DMF - Are You Ready?
•
0 likes•49 views
Short presentation on how you should prepare for upcoming DMF submission mandates. DMF in eCTD will become mandatory. Learn how Cunesoft can help you with eCTD submission software - cune-eCTD. Check it our here: https://cunesoft.com/en/products/ectd/ or write us directly at info@cunesoft.com
Report
Share
Report
Share
Download to read offline
Recommended
More Related Content
Similar to DMF - Are You Ready?
Similar to DMF - Are You Ready? (20)
How to Prepare for an FDA Inspection and Respond to FDA 483's / Warning Letters

How to Prepare for an FDA Inspection and Respond to FDA 483's / Warning Letters
Synchrogenix Inc Capabilities Presentation Jan 2010 Final

Synchrogenix Inc Capabilities Presentation Jan 2010 Final
Submitting electronic Drug Master Files (DMF) and Active Substance Master Fil...

Submitting electronic Drug Master Files (DMF) and Active Substance Master Fil...
Product Complaints: Complaint Handling from Intake to Closure

Product Complaints: Complaint Handling from Intake to Closure
Quality & compliance excellence in pharmaceuticals

Quality & compliance excellence in pharmaceuticals
Taiwan medical device registration and approval chart - EMERGO

Taiwan medical device registration and approval chart - EMERGO
More from V E R A
More from V E R A (7)
Your next RIM solution - 3 key considerations for your move

Your next RIM solution - 3 key considerations for your move
The IDMP Challenge - Whitepaper on ISO IDMP by Cunesoft

The IDMP Challenge - Whitepaper on ISO IDMP by Cunesoft
Regulatory Information Management - Everybody deserves the Truth

Regulatory Information Management - Everybody deserves the Truth
Recently uploaded
Recently uploaded (20)
Top Rated Bangalore Call Girls Mg Road ⟟ 9332606886 ⟟ Call Me For Genuine S...

Top Rated Bangalore Call Girls Mg Road ⟟ 9332606886 ⟟ Call Me For Genuine S...
Call Girls Visakhapatnam Just Call 8250077686 Top Class Call Girl Service Ava...

Call Girls Visakhapatnam Just Call 8250077686 Top Class Call Girl Service Ava...
Call Girls Kochi Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Kochi Just Call 8250077686 Top Class Call Girl Service Available
Call Girls Cuttack Just Call 9907093804 Top Class Call Girl Service Available

Call Girls Cuttack Just Call 9907093804 Top Class Call Girl Service Available
Mumbai ] (Call Girls) in Mumbai 10k @ I'm VIP Independent Escorts Girls 98333...![Mumbai ] (Call Girls) in Mumbai 10k @ I'm VIP Independent Escorts Girls 98333...](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Mumbai ] (Call Girls) in Mumbai 10k @ I'm VIP Independent Escorts Girls 98333...](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
Mumbai ] (Call Girls) in Mumbai 10k @ I'm VIP Independent Escorts Girls 98333...
Call Girls Aurangabad Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Aurangabad Just Call 8250077686 Top Class Call Girl Service Available
Premium Bangalore Call Girls Jigani Dail 6378878445 Escort Service For Hot Ma...

Premium Bangalore Call Girls Jigani Dail 6378878445 Escort Service For Hot Ma...
Call Girls Guntur Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Guntur Just Call 8250077686 Top Class Call Girl Service Available
Call Girls Jabalpur Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Jabalpur Just Call 8250077686 Top Class Call Girl Service Available
Night 7k to 12k Chennai City Center Call Girls 👉👉 7427069034⭐⭐ 100% Genuine E...

Night 7k to 12k Chennai City Center Call Girls 👉👉 7427069034⭐⭐ 100% Genuine E...
Best Rate (Hyderabad) Call Girls Jahanuma ⟟ 8250192130 ⟟ High Class Call Girl...

Best Rate (Hyderabad) Call Girls Jahanuma ⟟ 8250192130 ⟟ High Class Call Girl...
Best Rate (Guwahati ) Call Girls Guwahati ⟟ 8617370543 ⟟ High Class Call Girl...

Best Rate (Guwahati ) Call Girls Guwahati ⟟ 8617370543 ⟟ High Class Call Girl...
Top Rated Bangalore Call Girls Richmond Circle ⟟ 9332606886 ⟟ Call Me For Ge...

Top Rated Bangalore Call Girls Richmond Circle ⟟ 9332606886 ⟟ Call Me For Ge...
College Call Girls in Haridwar 9667172968 Short 4000 Night 10000 Best call gi...

College Call Girls in Haridwar 9667172968 Short 4000 Night 10000 Best call gi...
(Low Rate RASHMI ) Rate Of Call Girls Jaipur ❣ 8445551418 ❣ Elite Models & Ce...

(Low Rate RASHMI ) Rate Of Call Girls Jaipur ❣ 8445551418 ❣ Elite Models & Ce...
All Time Service Available Call Girls Marine Drive 📳 9820252231 For 18+ VIP C...

All Time Service Available Call Girls Marine Drive 📳 9820252231 For 18+ VIP C...
Call Girls Dehradun Just Call 9907093804 Top Class Call Girl Service Available

Call Girls Dehradun Just Call 9907093804 Top Class Call Girl Service Available
Call Girls Faridabad Just Call 9907093804 Top Class Call Girl Service Available

Call Girls Faridabad Just Call 9907093804 Top Class Call Girl Service Available
Call Girls Agra Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Agra Just Call 8250077686 Top Class Call Girl Service Available
Call Girls Ooty Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Ooty Just Call 8250077686 Top Class Call Girl Service Available
DMF - Are You Ready?
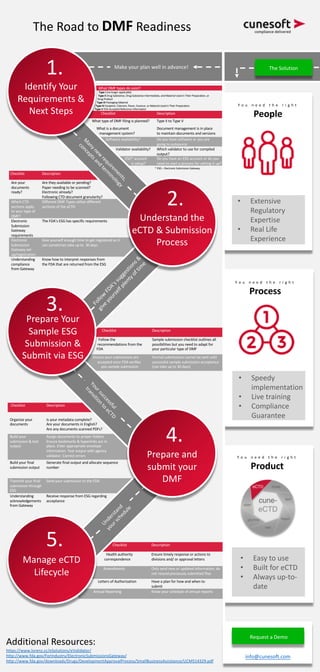
- 1. Checklist Description Health authority correspondence Ensure timely response or actions to divisions and/ or approval letters Amendments Only send new or updated information; do not resend previously submitted files Letters of Authorization Have a plan for how and when to submit Annual Reporting Know your schedule of annual reports Checklist Description Organize your documents Is your metadata complete? Are your documents in English? Are any documents scanned PDFs? Build your submission & test output Assign documents to proper folders Ensure bookmarks & hyperlinks are in place. Enter appropriate envelope information. Test output with agency validator. Correct errors Build your final submission output Generate final output and allocate sequence number Transmit your final submission through ESG Send your submission to the FDA Understanding acknowledgements from Gateway Receive response from ESG regarding acceptance Checklist Description Follow the recommendations from the FDA Sample submission checklist outlines all possibilities but you need to adapt for your particular type of DMF Ensure your submissions are accepted once FDA verifies you sample submission Formal submissions cannot be sent until successful sample submission acceptance (can take up to 30 days) Checklist Description Are your documents ready? Are they available or pending? Paper needing to be scanned? Electronic already? Following CTD document granularity? Which CTD sections apply to your type of DMF? Different DMF Types utilize different sections of the eCTD Electronic Submission Gateway requirements The FDA’s ESG has specific requirements Electronic Submission Gateway set- up/registration Give yourself enough time to get registered as it can sometimes take up to 30 days Understanding compliance from Gateway Know how to interpret responses from the FDA that are returned from the ESG What DMF types do exist? Type I (no longer applicable) Type II Drug Substance, Drug Substance Intermediate, and Material Used in Their Preparation, or Drug Product Type III Packaging Material Type IV Excipient, Colorant, Flavor, Essence, or Material Used in Their Preparation Type V FDA Accepted Reference Information Checklist Description What type of DMF filing is planned? Type II to Type V What is a document management system? Document management is in place to maintain documents and versions Software availability? Do you have software or you are going to outsource Validator availability? Which validator to use for compiled output? ESG* account is setup? Do you have an ESG account or do you need to start a process for setting it up? The Road to DMF Readiness Make your plan well in advance! M anynew requirem ents, conceptsand term inology Follow FDA’ssuggestions& giveyourselfplentyoftim e Yoursuccessful transition to eCTD Understand your schedule Identify Your Requirements & Next Steps 1. Understand the eCTD & Submission Process 2. Prepare and submit your DMF 4. Manage eCTD Lifecycle 5. Prepare Your Sample ESG Submission & Submit via ESG 3. Additional Resources: https://www.lorenz.cc/eSolutions/eValidator/ http://www.fda.gov/ForIndustry/ElectronicSubmissionsGateway/ http://www.fda.gov/downloads/Drugs/DevelopmentApprovalProcess/SmallBusinessAssistance/UCM514329.pdf Y o u n e e d t h e r i g h t People • Extensive Regulatory Expertise • Real Life Experience • Speedy implementation • Live training • Compliance Guarantee • Easy to use • Built for eCTD • Always up-to- date Request a Demo info@cunesoft.com The Solution * ESG – Electronic Submission Gateway Y o u n e e d t h e r i g h t Process Y o u n e e d t h e r i g h t Product