Embed presentation
Download to read offline












































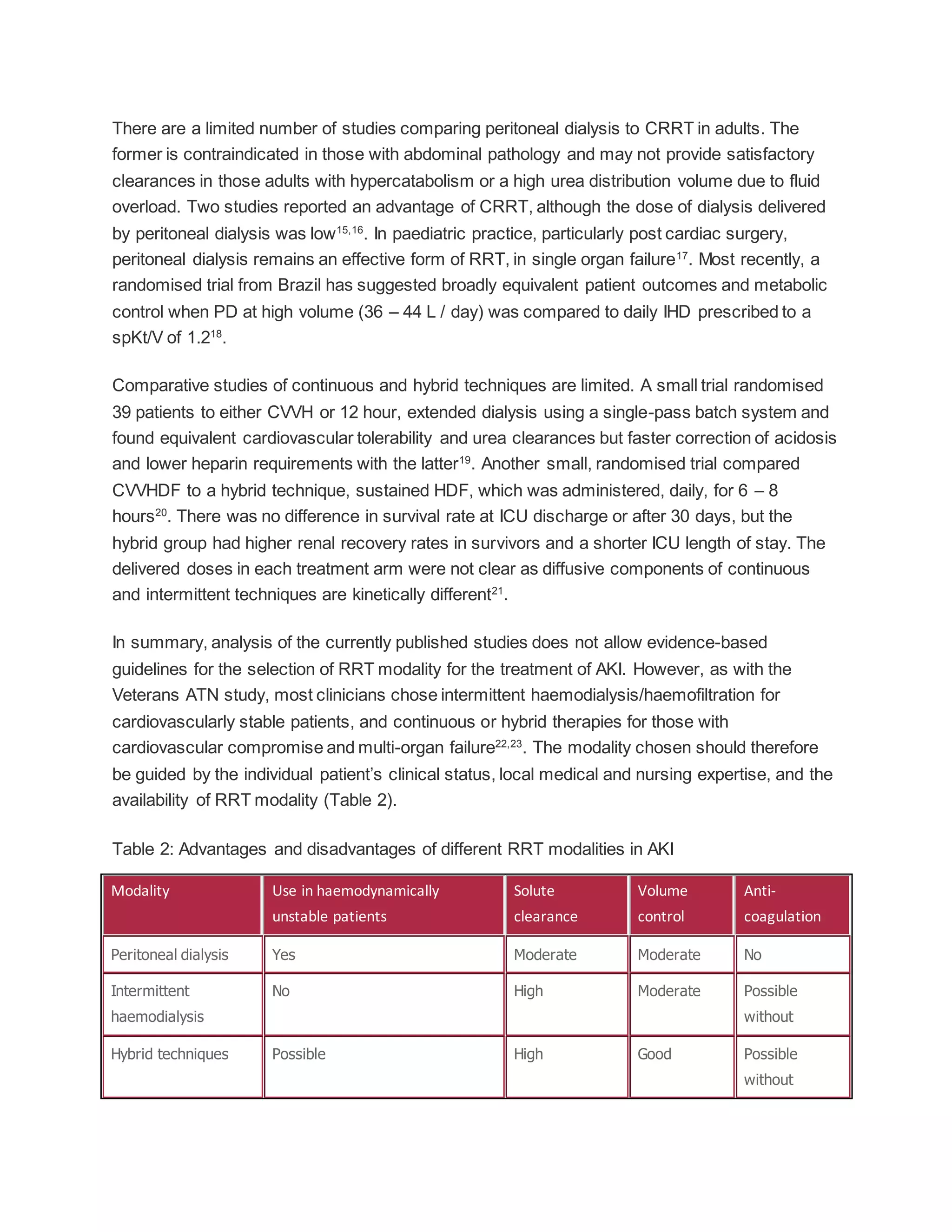
























This document provides clinical practice guidelines for the management of Acute Kidney Injury (AKI). It discusses the definition and staging systems for AKI, epidemiology and outcomes. Prevention, management, treatment facilities and timing of renal replacement therapy are covered. Guidelines are provided on choice of renal replacement modality, dialyser membranes, vascular access, anticoagulation, and prescription of renal replacement therapy. There is a lack of evidence to guide optimal care and timing of renal replacement therapy. The document aims to standardize care and improve outcomes of patients with AKI.




































































