PANTOPRAZOLE IV
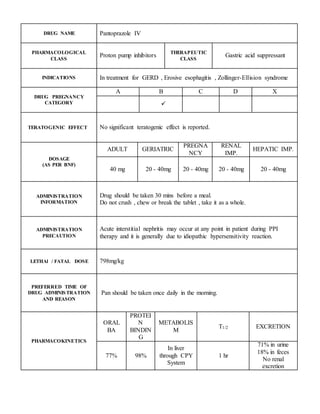
- 1. DRUG NAME Pantoprazole IV PHARMACOLOGICAL CLASS Proton pump inhibitors THERAPEUTIC CLASS Gastric acid suppressant INDICATIONS In treatment for GERD , Erosive esophagitis , Zollinger-Ellision syndrome DRUG PREGNANCY CATEGORY A B C D X TERATOGENIC EFFECT No significant teratogenic effect is reported. DOSAGE (AS PER BNF) ADULT GERIATRIC PREGNA NCY RENAL IMP. HEPATIC IMP. 40 mg 20 - 40mg 20 - 40mg 20 - 40mg 20 - 40mg ADMINISTRATION INFORMATION Drug should be taken 30 mins before a meal. Do not crush , chew or break the tablet , take it as a whole. ADMINISTRATION PRECAUTION Acute interstitial nephritis may occur at any point in patient during PPI therapy and it is generally due to idiopathic hypersensitivity reaction. LETHAl / FATAL DOSE 798mg/kg PREFERRED TIME OF DRUG ADMINISTRATION AND REASON Pan should be taken once daily in the morning. PHARMACOKINETICS ORAL BA PROTEI N BINDIN G METABOLIS M T1/2 EXCRETION 77% 98% In liver through CPY System 1 hr 71% in urine 18% in feces No renal excretion
- 2. PHARMACODYNAMICS / MOA Inhibits the action of PPI by binding to hydrogen-potassium adenosine triphosphate,located at the parietal cells and there by suppress the secretion of gastric acid. CONTRAINDICATION IN YES/NO PEDIATRIC GERIATRIC PREGNANCY LACTATION RI HI - - - - - - REASON OF CONTRAINDICATION - - - - - - MAJOR DRUG INTERACTIONS Drug 1 Drug 2 Drug 3 Drug 4 Drug 5 NAME OF THE DRUG(S) Atazanavir Ceritinib Dipyridamo le Ketoconazol e Methotrexate EFFECT OF INTERACTIONS Severe Moderate Moderate Moderate Severe MANAGEMENT Avoid or adjust the dose Change the frequency of the drug Change the frequency of the drug Change the frequency of the drug Avoid or adjust the dose ADVERSE DRUG REACTIONS (ADRS) MILD MODERATE SEVERE Confusion Depression Malaise Vision disorder Peripheral odema Agranulocytosis Hallucination Gynaecomastia ADDITIONAL INFORMATION/ PRECAUTIONS OF ADRs Monitoring liver function in hepatic disorder Patient at risk of osteoporosis should maintain a adequate intake of Ca and Vitamin D Monitor Complete blood count in patients. PREFERRED DURATION (MIN-MAX) & DESCRIPTION Pan can be administered for maximum 2 months. THERAPY TERMINATION PROCEDURE Terminate medication unless any side effects is seen in the patient. ALTERING LABORATORY PARAMETERS Electrolytes – Potassium , magnesium ; Urine test
- 3. OVER DOSE / POISON’S SYMPTOMS & EFFECT Hypoactivity , ataxia , hunched sitting , limb splay , absence of ear reflex and tremor. MANAGEMENT OF OVER DOSE Discontinue the drug and give patient supportive care.
