 antagonists do not suppress gastricsecretion. However, the antimuscarinic properties of many H1 antagonists may contribute to lessened secretion. They possess anticholinergiceffects.
- 39. Autacoids 1. Amine Autacoids Histamine receptor antagonist: Anticholinergic effects of 1st generationantihistamines
- 40. Autacoids 1. Amine Autacoids Histamine receptor antagonists 1. Effects of antihistamines on physiological system:
- 41. Autacoids 1. Amine Autacoids Histamine receptor antagonists 1. Effects of antihistamines on physiological system: • MastCell–Stabilizing and Anti-inflammatory Properties: Act as mast cells stabilizers tooduring allergic response(Many secondgeneral H1 blockers: citrizine, fexofenedine etc). These agents also have anti-inflammatory properties, which can include reducedcytokine secretion, decreasedadhesionmolecule expression, and inhibitionof eosinophil infiltration. These effects can be both H1 receptor dependent and independent, but precise mechanisms are still unclear, and it is unknownwhat role they play at normal therapeutic doses of these drugs.
- 42. Autacoids 1. Amine Autacoids Histamine receptor antagonists 1. Effects of antihistamines on physiological system: • CNS: H1 antagonists can bothstimulate and depress the CNS. Stimulationoccasionally is encounteredin patients given conventional doses; the patients become restless, nervous, and unable to sleep. Central excitationalso is a striking feature of overdose, which commonly results in convulsions, particularly in infants. Central depressionincludes, Diminishedalertness, slowedreactiontimes, and somnolence are commonmanifestations. Patients vary in their susceptibility and responses to individual drugs. Firstgenerationcause sedation, therefore these drugs cannot be toleratedor usedsafely by many patients except at bedtimes. Patientsmay experience an antihistamine “hangover” in the morning, resulting in sedationwithor withoutpsychomotor impairment.
- 43. Autacoids 1. Amine Autacoids Histamine receptor antagonists 1. Effects of antihistamines on physiological system: • CNS: Second-generationH1 antagonists are termed nonsedating because they do not cross the blood- brainbarrier appreciably. This is due to their decreasedlipophilicity and because they are substrates of P-glycoprotein*, which pumps themout of the blood-brainbarrier capillary endothelial cells and back intothe capillary lumen. *P-glycoprotein 1 (permeability glycoprotein,) also known as multidrug resistance protein 1 (MDR1) or ATP-binding cassette sub- family B member 1 (ABCB1) or cluster of differentiation 243 (CD243) is an important protein of the cell membrane that pumps many foreign substances out of cells. More formally, it is an ATP-dependent efflux pump with broad substrate specificity. It exists in animals, fungi, and bacteria, and it likely evolved as a defense mechanism against harmful substances.
- 44. Autacoids 1. Amine Autacoids Histamine receptor antagonists 1. Effects of antihistamines on physiological system: • Local anestheticeffects: Some H1 antagonists have local anestheticactivity. However, the concentrations requiredfor this effectare much higher than those that antagonize histamine’s interactions with its receptors.
- 45. Autacoids 1. Amine Autacoids Histamine receptor antagonists 2. Pharmacokinetics (ADME) of antihistamines: Well absorbedfromGI tract after oral administration. Peak plasmaconcentrationis achievedin 1 to 3 hours. Firstgenerationantihistamines’ effects last for 4 to 6 hours. Secondgenerationantihistamineshave much longer effects. These agents are distributedwidely throughout the body, including the CNS for the first-generation agents. Peak concentrations of these drugs in the skinmay persist long after plasmalevels have declined. Thus, inhibitionof “wheal-and-flare” responses to the intradermal injectionof histamine or allergen can persistfor 36 h or more after initial treatment and up to 7 days after discontinuationof treatmentin patients who regularly use an H1 antagonist for 1 week or more. All first-generationand mostsecond-generation H1 antagonists are metabolizedby CYPs and little is excretedunchangedin the urine.
- 46. Autacoids 1. Amine Autacoids Histamine receptor antagonists 2. Pharmacokinetics (ADME) of antihistamines: Exceptions are: Citrizine and Acrivastin, <40%metabolized. • Fexofenadine, levocetirizine, andepinastine, <10%metabolized. Cetirizine, levocetirizine, and acrivastine are excretedprimarily intothe urine. fexofenadine is mainly excretedin the feces Epinastine is excretedboth in urine and feaces. The H 1 antagonists that are metabolized are eliminatedmore rapidly by children thanby adults and more slowly in those withsevere liver disease. These antagonists also have higher potential for drug interactions. For example, plasma levels of H1 antagonists may be reducedwhencoadministered with drugs that induce CYP synthesis (e.g., benzodiazepines) or elevatedwhen takenwithdrugs that compete withor inhibit the same CYP isoform(e.g., erythromycin, ketoconazole, antidepressants).
- 47. Autacoids 1. Amine Autacoids Histamine receptor antagonists 2. Pharmacokinetics (ADME) of antihistamines: Clinically relevantinteractions are more likely with first-generationthansecond-generationdrugs. However, two second-generationH1 antagonists marketedpreviously, terfenadine and astemizole, were foundin rare cases to prolong the QTc interval and induce a potentially fatal arrhythmia, due to their capacity to inhibit a cardiac K+ channel whentheir metabolismwas impairedand their plasmaconcentrations rose toohigh, due, for instance, to liver disease or to drugs that inhibitedthe CYP3A family. This led to the withdrawal of terfenadine and astemizole fromthe market. Astemizole and an active hydroxylatedmetabolite naturally have very long half-lives. Terfenadine is a prodrug, metabolizedby hepatic CYP3A4 to fexofenadine, which is its replacement and lacks noticeable cardiotoxicity.
- 48. Autacoids 1. Amine Autacoids Histamine receptor antagonists 3. Therapeuticuses of antihistamines: H1 antagonists for acute allergic reactions that present with symptoms of rhinitis, urticaria, and conjustivitis. In bronchial asthmabut not usedas a sole therapy. In the treatment of systemicanaphylaxis where autocoids other thanhistamine are important, the mainstay of therapy is epinephrine; histamine antagonists have only a subordinate and adjuvant role. The same is true for severe angioedema, in which laryngeal swelling constitutes a threat to life. Urticaria(a raised, itchy rash that appears on the skin). Contact dermatitis (an itchy rash causedby direct contact witha substance or an allergic reaction to it.) although topical corticosteroids are more effective. H1 antagonists can be usedin combating the commoncold. The weakanticholinergic effects of the older agents may tendto lessenrhinorrhea, but this drying effect may do more harm thangood.
- 49. Autacoids 1. Amine Autacoids Histamine receptor antagonists Adverse Effects of antihistamines: The mostfrequentside effectof first-generationH1 antagonists is sedation. Concurrent ingestionof alcohol or other CNS depressants produces an additive effect that impairs motor skills. Other untowardcentral actions include dizziness, tinnitus, lassitude (state of physical or mental weakness or lethargy), incoordination, fatigue, blurredvision, diplopia(double visionor seeing double), euphoria, nervousness, insomnia, and tremors. Other potential side effects, including loss of appetite, nausea, vomiting, epigastric distress, and constipationor diarrhea, may be reducedby taking the drugs with meal. H1 antagonists such as cyproheptadine may increase appetite and cause weight gain. Other side effects, owing to the antimuscarinic actions of some first-generationH1 antagonists, include dryness of the mouth and respiratory passages (sometimes inducing cough), urinary retentionor frequency, and dysuria.
- 50. Autacoids 1. Amine Autacoids Histamine receptor antagonists Adverse Effects of antihistamines: Hypersensitivity reactions include drug fever and photosensitization. Hematological complications, such as leukopenia (low white bloodcell count ), agranulocytosis (Agranulocytosis is a life-threatening blooddisorder. It happens whenthe body doesn'tmake enough of a type of white bloodcells calledneutrophils), and hemolytic anemia(Hemolytic anemia is a disorder in which red bloodcells are destroyedfaster than they can be made.), are very rare. H1 antihistamines cross the placenta, caution is advisedfor womenwho are or may become pregnant. Antihistamines can be excretedin small amounts in breast milk, and first-generation antihistamines takenby lactating mothers may cause symptoms such as irritability, drowsiness, or respiratory depressionin the nursing infant.
- 51. Autacoids 1. Amine Autacoids Histamine receptor antagonists Adverse Effects of antihistamines: In acute poisoning with first-generationH1 antagonists, their central excitatory effects constitute the greatest danger. The syndrome includes hallucinations, excitement, ataxia, incoordination, athetosis, andconvulsions, fixed, dilatedpupils with a flushed face, together withsinus tachycardia, urinary retention, dry mouth, andfever. The syndrome exhibits a remarkable similarity to that of atropine poisoning. Terminally, there is deepening coma with cardiorespiratory collapse and death usually within2–18 h. First-generationantihistamines are not recommendedfor use in childrenbecause their sedative effects canimpair learning and school performance
- 52. Autacoids 1. Amine Autacoids Histamine and anti-histamines Biosynthesis and Distribution of Histamine. Histamine is produced by decarboxylation of the amino acid histidine. (1) It la synlhe1ized in the mast cells and basophils of the immune system, in the enterochramaffin-like cells (ECL) of the gastric mucosa, and in different neurons of the central nervous system (CNS). (a) Mast cells and basophils store histamine in secretory granules. Its release is induced by immunoglobulin B (lgB) fixation to mast cells (sensitization) and subsequent exposure to a specific antigen. i. Allergic processes or anaphylaxis can trigger degranulation. (b) ECL cells and histaminergic CNS neurons continuously release histamine as required for gastric acid secretion and neurotransmission, respectively.
- 53. Autacoids 1. Amine Autacoids Physiological effects of Histamine. All four receptor subtypes H1, H2, H3 and H4 are GPCRs. 1. Histamine (H1) Receptors Located in smooth muscles, vascular endothelium and brain. They are Gq coupled and activate the second messenger inositol triphosphate (IP3) and diacylglycerol (DAG). H1 receptors commonly mediate inflammatory and allergic reactions. Activation in the: (a) Lungs can lead to bronchoconstriction and asthma-like symptoms. (b) Vascular smooth muscle may cause vasodilation, leading to erythema (redness of the skin caused by injury or another inflammation-causing condition). (c) Vascular endothelium can lead to edema. (d) Peripheral nerves can lead to itchiness and pain.
- 54. Autacoids 1. Amine Autacoids Physiological effects of Histamine. 2. Histamine (H2) Receptors (1) These receptors are commonly found in parietal cells and cardiac muscles. (2) They are Gs coupled and lead to the activation of adanylyl cyclase and increased cyclic AMP (cAMP) production. (3) Activation of H2-receptors: (a) Increases gastric acid production, potentially leading to increased risk of peptic ulcer disease or heartburn. (b) May also cause minor increases in heart rate. 3. H3 and H4 receptors are located in the brain, hematopoietic cells, and thymus. (1) There is no approved drugs that act through these receptor.
- 55. Autacoids 1. Amine Autacoids Anti histamine classification First Generation Antihistamines: • Doxepin HCl. • Carbinoxamine Maleate. • Clemastine fumarate. • Diphenhydramine HCl. • Dimenhydrinate. • Chlorpheniramine maleate. • Brompheniramine maleate. • Hydroxyzine HCl. • Hydroxyzine pamoate. • Cyclizine HCl. • Meclizine HCl. • Promethazine HCl. • Cyproheptadine HCl.
- 56. Autacoids 1. Amine Autacoids Anti histamine classification Second Generation Antihistamines: • Olopatadine HCl. • Acrivastine. • Cetrizine HCl. • Levocetrizine HCl. • Alcaftadine. • Bepotastine besilate. • Desloratadine. • Fexofenadine HCl. • Ketotifen fumarate. • Loratadine. • Azelastine HCl. • Emedastine. • Epinastine. • Ebastine.
- 57. Autacoids 1. Amine Autacoids Histamine (H1) receptor antagonist. 2. Histamine (H1) Receptors a) Mechanism of action. These drugs are competitive inhibitors at the H1-receptor. They relax histamine-induced contraction of bronchial smooth muscle and have some use in allergic bronchospasm. They block the vasodilator action of histamine and inhibit histamine- induced increases in capillary permeability. They do not affect the release of histamine from secretory granules and are more effective when given prior to histamine release.
- 58. Autacoids 1. Amine Autacoids Histamine (H1) receptor antagonist. 2. Histamine (H1) Receptors a) First-generation agents Diphenhydramine HCl, Dimenhydrinate, Chlorpheniramine maleate, Cyclizine HCl, Meclizine HCl, Promethazine HCl As a class, these agents are lipid soluble, readily cross the blood-brain barrier, and have significant CNS actions, mediated in large part by their anticholinergic activity. 1) Their structures are similar to antimuscarinic agents. Indications: a.These agents are commonly used for management of alIergic symptoms, insomnia, motion sickness, and nausea and vomiting.
- 59. Autacoids 1. Amine Autacoids Histamine (H1) receptor antagonist. Adverse effects: a. Include sedation (synergistic with alcohol and other depressants), dizziness, and loss of appetite. b. They also produce anticholinergic effects (dry mouth, blurred vision, and urine retention). b) Second-generation agents Cetrizine HCl, Levocetrizine HCl, Fexofenadine HCl, Loratadine, desloratadine, Ebastine These agents are lipophobic and were developed to avoid the sedation and anticholinergic activity of the first-generation drugs. Indications: a.These agents are preferred over first-generation agents for the treatment of allergic rhinitis, as they have similar efficacy and less CNS effects.
- 60. Autacoids 1. Amine Autacoids Histamine (H1) receptor antagonist. Adverse effects: a. Significantly reduced with second-generation agents. Histamine (H2) receptor antagonist. a) Mechanism of action. These agents are competitive antagonists at the H2 receptor in the gastric parietal cell; they inhibit gastric acid secretion. b) Specific agents: Cimetidine, Ranitidine, famotidine, and nizatidine. c) Indications: They are used in the treatment of heartburn and acid-induced indigestion. They promote the healing of gastric and duodenal ulcers and are used in the management of gastroesophageal reflux disease (GERD).
- 61. Autacoids 1. Amine Autacoids Histamine (H2) receptor antagonist. d) Adverse Effects: H2 antagonists are very safe drugs, and adverse effects are rare. Cimetidine is an androgen-receptor antagonist and may cause gynecomastia (increase in the amount of breast gland tissue in boys or men, caused by an imbalance of the hormones estrogen and testosterone.) and Impotence e) Drug interactions: Cimetidine is also a potent cytochrome P-450 (CYP450) inhibitor and has the potential for many drug interactions.
- 62. Autacoids 1. Amine Autacoids Mast cells stabilizers. a) Mechanism of action. Mast cell stabilizers inhibit the release of histamine and other autocoids from mast cells.. b) Specific agents: Cromolyn and nedocromil. c) Indications: a. Cromolyn is available in several dosage forms: 1) Oral solution for systemic mastocytosis (a rare condition caused by an excess number of mast cells gathering in the body's tissues). 2) Oral inhalation (nebulization) for asthma and prevention of bronchospasm 3) Nasal spray for allergic rhinitis 4) Ophthalmic solution for conjunctivitis and keratitis.
- 63. Autacoids 1. Amine Autacoids c) Indications: Nedocromil is available as an ophthalmic solution for allergic conjunctivitis. d) Adverse effects Include headache, dry mouth, and dry eyes.
- 64. Autacoids 1. Amine Autacoids Serotonin agonist and Antagonist Serotonin (5-hydraxytryptllmine, 5-HT) 1. Biosynthesis and Distribution: a) Serotonin is synthesized from the amino acid tryptophan by hydroxylation and decarboxylation, in a two-step pathway. b) Most serotonin is synthesized in the ECL cel1s of the gastrointestinal (GI) tract. It is also found in platelets and neurons of the CNS and enteric nervous system. 1. In the CNS, cells in the raphe nuclei of the brainstem predominantly synthesize serotonin. c) Serotonin is also a precursor of melatonin.
- 65. Autacoids 1. Amine Autacoids Serotonin agonist and Antagonist Serotonin (5-hydraxytryptllmine, 5-HT) Physiological actions of serotonin: a) There are multiple 5-IIT-receptor subtypes that mediate a wide variety of physiological effects. b) 5-HT1-receptors are Gi, coupled and Inhibit adenylyl cyclase and decrease cAMP. (1) These receptors are located in the brain and smooth muscle tissues. c) 5-HT2 receptors are Gq coupled and increase Phospholipase C activity. (1) These receptors are located in the CNS and mediate hallucinogenic effects. (2) Stimulation can also cause contraction of vascular and intestinal
- 66. Autacoids 1. Amine Autacoids Serotonin agonist and Antagonist Serotonin (5-hydraxytryptllmine, 5-HT) Physiological actions of serotonin: smooth muscle, increase microcirculation and vascular permeability, and lead to platelet aggregation. d) 5-HT3 receptors are ligand gated sodium and potassium ion channels. (1) Stimulation in the area postrema causes nausea and vomiting. (2) Stimulation on peripheral sensory neurons causes pain. e) 5-HT4-receptors are Gs coupled and stimulate adenylyl cyclase and increase cAMP. (1) In the GI tract, they mediate an increase in secretion and peristalsis.
- 67. Autacoids 1. Amine Autacoids Serotonin agonist and Antagonist Serotonin (5-hydraxytryptllmine, 5-HT) Serotonin Agonists: 1) Buspirone (a) Mechanism of Action: Buspirone is a 5-HT1A selective partial agonist It also possess weak dopamine receptor antagonism. (b) Indication: It is used for the management of generalized anxiety disorder. (1) It may take 2 weeks for the beneficial effects to appear. (2) It does not affect GABA receptors; therefore, it is not addictive and causes less sedation than other anxiety medications like benzodiazepines. (C) Adverse effects: dizziness, drowsiness, and nasuea.
- 68. Autacoids 1. Amine Autacoids Serotonin agonist and Antagonist Serotonin (5-hydraxytryptllmine, 5-HT) Serotonin Agonists: 2) Triptans (a) Specific Agents include: Sumatriptan, rizatriptan, eletriptan, zolmitriptan, almotriptan, frovatriptan, and naratriptan. (1) All are available as oral agents. Some are also available as nasal sprays and subcutaneous injection. (b) Mechanism of Action: Triptans are 5HT1B/1D-receptor agonists. They cause vasoconstriction and reduce inflammation in intracranial blood vessels and sensory nerves of the trigeminal system. (C) Indications: They are commonly used for the treatment of acute migraines and cluster headaches. About 50%-80% of patients report relief from pain within 2 hours.
- 69. Autacoids 1. Amine Autacoids Serotonin agonist and Antagonist Serotonin (5-hydraxytryptllmine, 5-HT) Serotonin Agonists: (D) Adverse effects: May include dizziness, drowsiness, flushing, and chest pain. (1) They can cause coronary vasospasm and are contraindicated in patients with coronary artery diseases (CAD) or angina.
- 70. Autacoids 1. Amine Autacoids Serotonin agonist and Antagonist Serotonin (5-hydraxytryptllmine, 5-HT) Serotonin Antagonists: (a) Mechanism of Action: They block serotonin receptors centrally in the chemoreceptor trigger zone and peripherally on vagal nerve terminals. (b) Specific agents: Ondansetron, granisetron, dolasetron, and palonosetron. (c) Indication: They are used for the prevention of nausea and vomiting due to chemotherapy and radiation therapy. They are also used to prevent postoperative nausea and vomiting. (d) Adverse effects: Include headache and constipation. They also have the potential to cause QTs prolongation and arrhythmias.
- 71. Autacoids 1. Amine Autacoids Serotonin agonist and Antagonist Serotonin (5-hydraxytryptllmine, 5-HT) Serotonin Antagonists: Alosetron is a 5-HT3 antagonist approved for diarrhoea-predominant irritable bowel syndrome in women. It may cause serious GI side effects, including ischemic colitis and severe constipation.
- 73. Autacoids 2. Lipid derived Autacoids It includes Prostaglandins Leukotrienes Platelet-activating factor (non-eicosanoids)
- 74. Autacoids Eicosanoids are a large group of autocoids with potent effects on many tissues in the body. Eicosanoids are biologically active lipid derivatives of unsaturated fatty acids containing 20 carbons. Eicosanoids are locally acting bioactive hormones that act near the point of hormone synthesis and included in the class of paracrine hormones. Eicosanoids are derived from arachidonic acid and related polyunsaturated fatty acids (PUFAs) such as eicosapentaenoic acid (EPA). The eicosanoids include the prostaglandins, thromboxanes, leukotrienes, hydroperoxyeicosatetraenoic acids (HPETEs), and hydroxyeicosatetraenoic acids (HETEs). 2. Lipid derived Autacoids
- 75. Autacoids Synthesis of eicosanoids Primary precursor and synthesis (a) The primary precursor of the prostaglandins and other related eicosanoids is the Arachidonic acid, containing 20 carbons. 2. Lipid derived Autacoids
- 76. Autacoids Synthesis of eicosanoids Primary precursor and synthesis (b) Arachidonic acid is primarily the constituent of cell membrane phospholipids, mainly found in phosphatidylinositol and in other complex forms of lipid. (c) The release of free arachidonic acid, associated with phospholipids, is performed mainly by the activity of an enzyme called phospholipase A2 and other acyl hydrolases also play some role. (d) The release of free arachidonic acid is under controlled of hormones and other stimuli. 2. Lipid derived Autacoids
- 77. Autacoids Synthesis of eicosanoids Primary precursor and synthesis 2. Lipid derived Autacoids
- 78. Autacoids Synthesis of eicosanoids Primary precursor and synthesis 2. Lipid derived Autacoids
- 79. Autacoids Synthesis of eicosanoids Primary precursor and synthesis 1. Eicosanoids are synthesized by two pathways: Cyclooxygenase and Lipoxygenase. a. Cyclooxygenase (COX) pathway (there are two forms of COX) The COX-1 enzyme is located in the kidney, GI tract, and platelets, as well as many other locations. (1) It is expressed at fairly constant level and provides protective action on gastric mucosa, the endothelium, and in the kidney. The COX-2 enzyme is found in great abundance in connective tissues, in the kidney, and in the endothelium. It is highly inducible by numerous factors associated with inflammation. 2. Lipid derived Autacoids
- 80. Autacoids Synthesis of eicosanoids Primary precursor and synthesis The cox pathway produces thromboxane (TXA2), prostaglandin E(PGE), prostaglandin F(PGF), prostaglandin D (PGD), and prostacyclin (PGI2). b. Lipoxygenase (LOX) pathway 5-LOX association with 5-LOX-activating protein (FLAP) produces 5-HPETEs, which are subsequently converted to 5- HETBs and then to leukotrienes. 2. Eicosanoids are synthesized through out the body (often tissue specific) and have various physiological effects. PGI2 is synthesized in endothelial and vascular smooth muscle cells. Thromboxane synthesis occurs primarily in platelets. 2. Lipid derived Autacoids
- 81. Autacoids Synthesis of eicosanoids Primary precursor and synthesis Thromboxane synthesis occurs primarily in platelets. HPETEs, HETEs, and the leukotrienes are synthesized predominantly in mast cells, white blood cells, airway epithelium, and platelets. 2. Lipid derived Autacoids Table: Location and physiological actions of Eicosanoids.
- 82. Autacoids Synthesis of eicosanoids Primary precursor and synthesis 2. Lipid derived Autacoids
- 83. Autacoids Indications of eicosanoids 1. Cervical Ripening: Dinoprostone (PGE2), naturally occurring prostaglandin, reduces the collagen network within the cervix. It softens the cervix and relaxes the smooth muscles, allowing dilation and passage of the fetus through the birth canal. 2. Termination of pregnancy: Carboprost tromethamine (PGF2α) and dinoprostone stimulate uterine contractions similar to natural labor contractions. Currently, these prostaglandins are combined with mifepristone, an antiprogesterone, which blocks the effects Of progesterone to cause contraction- inducing activity in the myometrium. 2. Lipid derived Autacoids
- 84. Autacoids Indications of eicosanoids 3. Maintenance of ductus arteriosus*: Alprostadil (PGE1) will maintain patency of the ductus arteriosus, which may be desirable before surgery. 4. Nonsteroidal anti-inflammatory drug (NSAID)-induced ulcers: Misoprostol (PGE1 analog) replaces the protective prostaglandins (gastromucosal defense mechanisms) consumed with prostaglandin-inhibiting therapies, such as NSAIDs. 5. Erectile dysfunction: Alprostadil (PGE1) can be injected directly into the corpus cavernosum or administered as a transurethral suppository to cause vasodilation and enhance tumescence. It relaxes trabecular smooth muscle, which allows blood flow to the lacunar spaces of the penis. 6. Glaucoma: Latanoprost (PGF2α analog) reduces intraocular pressure by increasing the outflow of aqueous humor. 2. Lipid derived Autacoids
- 85. Autacoids 2. Lipid derived Autacoids *Ductus arteriosus: Before a baby is born, the fetus's blood does not need to go to the lungs to get oxygenated. The ductus arteriosus is a hole that allows the blood to skip the circulation to the lungs. However, when the baby is born, the blood must receive oxygen in the lungs and this hole is supposed to close. If the ductus arteriosus is still open (or patent) the blood may skip this necessary step of circulation. The open hole is called the patent ductus arteriosus.
- 86. Autacoids Indications of eicosanoids 7. Pulmonary hypertension: PGI2 analogs (epoprostenol, treprostinil, and iloprost) are strong vasodilators of pulmonary and systemic vascular beds. 2. Lipid derived Autacoids
- 87. Autacoids Adverse effects of eicosanoids Adverse effects of eicosanoids include 1. Local pain 2. Irritation 3. Bronchospasm 4. GI disturbances, Including nausea Vomiting, cramping, and diarrhoea. 2. Lipid derived Autacoids
- 88. Autacoids Inhibitors and Antagonists 1. NSAIDs are used to suppress the symptoms of inflammation and relieve pain (analgesic action) and fever (antipyretic action). 2. Anti-inflammatory effect a) PGE2 and PGl2 are primarily involved with inflammation. They promote blood flow in the inflamed region, enhance edema formation, and enhance leukocyte infiltration. b) The anti-inflammatory effect of NSAIDs is due to the inhibition of COX- 1 and COX-2. COX-2 plays an important role in the inflammatory process, although the effect of its inhibition on Inflammation is not fully understood. (1) Aspirin irreversibly inactivates COX-1 and COX-2 by acetylation of a specific serine residue. This distinguishes it from other NSAIDs, which reversibly inhibit COX-1 and COX-2. 2. Lipid derived Autacoids
- 89. Autacoids Inhibitors and Antagonists (2) COX-2 selective agents inhibit COX-2 more than COX-1. They inhibit COX-2-mediated prostacyclin synthesis in the vascular endothelium. a) Rationale for development: Inhibition of COX-2 would reduce the inflammatory response and pain but not inhibit the cytoprotective action of prostaglandins in the stomach. Unfortunately, this caused an increased risk for serious cardiovascular thrombotic events. (3) NSAIDs have no effect on lipoxygenase and therefore do not inhibit the production of leukotrienes 2. Lipid derived Autacoids
- 90. Autacoids Inhibitors and Antagonists 3. Analgesic effect (decrease pain): PGE2 and PGl2 are the most important prostaglandins involved in pain. They are hyperalgesic in the periphery and centrally. Inhibition of their synthesis is a primary mechanism of NSAID- mediated analgesia. 4. Antipyretic effect: The antipyretic effect of NSAIDs is believed to be related to inhibition of production of prostaglandins induced by interleukin-1 (IL-1) and interleukin-6 (lL-6) in the hypothalamus and the resetting of the thermoregulatory system, leading to vasodilatation and increased heat loss. 2. Lipid derived Autacoids
- 91. Autacoids Inhibitors and Antagonists Indications 1. NSAIDs are first-line drugs used to arrest inflammation and the accompanying pain of rheumatic and nonrheumatic diseases. a. They are used in rheumatoid arthritis, juvenile arthritis, osteo arthritis, psoriatic arthritis, ankylosing spondylitis, reactive arthritis (Reiter syndrome), dysmenorrhea, bursitis, and tendonitis. 1. NSAIDs suppress the signs of underlying inflammatory response but may not reverse or resolve the inflammatory process. b. Treatment of chronic inflammation requires higher doses than those used for analgesia and antipyresis; consequently, the incidence of adverse drug effects is increased. c. In some cases, anti-inflammatory effects may develop only after several weeks of treatment. 2. Lipid derived Autacoids
- 92. Autacoids Inhibitors and Antagonists Rheumatoid arthritis: Rheumatoid arthritis, or RA, is an autoimmune and inflammatory disease, which means that your immune system attacks healthy cells in your body by mistake, causing inflammation (painful swelling) in the affected parts of the body. RA mainly attacks the joints, usually many joints at once. Juvenile arthritis: The most common type of childhood arthritis is juvenile idiopathic arthritis (JIA), also known as juvenile rheumatoid arthritis. Childhood arthritis can cause permanent physical damage to joints. This damage can make it hard for the child to do everyday things like walking or dressing and can result in disability. Osteo arthritis: Osteoarthritis (OA) is the most common form of arthritis. Some people call it degenerative joint disease or “wear and tear” arthritis. It occurs most frequently in the hands, hips, and knees. With OA, the cartilage within a joint begins to break down and the underlying bone begins to change. 2. Lipid derived Autacoids
- 93. Autacoids Inhibitors and Antagonists Psoriatic arthritis: Psoriatic arthritis is a type of arthritis linked with psoriasis, a chronic skin and nail disease. Psoriasis causes red, scaly rashes and thick, pitted fingernails. Psoriatic arthritis is similar to rheumatoid arthritis (RA) in symptoms and joint swelling (inflammation). But it tends to affect fewer joints than RA. Ankylosing spondylitis: Ankylosing spondylitis is a type of arthritis that causes inflammation in the joints and ligaments of the spine. Normally, the joints and ligaments in the spine help us move and bend. If you have ankylosing spondylitis, over time, the inflammation in the joints and tissues of the spine can cause stiffness. Reactive arthritis (Reiter syndrome): Reactive arthritis is joint pain and swelling triggered by an infection in another part of the body — most often the intestines, genitals or urinary tract. This condition usually targets the knees, ankles and feet. Inflammation also can affect the eyes, skin and the tube that carries urine out of the body (urethra). 2. Lipid derived Autacoids
- 94. Autacoids Inhibitors and Antagonists Dysmenorrhea: Menstrual cramps (dysmenorrhea) are throbbing or cramping pains in the lower abdomen. Many women have menstrual cramps just before and during their menstrual periods. For some women, the discomfort is merely annoying. Bursitis: A painful condition that affects the small, fluid-filled sacs — called bursae (bur-SEE) — that cushion the bones, tendons and muscles near your joints. Bursitis occurs when bursae become inflamed. The most common locations for bursitis are in the shoulder, elbow and hip. Tendinitis: Tendinitis is inflammation or irritation of a tendon — the thick fibrous cords that attach muscle to bone. The condition causes pain and tenderness just outside a joint. While tendinitis can occur in any of your tendons, it's most common around your shoulders, elbows, wrists, knees and heels. 2. Lipid derived Autacoids
- 95. Autacoids Inhibitors and Antagonists Indications 2. NSAIDs are used to alleviate mild to moderate pain. a. They are more effective for pain associated with integumental structures (pain of muscular and vascular origin, arthritis, and bursitis) than with pain associated with the viscera. b. They are less effective than opioids. 3. NSAIDs are used to reduce elevate body temperature. They have little effect on normal body temperature. 4. Aspirin reduces the formation of thrombi. a. At low doses, aspirin is more selective for COX-1. b. It has significantly greater antithrombotic activity than other NSAIDs and is useful in preventing or reducing the risk of myocardial infarction in patients with a history of myocardial infarction, angina, cardiac surgery, and cerebral or peripheral vascular disease. 2. Lipid derived Autacoids
- 96. Autacoids Inhibitors and Antagonists Indications c. It is also used prophylactically to reduce recurrent transient ischemia, unstable angina, and the incidence of thrombosis after coronary artery by pass grafts. 5. Salicylic acid is used topically to treat plantar warts, fungal infections, and corns. a. It causes destruction of keratinocytes and dermal epithelia by the free acid. 2. Lipid derived Autacoids
- 97. Autacoids Inhibitors and Antagonists Adverse Effects 1. Gastrointestinal events GI problems are the most common adverse effects. These may include nausea, vomiting, diarrhea, constipation, dyspepsia, epigastric pain, bleeding, and ulceration of stomach, duodenum, and small intestine. Elderly patients and patients with a history of GI bleeding or peptic ulcer disease are at greatest risk. These effects are most likely due to a decrease in the production and cytoprotective activity of prostaglandins, as well as a direct chemical effect on gastric cells. Substitution of enteric-coated or timed-release preparations, or the use of nonacetylated salicylates, may decrease gastric irritation. 2. Lipid derived Autacoids
- 98. Autacoids Inhibitors and Antagonists Adverse Effects 2. Hypersensitivity (intolerance) (also known as aspirin or NSAID- exacerbated respiratory disease) Hypersensitivity is relatively uncommon, but can result in rash, bronchospasm, rhinitis, edema, or an anaphylactic reaction with shock, which may be life-threatening. The incidence is highest in patients with asthma and nasal polyps. Cross-hypersensitivity may exist between aspirin and other NSAIDs. It is thought that inhibiting the COX pathway diverts arachidonic acid metabolites to the lipoxygenase pathway, leading to increased synthesis of cysteinyl leukotrienes (proinflammatory mediators that have an important role in the pathophysiology of asthma). Leukotriene-modifying agents, such as a leukotriene-receptor antagonist (montelukast or zafirlukast), can help prevent exacerbations. 2. Lipid derived Autacoids
- 99. Autacoids Inhibitors and Antagonists Adverse Effects 3. Reye syndrome Reye syndrome is an illness characterized by vomiting, hepatic disturbances, and encephalopathy. It occurs when aspirin or other salicylates are used to control fever during viral infections (influenza and chickenpox) in children and adolescents. Acetaminophen is recommended as a substitute for children with fever of unknown etiology. 4. Renal impairment NSAIDs may reduce renal blood flow and compromise existing renal function. 2. Lipid derived Autacoids
- 100. Autacoids Inhibitors and Antagonists Adverse Effects 5. Cardiovascular events Although aspirin has cardioprotective effects, other NSAIDs can cause an increased risk of serious cardiovascular thrombotic events, such as myocardial infarction or stroke. Compared to other nonselective NSAIDs, COX-2 selective inhibitors have an increased incidence of heart attack and stroke. One potential reason for the increased risk of serious (and potentially fatal) adverse cardiovascular thrombotic events: inhibition of COX-2- mediated production of the vasodilator, PGl21 by endothelial cells, while not affecting the prothrombotic actions of COX-I in platelets, increases the chance of blood clots. 2. Lipid derived Autacoids
- 101. Autacoids Inhibitors and Antagonists Adverse Effects 5. Cardiovascular events Prolonged bleeding time Platelet adhesion and aggregation may be decreased. Patients on anticoagulant therapy or with coagulation disorders must be monitored closely. Aspirin irreversibly inhibits platelet COX-1 and COX-2 and, therefore, irreversibly inhibits TXA2 production, suppressing platelet adhesion and aggregation. Sulfonamide allergy COX-2 selective inhibitors contain the sulfonamide structure; therefore, caution must be used in those with a sulfa allergy. 2. Lipid derived Autacoids
- 102. Autacoids Inhibitors and Antagonists Drug Interactions 1. Anticoagulants and antiplatelet agents The antiplatelet properties of salicylates and NSAIDs in combination with other anticoagulant or antiplatelet agents may increase the risk of bleeding. 2. Sulfonylureas (glyburide or Glibenclamide, glimepiride, glipizide) The hypoglycemic action of sulfonylureas may be enhanced when given with aspirin and certain NSAIDs due to displacement from their binding sites on serum albumin. 3. Methotrexate a. NSAIDs or salicylates should not be given in combination with methotrexate due to the risk of methotrexate toxicity, which may include hematologic toxicity (neutropenia, thrombocytopenia), naphrotoxicity, and hepatotoxicity. 2. Lipid derived Autacoids
- 103. Autacoids Inhibitors and Antagonists Drug Interactions There are several potential mechanisms for this interaction, including decreased renal excretion of methotrexate. 4. Alcohol Patients should be advised that regularly consuming alcohol when taking NSAIDs or salicylates could increase the bleeding risk. 5. Aspirin interaction with NSAIDs Some nonselective NSAIDs may exhibit greater affinity than aspirin for the active site on the COX enzyme, limiting aspirin's irreversible inhibition of COX. COX-2 selective NSAIDs have less risk for this potential interaction. 2. Lipid derived Autacoids
- 104. Autacoids Inhibitors and Antagonists NSAIDs classification: Propionic acid derivative& a)Ibuprofen, naproxen, fenoprofen, and ketoprofen Acetic acid derivatives a)Indomethacin, sulindac, ketorolac, and diclofenac b)Diclofenac is a potent anti-inflammatory agent c)Ketorolac is a potent analgesic and is used for the management of moderately severe acute pain that requires analgesia at the opioid level. Due to the potential for severe adverse effects, it is only indicated for short-term use (use should not exceed 5 days). Oxicam derivatives a)Piroxicam 2. Lipid derived Autacoids
- 105. Autacoids Inhibitors and Antagonists NSAIDs classification: Fenamate derivatives a)Mefenamate and meclofenamate Ketones a)Nabumetone COX-2 selective agents a)Celecoxib 2. Lipid derived Autacoids
- 106. Autacoids Inhibitors and Antagonists NSAIDs classification: 2. Lipid derived Autacoids
- 107. Autacoids Inhibitors and Antagonists Other anti-inflammatory drugs: Other anti-Infammatory drugs are used in the more advanced stages of some rheumatoid diseases. 1. Gold sodium Thiomalate and auranofin a. These are gold compounds that may retard the destruction of bone and joints by an unknown mechanism. b. These agents can produce serious GI disturbances, dermatitis, and mucous membrane lesions. c. Less common effects may include hematologic disorders such as aplastic anemia and proteinuria, with occasional nephrotic syndrome. 2. Lipid derived Autacoids
- 108. Physiological Regulation of Arterial Blood Pressure Maaz Bin NASIM, PhD. Assistant Professor
- 109. Objectives By the end of this session, the student should be able to: a) Outline the different mechanisms involved in regulation of ABP. b) Discuss the role of reflexes especially baroreceptor reflex in regulation of ABP. c) Discuss the role of renin-angiotensin system in regulation of ABP. d) Discuss the role of renal-body fluid in long-term regulation of ABP.
- 110. Mechanisms Involved for Regulation of Arterial Blood Pressure
- 111. Changes in mean arterial pressure after rapid hemorrhage A-return of MAP to normal by compensatory mechanism B-failure of compensatory mechanism leading to hypovolemic shock and death
- 112. Mechanisms Involved for Regulation of Arterial Blood Pressure Rapidly acting mechanism for regulation of blood pressure Mostly the nervous control mechanism • Baro receptor feed back mechanism • Central nervous system ischemic mechanism • Chemoreceptor mechanism Intermediate acting mechanism for control of blood pressure • Renin angiotensin vasoconstrictor mechanism • Stress relaxation of vasculature • Fluid shift across the capillary for adjustment of blood volume Long term mechanism for control of blood pressure • Renal blood volume pressure control mechanism
- 114. The baroreceptor system for controlling arterial pressure
- 115. Role of the Nervous System in Rapid Control of Arterial Pressure Nervous control of arterial pressure is by far the most rapid of all our mechanisms for pressure control. When there is drop in arterial blood pressure there are three major changes that occur simultaneously, each of which helps to increase arterial pressure. They are as follows 1. Most arterioles of the systemic circulation are constricted. 2. The veins especially (but the other large vessels of the circulation as well) are strongly constricted. This displaces blood out of the large peripheral blood vessels toward the heart, thus increasing the volume of blood in the heart chambers. 3. Finally, the heart itself is directly stimulated by the autonomic nervous system, further enhancing cardiac pumping.
- 116. Reflex Mechanisms for Maintaining Normal Arterial Pressure
- 117. Reflex Mechanisms for Maintaining Normal Arterial Pressure Baroreceptor reflex •It is the baroreceptor arterial pressure control system and it is the best known nervous mechanism for control of arterial pressure. •Basically, this reflex is initiated by stretch receptors, called either baroreceptors or pressoreceptors, located at specific points in the walls of several large systemic arteries. •Baroreceptors are spray-type nerve endings that lie in the walls of the arteries; they are stimulated when stretched. They are extremely abundant in (1) the wall of each internal carotid artery slightly above the carotid bifurcation, an area known as the carotid sinus, and (2) the wall of the aortic arch, the aortic sinus.
- 118. •The baroreceptors respond much more to a rapidly changing pressure than to a stationary pressure. •Carotid sinus <Hering’s nerve<Glossopharyngeal nerve<NTS in the medulla; Aortic sinus < vagus nerve< NTS in medulla •Excitation of the baroreceptors by high pressure in the arteries reflexly causes the arterial pressure to decrease because of both a decrease in peripheral resistance and a decrease in cardiac output. Conversely, low pressure has opposite effects, reflexly causing the pressure to rise back toward normal. •Function of the Baroreceptors -During Changes in Body Posture •Because the baroreceptor system opposes either increases or decreases in arterial pressure, it is called a pressure buffer system and the nerves from the baroreceptors are called buffer nerves. •The long-term regulation of mean arterial pressure by the baroreceptors requires interaction with additional systems, principally the renal-body fluid-pressure control system (along with its associated nervous and hormonal mechanisms).
- 119. Changes in mean aortic pressure in response to 8% blood loss in three groups of individual (2)
- 120. Control of Arterial Pressure by the Carotid and Aortic Chemoreceptors-chemoreceptor reflex •Whenever the arterial pressure falls below a critical level, the chemoreceptors become stimulated because diminished blood flow causes decreased oxygen, as well as excess buildup of carbon dioxide and hydrogen ions that are not removed by the slowly flowing blood. The signals transmitted from the chemoreceptors excite the vasomotor center, and this elevates the arterial pressure back toward normal. •However, this chemoreceptor reflex is not a powerful arterial pressure controller until the arterial pressure falls below 80 mm Hg. Therefore, it is at the lower pressures that this reflex becomes important to help prevent further decreases in arterial pressure
- 121. Atrial and Pulmonary Artery Reflexes Regulate Arterial Pressure Both the atria and the pulmonary arteries have in their walls stretch receptors called low-pressure receptors These low-pressure receptors play an important role, especially in minimizing arterial pressure changes in response to changes in blood volume They do detect simultaneous increases in pressure in the low- pressure areas of the circulation caused by increase in volume, and they elicit reflexes parallel to the baroreceptor reflexes to make the total reflex system more potent for control of arterial pressure
- 122. Atrial Reflexes That Activate the Kidneys-The "Volume Reflex." •Stretch of the atria also causes significant reflex dilation of the afferent arterioles in the kidneys. Signals are also transmitted simultaneously from the atria to the hypothalamus to decrease secretion of antidiuretic hormone (ADH). Combination of these two effects-increase in glomerular filtration and decrease in reabsorption of the fluid-increases fluid loss by the kidneys and reduces an increased blood volume back toward normal •Atrial stretch caused by increased blood volume also elicits a hormonal effect on the kidneys-release of atrial natriuretic peptide-that adds still further to the excretion of fluid in the urine and return of blood volume toward normal
- 123. Atrial Reflex Control of Heart Rate (the Bainbridge Reflex) •An increase in atrial pressure also causes an increase in heart rate, sometimes increasing the heart rate as much as 75 percent •The stretch receptors of the atria that elicit the Bainbridge reflex transmit their afferent signals through the vagus nerves to the medulla of the brain. Then efferent signals are transmitted back through vagal and sympathetic nerves to increase heart rate and strength of heart contraction. Thus, this reflex helps prevent damming of blood in the veins, atria, and pulmonary circulation Central Nervous System Ischemic Response This arterial pressure elevation in response to cerebral ischemia is known as the central nervous system (CNS) ischemic response. It is one of the most powerful of all the activators of the sympathetic vasoconstrictor system
- 124. The Renin-Angiotensin System: Its Role in Arterial Pressure Control
- 125. Components of the Renin-Angiotensin System Renin is synthesized and stored in an inactive form called prorenin in the juxtaglomerular cells (JG cells) of the kidneys. The JG cells are modified smooth muscle cells located in the walls of the afferent arterioles immediately proximal to the glomeruli. When the arterial pressure falls, intrinsic reactions in the kidneys themselves cause many of the prorenin molecules in the JG cells to split and release renin. Most of the renin enters the renal blood and then passes out of the kidneys to circulate throughout the entire body
- 128. Angiotensin II is an extremely powerful vasoconstrictor, and it also affects circulatory function in other ways as well. During its persistence in the blood, angiotensin II has two principal effects that can elevate arterial pressure. i. The first of these, vasoconstriction in many areas of the body, occurs rapidly. Vasoconstriction occurs intensely in the arterioles and much less so in the veins. Constriction of the arterioles increases the total peripheral resistance, thereby raising the arterial pressure. ii. The second principal means by which angiotensin II increases the arterial pressure is to decrease excretion of both salt and water by the kidneys. This long-term effect, acting through the extracellular fluid volume mechanism, is even more powerful than the acute vasoconstrictor mechanism in eventually raising the arterial pressure.
- 129. Angiotensin II causes the kidneys to retain both salt and water in two major ways: 1. Angiotensin II acts directly on the kidneys to cause salt and water retention. 2. Angiotensin II causes the adrenal glands to secrete aldosterone, and the aldosterone in turn increases salt and water reabsorption by the kidney tubules. Thus both the direct effect of angiotensin on the kidney and its effect acting through aldosterone are important in long- term arterial pressure control. However, research has suggested that the direct effect of angiotensin on the kidneys is perhaps three or more times as potent as the indirect effect acting through aldosterone-even though the indirect effect is the one most widely known.
- 131. Delayed Compliance in a Venous Segment
- 132. Delayed compliance (Stress relaxation) of vessels The principle of delayed compliance is the mechanism by which a blood vessels attempts to return back to its original pressure when it is loaded with blood or blood is withdrawn from it , this is a property of smooth muscles and is exhibited by blood vessels as well as hollow viscera like urinary bladder When a segment of a vein is exposed to increased volume , then immediately its pressure increases but after some time the pressure returns back to normal due to stretching of the vessel wall, similarly after drop in original volume due to any fluid loss the blood pressure decreases for some time and then it returns back to normal due to changes in the arrangement of smooth muscle. Similar phenomenon can be seen in urinary bladder.
- 133. Fluid shift across the capillary for adjustment of blood volume
- 134. •The arterial hypotension, arteriolar constriction, and reduced venous pressure during hemorrhagic hypotension lower hydrostatic pressure in the capillaries. The balance of these forces promotes the net reabsorption of interstitial fluid into the vascular compartment •Considerable quantities of fluid may thus be drawn into the circulation during hemorrhage. About 0.25 mL of fluid per minute per kilogram of body weight may be reabsorbed by the capillaries. Thus, approximately 1 L of fluid per hour might be autoinfused from the interstitial spaces into the circulatory system of an average individual after acute blood loss •Substantial quantities of fluid may shift slowly from the intracellular to the extracellular space. This fluid exchange is probably mediated by secretion of cortisol from the adrenal cortex in response to hemorrhage. Cortisol appears to be essential for the full restoration of plasma volume after
- 135. Role of renal-body fluid control mechanism in long-term regulation of ABP
- 136. An increase in arterial pressure in the human of only a few mm Hg can double renal output of water, which is called pressure diuresis, as well as double the output of salt, which is called pressure natriuresis.
- 137. Control of renal NaCl and water excretion Renal Sympathetic Nerves (↑ Activity: ↓ NaCl Excretion) ↓GFR ↑ Renin secretion ↑ Na+ reabsorption along the nephron Renin-Angiotensin-Aldosterone (↑ Secretion: ↓ NaCl Excretion) ↑ Angiotensin II stimulates reabsorption of Na+ along the nephron ↑ Aldosterone stimulates Na+ reabsorption in the thick ascending limb of Henle's loop, distal tubule, and collecting duct ↑ Angiotensin II stimulates secretion of ADH Natriuretic Peptides: ANP, BNP, and Urodilatin (↑ Secretion: ↑ NaCl Excretion) ↑ GFR ↓ Renin secretion ↓ Aldosterone secretion (indirect via ↓ in angiotensin II and direct on the adrenal gland) ↓ NaCl and water reabsorption by the collecting duct ↓ ADH secretion and inhibition of ADH action on the distal tubule and collecting duct ADH (↑ Secretion: ↓ H O Excretion)
- 138. Renal Urinary Output Curve The approximate average effect of different arterial pressure levels on urinary volume output by an isolated kidney, demonstrating markedly increased urine volume output as the pressure rises. This increased urinary output is the phenomenon of pressure diuresis. Not only does increasing the arterial pressure increase urine volume output, but it causes approximately equal increase in sodium output, which is the
- 139. Increases in cardiac output, urinary output, and arterial pressure caused by increased blood volume in dogs whose nervous pressure control mechanisms had been blocked. This figure shows return of arterial pressure to normal after about an hour of fluid loss into the urine
- 140. Near infinite feedback gain This return of the arterial pressure always back to the equilibrium point is the near infinite feedback gain principle for control of arterial pressure by the renal-body fluid mechanism.
- 141. Two ways in which the arterial pressure can be increased: A, by shifting the renal output curve in the right- hand direction toward a higher pressure level or B, by increasing the intake level of salt and water
- 142. Summary a) Outline the different mechanisms involved in regulation of ABP. b) Discuss the role of reflexes especially baroreceptor reflex in regulation of ABP. c) Discuss the role of renin-angiotensin system in regulation of ABP. d) Discuss the role of renal-body fluid in long-term regulation of ABP.
- 143. thanks
- 150. Cardiovascular Pharmacology Classification of Drugs Used in management of CVS pathologies 1. Angiotensin converting enzyme inhibitors Captopril Enalapril Lisinopril Ramipril Mechanism of Action: ACE (angiotensin converting enzyme) cleaves inactive angiotensin I to active angiotensin II which is a potent vasoconstrictor. It is also involved in breakdown of bradykinin, a peptide that increases production of nitric oxide and prostacyclin, both of which are potent vasodilators. ACE inhibitors competitively inhibit ACE and inhibit conversion of angiotensin I to angiotensin II.
- 151. Cardiovascular Pharmacology It results in prevention of: • Pressor effect of angiotensin II. • Stimulation of aldosterone synthesis and release resulting in decreased sodium and water retention. • Metabolism of bradykinin by ACE. Thus, vasodilation of both arterioles and veins occur as a result of decreased vasoconstriction (due to decreased angiotensin II) and enhanced vasodilation (due to increased bradykinin).
- 153. Cardiovascular Pharmacology Classification of Drugs Used in management of CVS pathologies 1. Angiotensin converting enzyme inhibitors Captopril Enalapril Lisinopril Ramipril
- 154. Cardiovascular Pharmacology Classification of Drugs Used in management of CVS pathologies Angiotensin converting enzyme inhibitors Adverse effects These agents have the potential to cause a dry, nonproductive cough, which resolves when therapy is discontinued (mostlikely due to increased bradykinin levels). Angioedema (life-threatening airway swelling and obstruction) is rare but can occur at any time, especially after the first dose (most likely due to increased bradykinin levels). Hyperkalemia is also a common adverse effect due to reduced aldosterone levels.
- 155. Cardiovascular Pharmacology Classification of Drugs Used in management of CVS pathologies Angiotensin converting enzyme inhibitors Adverse effects Modest reductions in glomerular filtration rate (GFR) and small increases in serum creatinine) may Occur with ACE inhibitors. Hypotension and dizziness are also potential adverse effects. Contraindications: Drugs that act on the renin-angiotensin system are teratogenic and should be discontinued in pregnancy.
- 156. Cardiovascular Pharmacology Classification of Drugs Used in management of CVS pathologies 2. Angiotensin receptor blockers Losartan Candesartan Valsartan Telmisartan Mechanism of Action: These agents bind to the AT1 angiotensin II receptor and prevent angiotensin II from binding; this blocks the effects of angiotensin II, including aldosterone release and vasoconstriction.
- 157. Cardiovascular Pharmacology Classification of Drugs Used in management of CVS pathologies 2. Angiotensin receptor blockers Losartan Candesartan Valsartan Telmisartan Mechanism of Action: AT1 receptors are coupled to the Gq protein and IP3 signal transduction pathway. ARBs have similar effects to ACE inhibitors, including reduced preload and afterload and decreased cardiac remodelling. Bradykinin levels are not increased since ARBs do not inhibit ACE.
- 158. Cardiovascular Pharmacology Classification of Drugs Used in management of CVS pathologies
- 159. Cardiovascular Pharmacology Classification of Drugs Used in management of CVS pathologies
- 160. Cardiovascular Pharmacology 3.Diuretics Carbonic anhydrase Inhibitors Acetazolamide, Dorzolamide, Brinzolamide Loop diuretics Furosemide, bumetanide, Torsemide, Ethacrynic acid Thiazide diuretics Hydrochlorothiazide, Chlorothiazide, chlorthalidone (thiazide-like) K+ sparing diuretics Spironolactone, eplerenone, Amiloride, triamterene
- 161. Cardiovascular Pharmacology 3.Diuretics SGLT2 Inhibitors Canagliflozin, dapagliflozin, Empagliflozin. Osmotic diuretics Mannitol ADH antagonists Conivaptan, Tolvaptan, Demeclocycline