New Visiongain report - Top 50 pharmaceutical CMO's market 2018
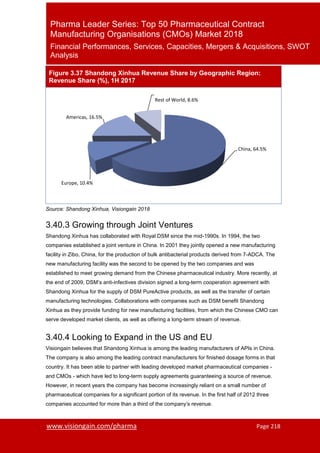
- 1. www.visiongain.com/pharma Page 218 Pharma Leader Series: Top 50 Pharmaceutical Contract Manufacturing Organisations (CMOs) Market 2018 Financial Performances, Services, Capacities, Mergers & Acquisitions, SWOT Analysis Figure 3.37 Shandong Xinhua Revenue Share by Geographic Region: Revenue Share (%), 1H 2017 3.40.3 Growing through Joint Ventures Shandong Xinhua has collaborated with Royal DSM since the mid-1990s. In 1994, the two companies established a joint venture in China. In 2001 they jointly opened a new manufacturing facility in Zibo, China, for the production of bulk antibacterial products derived from 7-ADCA. The new manufacturing facility was the second to be opened by the two companies and was established to meet growing demand from the Chinese pharmaceutical industry. More recently, at the end of 2009, DSM’s anti-infectives division signed a long-term cooperation agreement with Shandong Xinhua for the supply of DSM PureActive products, as well as the transfer of certain manufacturing technologies. Collaborations with companies such as DSM benefit Shandong Xinhua as they provide funding for new manufacturing facilities, from which the Chinese CMO can serve developed market clients, as well as offering a long-term stream of revenue. 3.40.4 Looking to Expand in the US and EU Visiongain believes that Shandong Xinhua is among the leading manufacturers of APIs in China. The company is also among the leading contract manufacturers for finished dosage forms in that country. It has been able to partner with leading developed market pharmaceutical companies - and CMOs - which have led to long-term supply agreements guaranteeing a source of revenue. However, in recent years the company has become increasingly reliant on a small number of pharmaceutical companies for a significant portion of its revenue. In the first half of 2012 three companies accounted for more than a third of the company’s revenue. China, 64.5% Europe, 10.4% Americas, 16.5% Rest of World, 8.6% Source: Shandong Xinhua, Visiongain 2018
- 2. www.visiongain.com/pharma Page 239 Pharma Leader Series: Top 50 Pharmaceutical Contract Manufacturing Organisations (CMOs) Market 2018 Financial Performances, Services, Capacities, Mergers & Acquisitions, SWOT Analysis Table 3.133 Piramal Pharma Solutions: Revenue ($m), Annual Growth Rate (%), CAGR (%), 2012-2017 2012 2013 2014 2015 2016 2017 Revenue ($m) 77.6 93.9 129.4 146.6 166.2 175.7 Annual Growth (%) 17.4% 27.4% 11.7% 11.8% 5.4% CAGR (%) 17.8% Figure 3.40 Piramal Pharma Solutions: Revenue ($m), 2012-2017 3.48.3 Investment in Technology based Delivery Platforms The Company is increasingly delivering its products and services through user-centric technology platforms. Its delivery modality has shifted dramatically from large, static research reports to digitally delivered, modular content that enables clients to obtain ‘just in time’ answers. For example, in FY2017, the Company combined 11 different legacy brands under a new, dynamic, web-based ‘Ask DRG’ Insights and Content Platform. It provides clients with an easy-to-use portal to access all of its expert forecasting, market sizing, and gold-standard epidemiology for over 140 diseases. This best-in-class technology gives its 9,000+ users both the ease of web browser-like searching and the ability to drill deep into the Company’s proprietary data sets. 0 20 40 60 80 100 120 140 160 180 200 2012 2013 2014 2015 2016 2017 Revenue($Mn) Year Source: Piramal, Visiongain 2018 Source: Piramal, Visiongain 2018
- 3. www.visiongain.com/pharma Page 243 Pharma Leader Series: Top 50 Pharmaceutical Contract Manufacturing Organisations (CMOs) Market 2018 Financial Performances, Services, Capacities, Mergers & Acquisitions, SWOT Analysis lucrative in the coming 10 years. Through a 2012 agreement with Enzon Pharmaceuticals, Zhejiang Hisun gained access to pegylation technology, for example. Growth for Zhejiang Hisun in the CMO market in the coming 10 years will be threatened by the company’s desire to expand in drug manufacturing and marketing. As well as deals signed with big pharma companies for generic product development in China, Zhejiang Hisun has licensed a number of novel pharmaceutical products from smaller drug developers. Deals include an agreement with Cleveland BioLabs, with Zhejiang Hisun gaining the exclusive rights to develop and market the synthetic lipopeptide CBLB612 in China. Table 3.135 Zhejiang Hisun Manufacturing: SWOT Analysis, 2017 Strengths Weaknesses The company exports more than 90 APIs to over 70 countries worldwide Zhejiang Hisun has a smaller product portfolio than developed market leaders The company has partnered with a number of leading big pharma clients Zhejiang Hisun is the market leader in antineoplastic and cardiovascular drug APIs; two of China’s fastest growing segments Opportunities Threats Further expansion in the US and EU through big pharma partnerships and new API approvals The company is increasingly focused on becoming a drug developer 3.50 Zhejiang Huahai Pharmaceuticals Zhejiang Huahai is much younger than the other companies discussed in this chapter. The company was founded in 1989 as a manufacturer of APIs, intermediates and formulations for the pharmaceutical industry. The company has 5,28,000 employees at eleven facilities in China and the US. Its US subsidiary was established in 2004 in New Jersey in order to provide marketing services for the North American market, as well as R&D, manufacturing and marketing for other developed markets. Zhejiang Huahai exports its products to more than 50 countries worldwide. It has a manufacturing presence in three provinces in China. Source: Visiongain 2018
