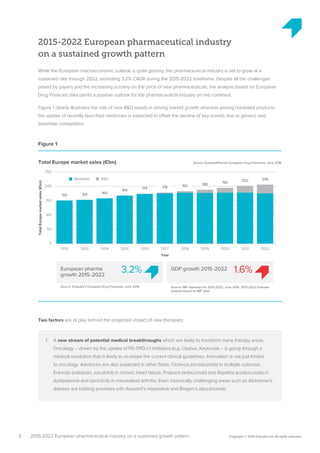
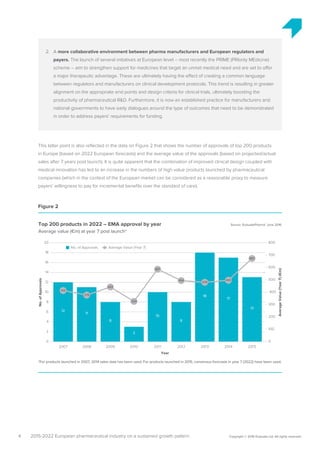
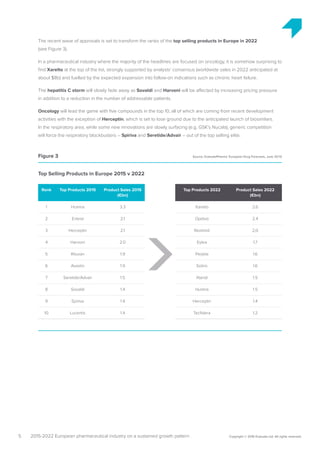
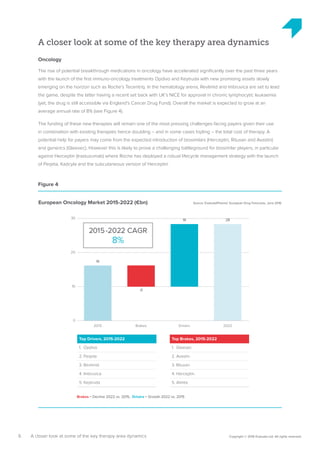
The pharmaceutical industry in Europe is expected to grow at an annual rate of 3.2% from 2015-2022, driven by new innovations emerging across therapy areas like oncology and immunology. While oncology drugs will account for much of the growth, the top-selling drug in Europe in 2022 is predicted to be Xarelto rather than a cancer drug. Five of the top 10 drugs will treat cancer. However, funding all these new high-cost therapies may challenge healthcare systems. Pharmaceutical companies and payers will need to collaborate more closely on new funding models to support continued innovation.