Efcorlin Inj drug information summary
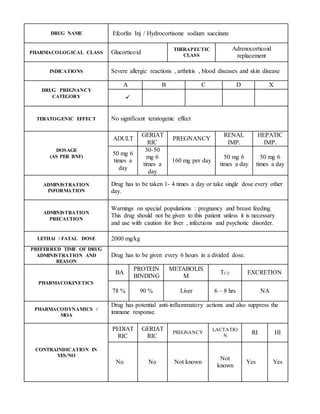
- 1. DRUG NAME Efcorlin Inj / Hydrocortisone sodium succinate PHARMACOLOGICAL CLASS Glucorticoid THERAPEUTIC CLASS Adrenocorticoid replacement INDICATIONS Severe allergic reactions , arthritis , blood diseases and skin disease DRUG PREGNANCY CATEGORY A B C D X TERATOGENIC EFFECT No significant teratogenic effect DOSAGE (AS PER BNF) ADULT GERIAT RIC PREGNANCY RENAL IMP. HEPATIC IMP. 50 mg 6 times a day 30-50 mg 6 times a day 160 mg per day 50 mg 6 times a day 50 mg 6 times a day ADMINISTRATION INFORMATION Drug has to be taken 1- 4 times a day or take single dose every other day. ADMINISTRATION PRECAUTION Warnings on special populations : pregnancy and breast feeding. This drug should not be given to this patient unless it is necessary and use with caution for liver , infections and psychotic disorder. LETHAl / FATAL DOSE 2000 mg/kg PREFERRED TIME OF DRUG ADMINISTRATION AND REASON Drug has to be given every 6 hours in a divided dose. PHARMACOKINETICS BA PROTEIN BINDING METABOLIS M T1/2 EXCRETION 78 % 90 % Liver 6 – 8 hrs NA PHARMACODYNAMICS / MOA Drug has potential anti-inflammatory actions and also suppress the immune response. CONTRAINDICATION IN YES/NO PEDIAT RIC GERIAT RIC PREGNANCY LACTATIO N RI HI No No Not known Not known Yes Yes
- 2. MAJOR DRUG INTERACTIONS Drug 1 Drug 2 Drug 3 Drug 4 Drug 5 NAME OF THE DRUG(S) Albutero l Aspirin Ibuprofen Paracetam ol Benedryl EFFECT OF INTERACTIONS Moderat e Severe Severe Mild Mild MANAGEMENT Avoid using this medicati on Stop using this medication Stop using this medication Change the frequency Taper the dose of the drug ADVERSE DRUG REACTIONS (ADRS) MILD MODERATE SEVERE Headache Sore throat Black / tarry stools Changes in vision Depression Changes in mood Delayed wound healing Convulsions Edema Severe allergic reaction ADDITIONAL INFORMATION/ PRECAUTIONS OF ADRs Not to use this medication when patient have infection which is untreated or treatment in progress Not recommended in patients having low immune power that causes decrease in blood cells This medication is not recommended for use if patient have known allergy to the drug PREFERRED DURATION (MIN-MAX) & DESCRIPTION From weeks to maximum 2 months of patient recovery THERAPY TERMINATION PROCEDURE Do not suddenly stop using it without physician knowledge ALTERING LABORATORY PARAMETERS Fluid and electrolytes imbalances : sodium , potassium , fluid retention and high blood pressure. OVER DOSE / POISON’S SYMPTOMS & EFFECT A single large dose of hydrocortisone is not expected to produce life threatening symptoms. However high doses taken can cause weight gain, roundness of face increased facial hair growth and muscle pain.
- 3. MANAGEMENT OF OVER DOSE Take this medication exactly as it was prescribed. Do not take the medication in larger amount or take it longer than recommended by the physician.
