Antiviral drugs
- 2. Introduction to the Viruses General properties of viruses: 1-They are very small in size, from 20-300 m. 2-They contain one kind of nucleic acid (RNA or DNA) as their genome. 3-They are metabolically inert because they do not possess enzyme systems necessary for the synthesis of new viral proteins. 4-so they are called as obligate intracellular parasites as they replicate inside living cells. 5-They are only seen by electron microscope. 2
- 3. Classification of Viruses Classical virus classification schemes have been based on the consideration of major properties of viruses: 1- The type of nucleic acid which is found in the virion (RNA or DNA, single stranded or double stranded) 2-The symmetry and shape of the capsid (Cubic, helical, complex). 3- The presence or absence of an envelope (enveloped, naked) 4- The size of the virus particle. 5- Antigenic properties . 6- Biologic properties, including natural host range, mode of transmission, vector relationship, pathogenicity etc. 3
- 4. Classification of Viruses: DNA Icosahedral Complex Pox viruses Naked Enveloped Papilloma virus Herpes viruses Hepatitis B 4
- 6. Viruses are different from other Microbes A virus cannot replicate on its own It must attach and enter a host cell Then the virus, uses the host cell’s energy to synthesize their DNA, RNA and protein. Viruses are difficult to kill because they live inside the cells Any drug that kills a virus may also kill cells (host) 6
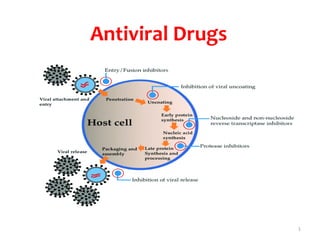
- 8. Viral replication: Steps of viral replication: 1. Recognization 2- Attachment/Adsorption: Virus attaches to the cell surface of the host. 3- Penetration by fusion (Entry): Some virus inject their nucleic acid through the cell membrane. The viral envelope of some virions fuses with the plasma membrane and the nucleocapsid is then introduced into the cell. Others enter the cell intact and are then uncoated (the virions are taken into the cell by endocytosis where the cell membrane wraps itself round the virion and is then pinched off to produce a vesicle called an endosome) 8
- 9. 4- Uncoating: Uncoating is the physical separation of viral nucleic acid from the outer structural components. This is done by lysosomes of host. 5- Transcription 6. protein synthesis & 7. Replication. Early genes takes over the host cell such that viral DNA and/or RNA is synthesized depending on the type of virus. 5- Synthesis of viral components: This involves the synthesis of viral proteins and viral genomes. 8- Assembly: late genes direct the synthesis of capsid & Viral nucleic acid is then taken into the capsid to form the nucleocapsid. 9- Release: Virus may be released from the host cell to attack other host cell. 9
- 10. Anti-Viral drugs Key characteristics of antiviral drugs Drugs used in treatment of viral infection are called antiviral drugs Many antiviral drugs are Purine or Pyrimidine analogs. Many antiviral drugs are Prodrugs. So, these drugs phosphorylated by viral cellular enzymes in order to become active. Anti-viral agents inhibits active replication. 10
- 11. Anti-viral how they act? Some drugs interfere with ability of virus to bind to cells. Drugs are able to enter the cells infected with virus and Interfere with viral nucleic acid synthesis and/or regulation. Some drugs stimulate the body’s immune system. A healthy immune system works synergistically with the drugs to eliminate or suppress viral activity 11
- 12. Classification of Antiviral Drugs CLASSES DRUGS 1. Anti-Herpes virus Idoxuridine, Trifluridine Acyclovir, Valacyclovir, Famciclovir Ganciclovir, Valganciclovir Cidofovir Foscarnet Fomivirsen 2. Anti-influenza virus Amantadine, Rimantadine Oseltamivir, Zanamivir 3. Anti-Hepatitis Lamivudine, Adefovir dipivoxil, Tenofovir Primarily for Hepatitis C Ribavirin, Interferon α 12
- 13. CLASSES DRUGS 4. Anti-retrovirus a. Nucleoside reverse transcriptase inhibitors (NRTIs) Zidovudine (AZT), Didanosine, Stavudine Lamivudine, Abacavir, Emtricitabine Tenofovir (Nt RTI) b. Non-nucleoside reverse transcriptase inhibitors (NNRTIs) Nevirapine, Efavirenz, Delavirdine c. Protease inhibitors Ritonavir, Atazanavir, Indinavir, Nelfinavir Saquinavir, Amprenavir, Lopinavir d. Entry (Fusion) inhibitor Enfuvirtide e. CCR5 receptor inhibitor Maraviroc f. Integrase inhibitor Raltegravir 13
- 14. Anti-Herpes virus Herpes virus is DNA virus, it causes various diseases such as cold sores, genital herpes, chicken pox, shingles, eye diseases, mononucleosis, Burkitt’s lymphoma and Kaposi’s sarcoma. Nucleoside analogues have been particularly effective Eg of drugs Purine Antagonist: Acyclovir, Vidarabine, Ganciclovir, Valacyclovir Pyrimidine Antagonist Cytarabine, Idoxuridine, Trifluorothymidine Phosphorus Derivatives: Foscarnet All these drugs are Inhibitors of viral DNA polymerase 14
- 15. Acyclovir was discovered and was introduced into the market in 1981. Acyclovir has a nucleoside-like structure and contains the same nucleic acid base as deoxyguanosine(GTP) However, it lacks the complete sugar ring. In virally infected cells, the drug is phosphorylated in three stages to form a triphosphate which is the active agent. so aciclovir is a prodrug Acyclovir (Zovirax) 15 Active form of drug
- 16. Acyclovir - MOA Step 1: Activation Inhibits DNA-polymerase irreversibly Aciclovir is a selectively up taken by infected cells & converted to the active triphosphate in infected cells. So it is prodrug Viral thymidine kinase is 100 times more effective for converting acyclovir to its monophosphate than host cell thymidine kinase. Therefore, An advantage is that Aciclovir doesn’t enter into uninfected human cells. 16
- 17. Acyclovir - MOA Step 2: Incorporation into growing DNA chain Gets incorporated in viral DNA As the sugar unit is incomplete and lacks the required -OH group normally present at position 3′ of the sugar ring, the nucleic acid chain cannot extend any further. Thus the drug act as chain terminator 17
- 18. Si HN Si H3C CH3 CH3 CH3 CH3 hexamethyl disilazane N N N H N OH H2N CH3 N N N H N O HN Si H3C CH3 CH3 Si H3C CH3 CH3 H2C CH2 O CH2 Cl O O 1-benzoyloxy-2-chloromethoxyethane N N N N O HN Si H3C CH3 CH3 Si H3C CH3 CH3 H2C CH2 O CH2 O O N N N N OH H2N H2C CH2 O CH2 OH MEOH/NH3 Synthesis of acyclovir Guanine 2-amino,6-hydroxr-9-(2-hydroxyethoxymethyl)-3H-purine 18
- 19. The oral bioavailability of acyclovir is quite low (15–30%). To overcome this, various prodrugs were developed to increase water solubility. Valaciclovir is an l-valyl ester prodrug absorbed from the gut far more effectively than acyclovir. Once valaciclovir is absorbed, it is hydrolysed to acyclovir in the liver and gut wall. Desciclovir is a prodrug of acyclovir which lacks the carbonyl group at position 6 of the purine ring and is more water soluble. In the body, it is metabolised by cellular xanthine oxidase to oxidizes the 6-position to give acyclovir. 19
- 20. Some viruses lack the enzyme thymidine kinase. As a result, phosphorylation fails to take place. Cidofovir was designed to combat this problem It is an analogue of deoxycytidine 5-monophosphate where the sugar and phosphate groups have been replaced by an acyclic group and a phosphonomethylene group . The latter group acts as a bioisostere for the phosphate Susceptible for enzymatic hydrolysis Resistant for enzymatic hydrolysis 20
- 21. The following drugs are phosphorylated equally by viral & cellular thymidine kinase, so there is less selectivity and more toxicity. Foscarnet has difficulty in crossing cell membrane due to high charge 21 Idoxuridine Trifluridine Vidarabine Foscarnet
- 22. Foscarnet (phosphonomethanoic acid) 22 Foscarnet is a structural mimic of the anion pyrophosphate that selectively inhibits the pyrophosphate binding site on viral DNA polymerases at concentrations that do not affect human DNA polymerases. Unlike acyclovir and ganciclovir, foscarnet is not activated by viral protein kinases, making it useful in acyclovir- or ganciclovir-resistant HSV and CMV infections Foscarnet has difficulty in crossing cell membrane due to high charge. Nephrotoxicity, Electrolyte disturbances, Genital ulceration CNS — paresthesia, irritability and hallucinations.
- 23. Therapeutic uses of Acyclovir Acyclovir is the drug of choice for: HSV Genital infections, encephalitis & Varicella zoster virus (VZV) HSV infections in immunocompromised patient Ganciclovir is the drug of choice for: CMV retinitis in immunocompromised patient Prevention of CMV disease in transplant patients Cytomegalovirus retinitis, also known as CMV retinitis, is an inflammation of the retina of the eye that can lead to blindness. Caused by cytomegalovirus, it occurs predominantly in people whose immune system has been compromised, 15-40% of those infected with AIDS. Valaciclovir is a valine prodrug of aciclovir and is particularly useful in the treatment of VZV infections. 23 CMV retinitis
- 24. Cidofovir (a nucleotide analog of cytosine) & foscarnet • It is approved for the treatment of CMV retinitis in immuno compromised patients. Vidarabin is used in Herpes Simplex Virus Kerato - conjunctivitis. Therapeutic uses Conti…… 24 Herpes Simplex
- 25. Adamantanes Amantadine and Rimantadine is used as Anti-influenza virus Amantadine and Rimantadine are both drugs that interfere with penetration of host cells by viruses and block early stage replication. Amantadine 1-adamantanamine hydrochloride (Symmetrel). Rimantadine, it is methyl-1-adamantane derivative These are unusual caged tricyclic amines with the following structures: 25
- 26. Amantadine has been used for years as a treatment for Parkinson disease. Both of these agents will specifically inhibit replication of the influenza type A viruses at low concentrations. Rimantadine is generally 4 to 10 times more active, less toxic than amantadine. The adamantanamines have two mechanisms in common: (a) they inhibit an early step in viral replication, most likely viral uncoating,and (b) in some strains, they affect by interfering with haemagglutinin (A viral protein, which causes haemagglutination)processing. 26
- 27. Synthesis of Amantadine 27 Adamantane 1-bromo adamantane 1-acetylamino adamantane Amantadine Ritter Reaction: Acetonitrile in presence of sulphuric acid
- 28. Vidarabine It is adenosine derivative Chemically, vidarabine (Vira-A), is 9—D arabinofuranosyladenine. Introduced in 1960 as a candidate anticancer agent, vidarabine was found to have broad- spectrum activity against DNA viruses. Vidarabine is also used to treat herpes zoster in AIDS patients A 3% ophthalmic ointment Vira-A is used in the treatment of acute keratoconjunctivitis and recurrent superficial keratitis caused by HSV-1 and HSV-2. 28
- 29. HSV encephalitis. Although the agent was initially prepared chemically, it is now obtained by fermentation with strains of Streptomyces antibioticus. Mechanism of Action The antiviral action of vidarabine is completely confined to DNA viruses. Vidarabine inhibits viral DNA synthesis by blocking viral DNA polymerase. Vidarabine triphosphate is also incorporated into cellular and viral DNA, where it acts as a chain terminator. 29
- 30. Anti-HIV Drugs 30 The red ribbon, as an awareness ribbon, is used as the symbol for the solidarity of people living with HIV/AIDS, and for the awareness and prevention of drug abuse and drunk driving.
- 31. HIV HIV = Human Immunodeficiency Virus (Causative organism) Destroys CD4 cells (T-cells and macrophages) AIDS = Acquired Immunodeficiency Virus (Disease) (~10 years after infection) HIV-1 = Most common HIV-2 = relatively uncommon and less infectious *http://en.wikipedia.org/wiki/Aids#Diagnosis 31
- 32. When a person with HIV is considered to have progressed to AIDS ? A person with HIV is considered to have progressed to AIDS when: The number of their CD4 cells falls below 200 cells per cubic millimeter of blood (200 cells/mm3). (In a healthy immune system, CD4 counts are between 500 and 1,600 cells/mm3.) OR They develop one or more opportunistic infections regardless of their CD4 count. 32
- 33. The Life Cycle of HIV • Free Virus • Binding and Fusion • Infection • Reverse Transcription • Integration • Transcription • Assembly • Budding • Maturation 33
- 34. 34 HIV surface protein gp120 co-receptor gp41 The binding of gp120 to a coreceptor either CCR5 or CXCR4. Virus penetration of the cell membrane by gp41: fusion. Binding, fusion, entry sequence
- 35. Binding, fusion, entry sequence HIV entry into a human cell requires the following steps in sequence. The binding of HIV surface protein gp120 to the CD4 receptor (Host) A conformational change in gp120, which both increases its affinity for a co-receptor and exposes gp41 The binding of gp120 to a co-receptor either CCR5 or CXCR4 The penetration of the cell membrane by gp41, which approximates the membrane of HIV and the T cell and promotes their fusion. The entry of the viral core into the cell Entry inhibitors work by interfering with one aspect of this process. 35
- 36. Reverse transcriptase/DNA polymerase Retroviruses possess DNA polymerase (Reverse transcriptase) that directs the synthesis of a DNA copies from the viral RNA. The formed DNA is duplicated, circularized, and incorporated into the DNA of an infected cell. RNA DNA Reverse transcriptase /DNA Polymerase 36
- 37. Classification of Anti-drugs HAART Entry Inhibitors Reverse Transcriptase Inhibitors (RTIs) Protease Inhibitors Nucleoside/ Nucleotide RTIs (NRTIs) Non-Nucleoside RTIs (NNRTIs) (Highly Active Anti Retroviral Therapy) 37
- 38. Anti HIV DRUGS i)Nucleoside Reverse Transcriptase Inhibitors(NRTIs) NRTIs block reverse transcriptase, an enzyme HIV needs to make copies of itself. Zidovudine Lamivudine Stavudine Abacavir Didanosine Tenofovir (component of atripla) Emtricitabine (component of atripla) 38
- 39. ii)Non-Nucleoside Reverse Transcriptase Inhibitors (NNRTIs) They don't have Nucleic acid structure and target reverse transcriptase but act different way as NRTIs Efavirenz, (component of atripla) Nevirapine, Rilpivirine iii) Protease Inhibitors (PIs), which target an HIV protein called protease. (maturing of proteins) Darunavir, Indinavir, Nelfinavir, Ritonavir, (now used in treatment of corona virus) Saquinavir 39
- 40. iv)Fusion inhibitors block HIV from entering the CD4 cells of the immune system/ Entry Inhibitors Enfuvirtide Maraviroc vi)Integrase inhibitors block HIV integrase, an enzyme HIV needs to make copies of itself. Dolutegravir, Elvitegravir Raltegravir 40
- 41. Fusion Inhibitors/Entry Inhibitors Fusion inhibitors block the HIV envelope from merging with the host CD4 cell membrane (fusion). This prevents HIV from entering the CD4 cell. Eg of drugs Enfuvirtide Maraviroc 41
- 42. Maraviroc (Selzentry) 1st oral entry inhibitor Maraviroc works by targeting CCR5, a co-receptor located on human helper T-cells. Maraviroc can cause serious, life- threatening side effects. These include liver problems, skin reactions, and allergic reactions 42 It also appeared to reduce graft-versus-host disease in patients treated with allogeneic bone marrow transplantation for leukemia,
- 43. Fusion Inhibitors/Entry Inhibitors (Enfuviritide) 43 Enfuvirtide is a peptide drug that must be injected and acts by interacting with the N-terminal heptad repeat of gp41 of HIV to form an inactive hetero six-helix bundle, therefore preventing infection of host cells
- 44. Reverse Transcriptase Inhibitors: NRTIs Tenofovir •Competitive inhibitors •No effect on host enzymes 44 Zidovudine Stavudine Emtricitabine
- 45. The enzyme Reverse Transcriptase is unique to HIV, it serves as an ideal drug target. These drugs have nucleoside-like structures. These drugs are phosphorylated by three cellular enzymes (not viral kinases) to form an active nucleotide triphosphate. Zidovudine was developed originally as an anticancer agent but was the first drug to be approved for use in the treatment of AIDS. It is an analogue of deoxythymidine where the sugar 3′- OH group has been replaced by an azido group. On conversion to the triphosphate, it inhibit Rtase. Since the sugar unit has an azide substituent at the 3′ position of the sugar ring, the nucleic acid chain cannot be extended any further 45
- 46. Zidovudine 3’-azido-3’-deoxythymidine or AZT, is an analog of thymidine that possesses antiviral activity against HIV-1, HIV-2. Lamivudine & Emtricitabine are deoxycytidine where the 3’ carbon has been replaced by Sulphur 46 Thymidine Natural nucleoside
- 47. The clinically useful NRTIs used against HIV and/or hepatitis B include abacavir (the only guanosine analogue), Stavudine (thymidine analogue) and zalcitabine (cytosine) NRTIs have good oral bioavailability, are bound minimally to plasma proteins, and are excreted through the kidneys. 47 Abacavir/Ziagen (ABC) Stavudine/Zerit (d4T) Thymine zalcitabine/HIVID (ddC) Cytosine Guanosine
- 48. Tenofovir It is a medication used to treat chronic hepatitis B and to prevent and treat HIV/AIDS. It is used for prevention of HIV/AIDS among those at high risk before exposure, and after a needle stick injury or other potential exposure. It is often recommended during pregnancy and appears to be safe. It is a nucleotide reverse transcriptase inhibitor and works by decreasing the ability of the viruses to replicate. 48 9-(2-Phosphonyl-methoxypropyly)adenine (PMPA) Tenofovir can be used for HIV prevention in people who are at high risk for infection through sexual transmission or injecting drug use.
- 50. Reverse Transcriptase Inhibitors: NNRTIs The NNRTIs are generally hydrophobic molecules that bind to an allosteric binding site which is hydrophobic in nature. Since the allosteric binding site is separate from the substrate binding site, the NNRTIs are non-competitive, reversible inhibitors. They include First-generation NNRTIs Nevirapine and Delavirdine Second-generation NNRTIs Efavirenz , Etravirine, Rilpivirine 50
- 51. Reverse Transcriptase Inhibitors: NNRTIs Nevirapine Delavirdine Efavirenz (component of atripla) •Only active against HIV-1 These drugs are vulnerable to resistance, rapid resistance emerges as a result of mutations in the NNRTI binding site—the most common being the replacement of Lys-103 with asparagine. This mutation is called K103N and is defined as a pan class resistance mutation. The resistance problem can be countered by combining an NNRTI with an NRTI from the start of treatment, as the binding sites are distinct. 51
- 52. 52
- 53. Protease Inhibitors (PIs) HIV protease is an enzyme that catalyze process of maturation and propagation of new virus. Inhibition of this post-translation step leads to total arrest of viral maturation & block virulence property of the virus. HIV PIs are designed to mimic the transition state of hydrolysis at the active site of proteins of HIV virus; so these drugs are called as transition state inhibitors. 53
- 54. Hydrolysis of a peptide bond proceeds through a transition state that is sp3 hybridized and, hence, tetrahedral. The analog inhibitors possess a preexisting sp3 hybridized center that will be drawn into the active site but stable to hydrolysis . So blocking viral post translation processes, maturation step & prevent the spread of cellular infection. Saquinavir (Invirase), Indinavir (Crixivan), Ritonavir (Norvir), Nelfinavir (Viracept), Amprenavir (Agenerase) have been approved for the treatment of HIV-infected patients. 54
- 55. Fosamprenavir Increased water solubility and improved oral bioavailability Metabolized to form amprenavir, which is the active ingredient Because it must be metabolized, it is time released and requires less dosages (4 instead of 16 pills per day) Possible Side Effects: Nausea, Vomiting, Diarrhea, Loose Stool, Hyperglycemia, and Fatigue 55 Fosamprenavir is a drug for the treatment of HIV infections. It is a pro-drug of drug Amprenavir.
- 56. Atripla A one-pill, once-a-day HIV-1 treatment. ATRIPLA® is a prescription medication that can be used alone as a complete regimen, or in combination with other anti-HIV- 1 medicines, to treat HIV-1 in patients who weigh at least 40 kg (88 lbs). Efavirenz(NNRTIs)/Emtricitabine/Tenofovir(both NRTIs) 56
- 57. Composition of atripla 57 Efavirenz Tenofovir disoproxil Emtricitabine
- 58. Thank You 58
