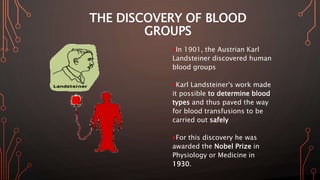
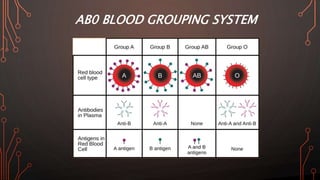
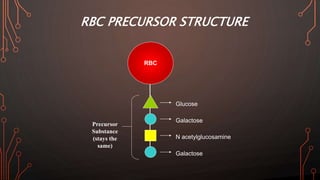
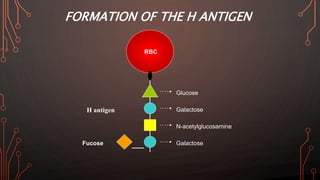
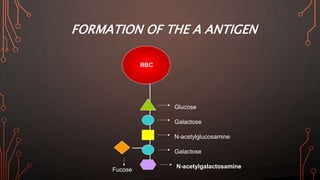
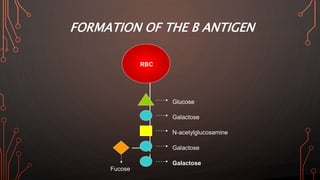
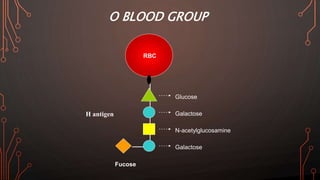
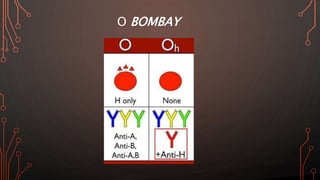
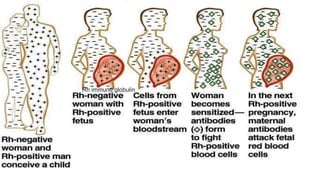
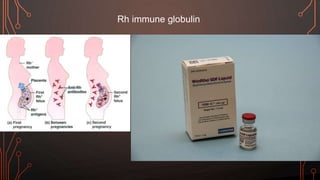
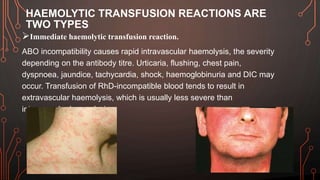
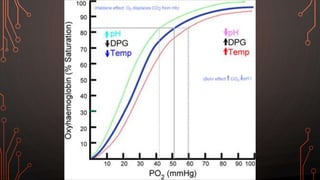
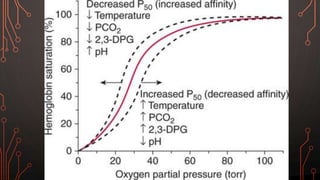
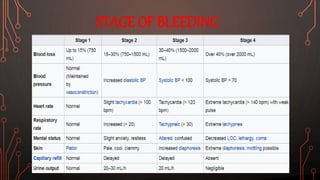
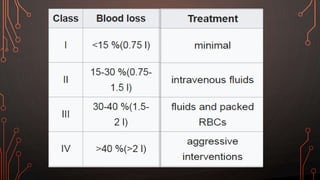
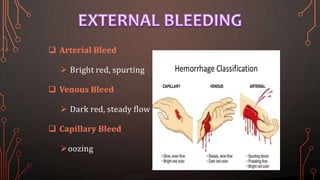
Karl Landsteiner discovered the ABO blood group system in 1901, allowing safe blood transfusions. There are four main blood groups - A, B, AB, and O - determined by antigens on red blood cells. Group O is the universal donor as it lacks A and B antigens. The Rh system was also discovered, with Rh+ and Rh- types important for transfusions. Improper blood matching can cause haemolytic transfusion reactions from antibody-mediated hemolysis. Massive transfusions carry risks like hypothermia, coagulopathy, hypocalcaemia, and hyperkalaemia that require careful management.