Combined sci c12 syllabus
•Download as PPT, PDF•
0 likes•641 views
Combined Sci
Report
Share
Report
Share
More Related Content
Viewers also liked
Viewers also liked (20)
Similar to Combined sci c12 syllabus
Similar to Combined sci c12 syllabus (20)
BT19ME016 Lokeshwar Presentation on IC Engine Fuels and its required Characte...

BT19ME016 Lokeshwar Presentation on IC Engine Fuels and its required Characte...
AQA Iinternational GCSE Chapter 12 :Carbon compounds as fuels(VERSION 1)

AQA Iinternational GCSE Chapter 12 :Carbon compounds as fuels(VERSION 1)
Production of Hydrocarbons from Palm Oil over NiMo Catalyst

Production of Hydrocarbons from Palm Oil over NiMo Catalyst
Waste Automotive Oil as Alternative Fuel for IC Engine

Waste Automotive Oil as Alternative Fuel for IC Engine
FUEL ( LPG,LNG,Coal,Natural Gas, Nuclear Fuel & Fuel Cell) 

FUEL ( LPG,LNG,Coal,Natural Gas, Nuclear Fuel & Fuel Cell)
More from cartlidge
More from cartlidge (20)
Recently uploaded
“Oh GOSH! Reflecting on Hackteria's Collaborative Practices in a Global Do-It...

“Oh GOSH! Reflecting on Hackteria's Collaborative Practices in a Global Do-It...Marc Dusseiller Dusjagr
Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"

Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"National Information Standards Organization (NISO)
Recently uploaded (20)
A Critique of the Proposed National Education Policy Reform

A Critique of the Proposed National Education Policy Reform
“Oh GOSH! Reflecting on Hackteria's Collaborative Practices in a Global Do-It...

“Oh GOSH! Reflecting on Hackteria's Collaborative Practices in a Global Do-It...
Contemporary philippine arts from the regions_PPT_Module_12 [Autosaved] (1).pptx![Contemporary philippine arts from the regions_PPT_Module_12 [Autosaved] (1).pptx](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Contemporary philippine arts from the regions_PPT_Module_12 [Autosaved] (1).pptx](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
Contemporary philippine arts from the regions_PPT_Module_12 [Autosaved] (1).pptx
18-04-UA_REPORT_MEDIALITERAСY_INDEX-DM_23-1-final-eng.pdf

18-04-UA_REPORT_MEDIALITERAСY_INDEX-DM_23-1-final-eng.pdf
Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"

Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"
Combined sci c12 syllabus
- 1. 12/04/16 C12 Organic ChemistryC12 Organic Chemistry Syllabus statementsSyllabus statements Statements from Cambridge IGCSE Combined Science syllabus 0653 (for exams in 2016 – 2018)
- 2. 12.1 Fuels12.1 Fuels 1. Recall coal, natural gas and petroleum as fossil fuels that produce carbon dioxide on combustion. 2. Name methane as the main constituent of natural gas. 3. Describe petroleum as a mixture of hydrocarbons and its separation into useful fractions by fractional distillation.
- 3. 12.1 Fuels12.1 Fuels 4. Understand the essential principle of fractional distillation in terms of differing boiling points (ranges) of fractions related to molecular size and intermolecular attractive forces.
- 4. 12.1 Fuels12.1 Fuels 5. State the use of: • refinery gas for bottled gas for heating and cooking, • gasoline fraction for fuel (petrol) in cars, • diesel oil / gas oil for fuel in diesel engines.
- 5. 12.2 Hydrocarbons12.2 Hydrocarbons 1. Describe the properties of alkanes (exemplified by methane) as being generally unreactive, except in terms of burning. 2. State that the products of complete combustion of hydrocarbons, exemplified by methane, are carbon dioxide and water.
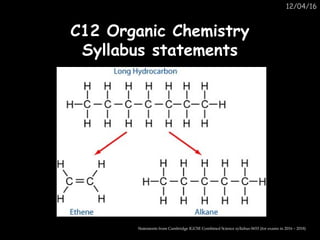
- 6. 12.2 Hydrocarbons12.2 Hydrocarbons 3. Name, identify and draw the structures of methane, ethane, ethene and ethanol. 4. Recognise the alkanes and alkenes from their chemical names or molecular structures. 5. Describe the manufacture of alkenes by cracking. 6. Distinguish between alkanes and alkenes by the addition reaction of alkenes with bromine.
- 7. 12.2 Hydrocarbons12.2 Hydrocarbons 3. Name, identify and draw the structures of methane, ethane, ethene and ethanol. 4. Recognise the alkanes and alkenes from their chemical names or molecular structures. 5. Describe the manufacture of alkenes by cracking. 6. Distinguish between alkanes and alkenes by the addition reaction of alkenes with bromine.