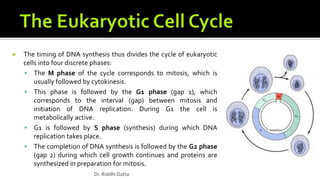
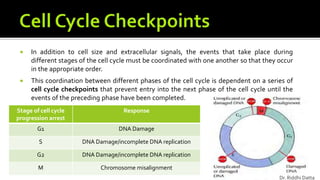
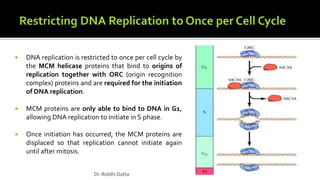
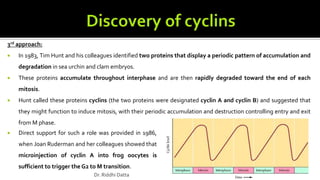
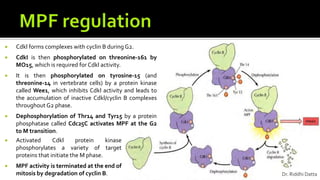
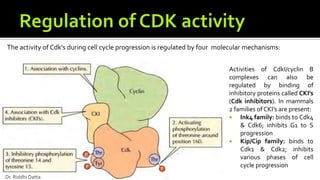
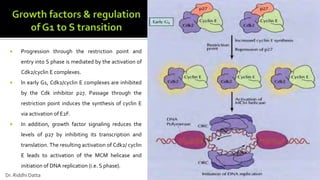
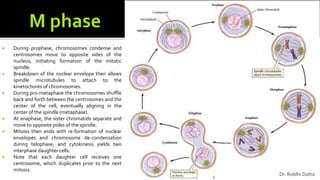
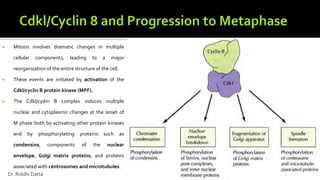
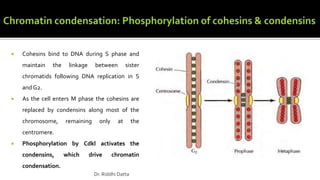
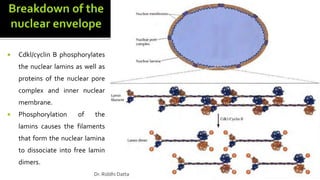
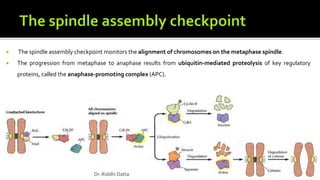
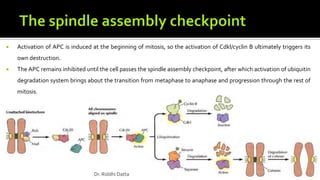
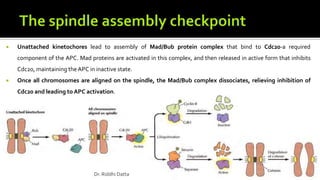
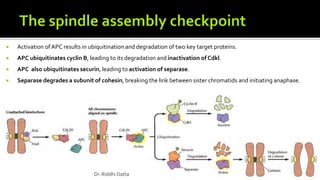
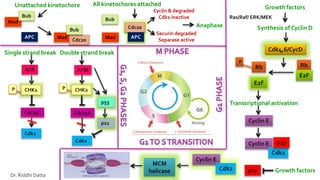
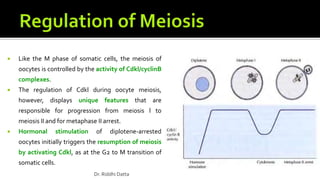
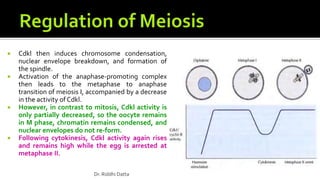
The document provides an extensive overview of the eukaryotic cell cycle, detailing its structure, phases (including G1, S, G2, and M), and regulatory mechanisms. It emphasizes how cell cycle progression is influenced by intracellular and extracellular signals, checkpoints, and the roles of various proteins including cyclins, CDKs, and tumor suppressors. Additionally, it highlights the implications of cell cycle dysregulation in cancer development.