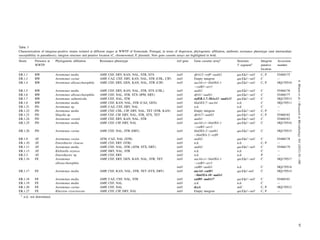
This study analyzed the occurrence and diversity of integrons in bacteria isolated from an urban wastewater treatment plant. A total of 697 isolates of Enterobacteriaceae and Aeromonas were screened for integrons. Three new gene cassettes were identified, including a novel aadA variant and genes involved in cell signaling and unknown functions. Thirteen different gene cassette arrays were detected, with four representing novel integrons. Approximately 80% of isolates were resistant to at least 3 antibiotic classes. The presence of novel integron structures in treated effluent suggests wastewater treatment plants may facilitate the formation and spread of antibiotic resistance genes.