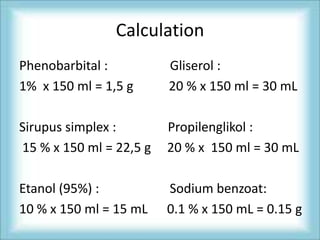
The document discusses the preparation of a phenobarbital elixir, including definitions of elixir and phenobarbital, properties and dosages of phenobarbital, additional substances used in the formulation such as preservatives and flavors, calculations for the ingredient amounts, weighing of the ingredients, and the preparation process which involves dissolving phenobarbital in ethanol and adding other solvents and ingredients to produce a 150mL elixir with 10mg/cc of phenobarbital.